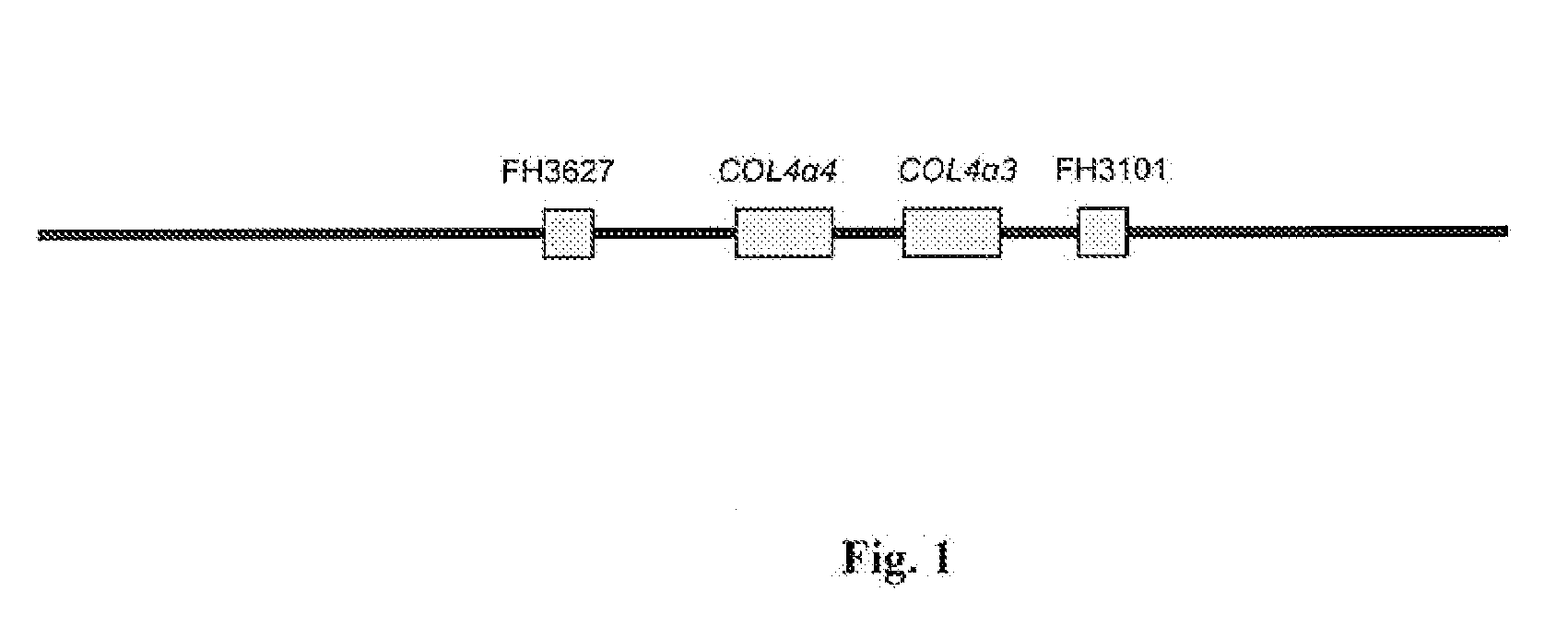
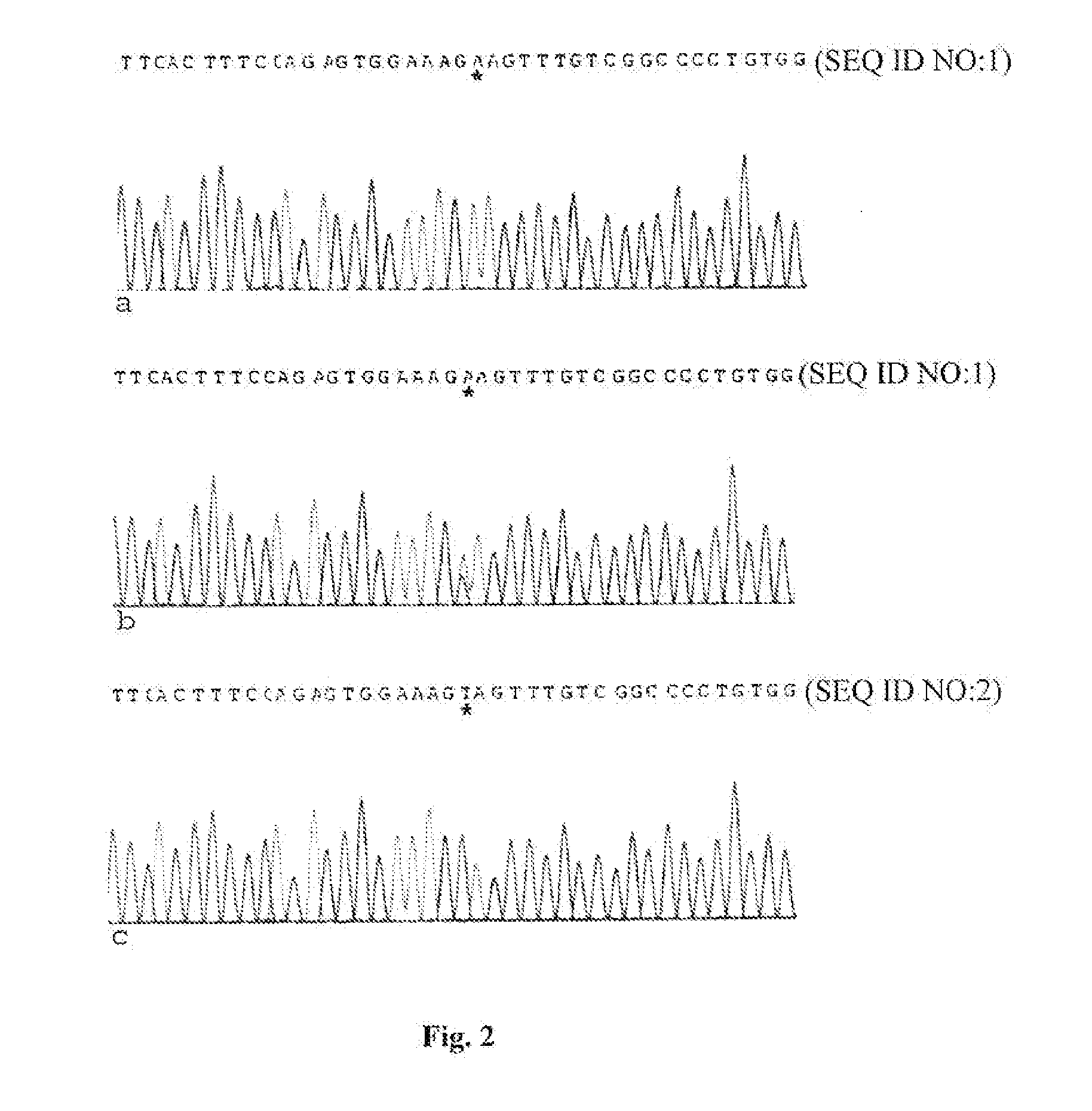
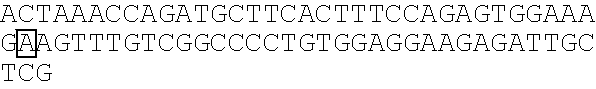Methods for identification of alport syndrome
a technology of alport syndrome and methods, applied in the field of gene testing in dogs, can solve the problems of improper assembly and non-identification of causal mutations in these genes
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
Definitions
[0012]“Genetic marker” or “marker” as used herein refers to a variable or polymorphic nucleotide sequence that is present in genomic DNA, and which is identifiable with specific oligonucleotides (e.g., distinguishable by nucleic acid amplification and observance of a difference in size or sequence of nucleotides due to the polymorphism). The “locus” of a genetic marker or marker refers to its place on the chromosome in relation to another locus. Markers, as illustrated herein, can be identified by any one of several techniques know to those skilled in the art, including microsatellite or short tandem repeat (STR) amplification, analyses of restriction fragment length polymorphisms (RFLP), single nucleotide polymorphism (SNP), detection of deletion or insertion sites, and random amplified polymorphic DNA (RAPD) analysis.
[0013]“Co-segregate” as used herein refers to inheritance together of two specific loci; e.g., the loci are located so physically close on the same chro...
PUM
| Property | Measurement | Unit |
|---|---|---|
| structure | aaaaa | aaaaa |
| nucleic acid amplification | aaaaa | aaaaa |
| size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More