Obesity and Metabolic Syndrome Treatment with Tanshinone Derivatives Which Increase Metabolic Activity
a technology of tanshinone and metabolic syndrome, which is applied in the direction of pill delivery, plant/algae/fungi/lichens ingredients, dispersion delivery, etc., can solve the problems of metabolic syndrome, inability of cells to perform normal functions, and insufficient glucose supply of insulin to cells, so as to prevent and treat metabolic syndrome and activate metabolism. , the effect of
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Isolation of Tanshinone Derivatives
[0094] 5 kg of Danshen (Salvia miltiorrhiza) material was purchased from a Chinese medicinal herb shop and other necessary materials were collected in fields and mountains or were purchased from the shop. Danshen was eluted with 50 L of methanol for 24 hours and concentrated under reduced pressure. 1500 mL of water was added to the resulting material. Then, an equal amount of n-hexane, dichloromethane (CH2Cl2) and ethyl acetate (EtOAc) were added and sequentially extracted two times so as to obtain a gelatinous red extract. When activity was examined on the respective layers thus obtained, the activity was highest in the dichloromethane layer.
[0095] Silica gel (Kieselgel 60, 230 to 460 mesh, Merck) was sufficiently swelled with 100% n-hexane and then packed into a column (530 cm high). 50 g of the extract obtained from the CH2Cl2 layer was dissolved in a trace amount of EtOAc and n-hexane and the resulting sample was loaded onto the column. After...
example 2
Structural Analysis of Separated Active Material
[0098] NMR analysis was performed to determine structures of cryptotanshinone, tanshinone I, tanshinone IIA and 15,16-dihydrotanshinone I separated in Example 1, respectively.
Cryptotanshinone
[0099] 1H-NMR (CDCl3): δ 7.42 (2H, ABq, J=8.0 Hz), 4.83 (1H, t, J=9.2 Hz), 4.31 (1H, dd, J=9.2 and 6.0 Hz), 3.55 (1H, m), 3.17 (2H, br t), 1.65 (4H, m), 1.40 (3H, d, J=6.8 Hz), 1.28 (6H, s)
[0100] 13C-NMR (CDCl3): δ 9.58 (C-1), 19.00 (C-2), 37.73 (C-3), 34.76 (C-4), 143.57 (C-5), 132.48 (C-6), 122.43 (C-7), 128.30 (C-8), 126.19 (C-9), 152.28 (C-10), 184.16 (C-11), 175.59 (C-12), 118.21 (C-13), 170.66 (C-14), 81.38 (C-15), 34.54 (C-16), 18.74 (C-17), 31.85 (C-18), 31.80 (C-19)
Tanshinone II-A
[0101] 1H-NMR (CDCl3, 300.40 MHz) δ 7.63 (1H, d, J=8.2 Hz), 7.54 (1H, d, J=8.2 Hz), 7.22 (1H, s), 3.18 (2H, t, J=6.6 Hz), 2.26 (3H, s), 1.78 (2H, m), 1.65 (2H, m), 1.31 (6H, s).
[0102] 13C-NMR (CDCl3, 75.45 MHz) δ 184.29, 176.43, 162.38, 150.80, 145.14, 14...
example 3
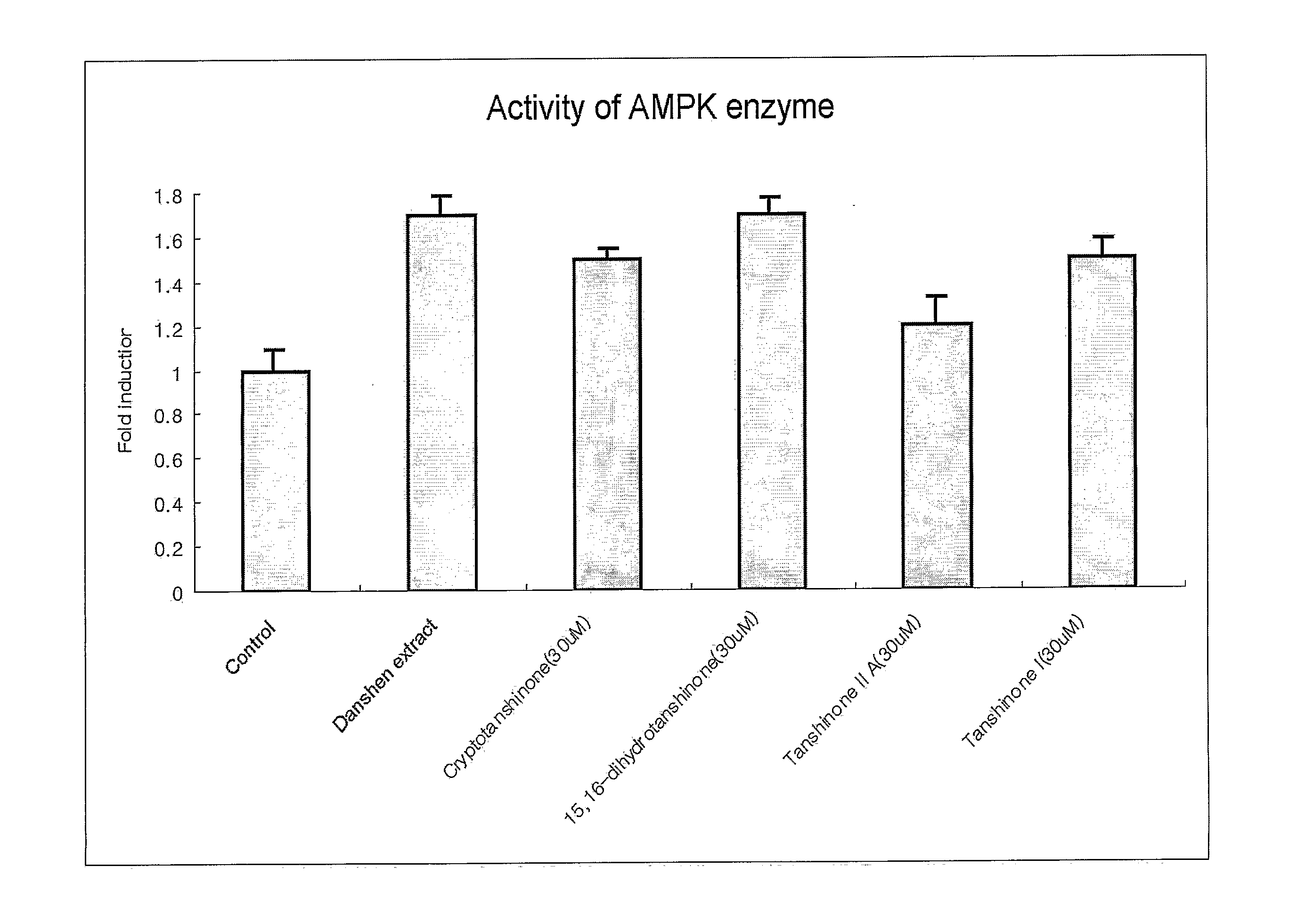
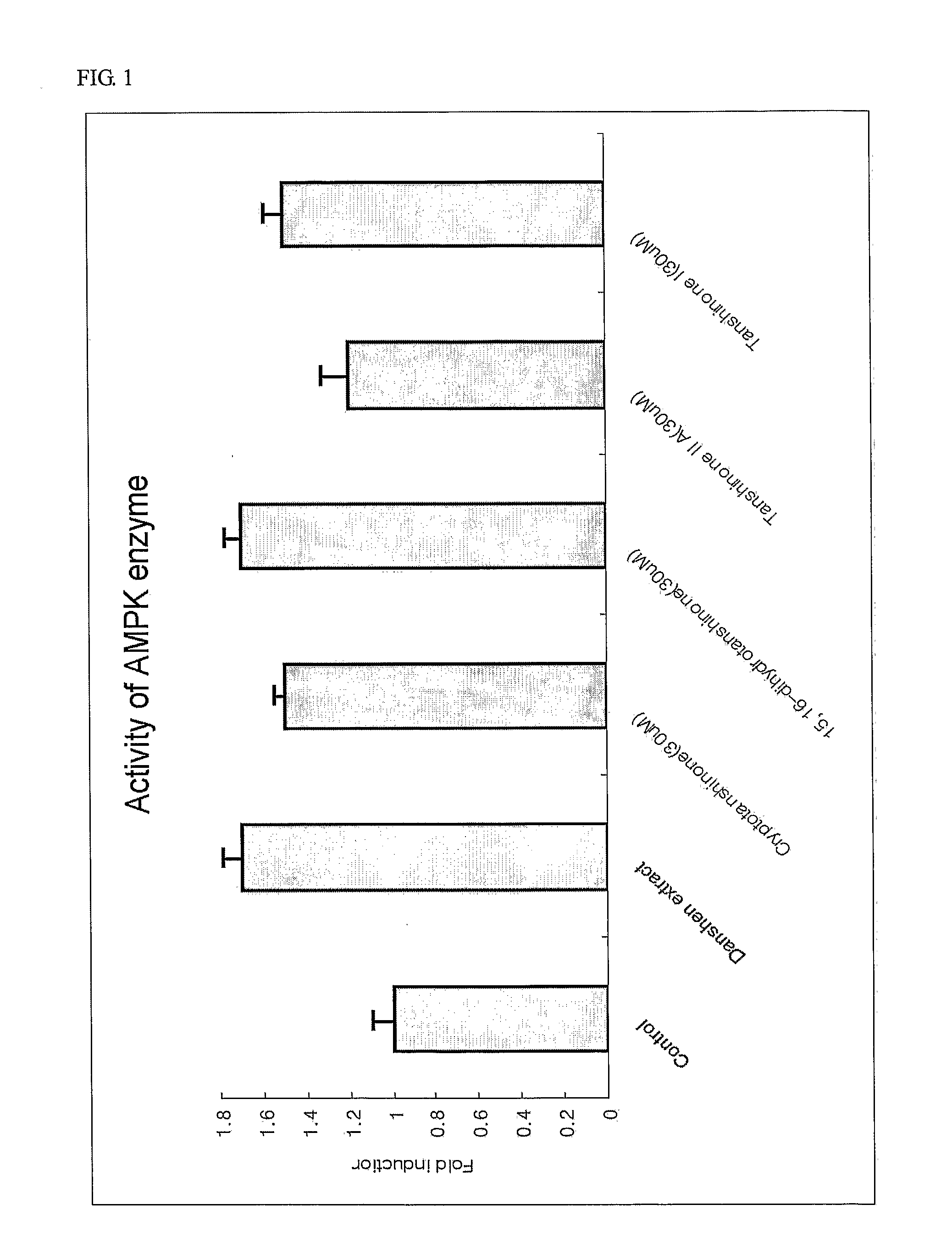
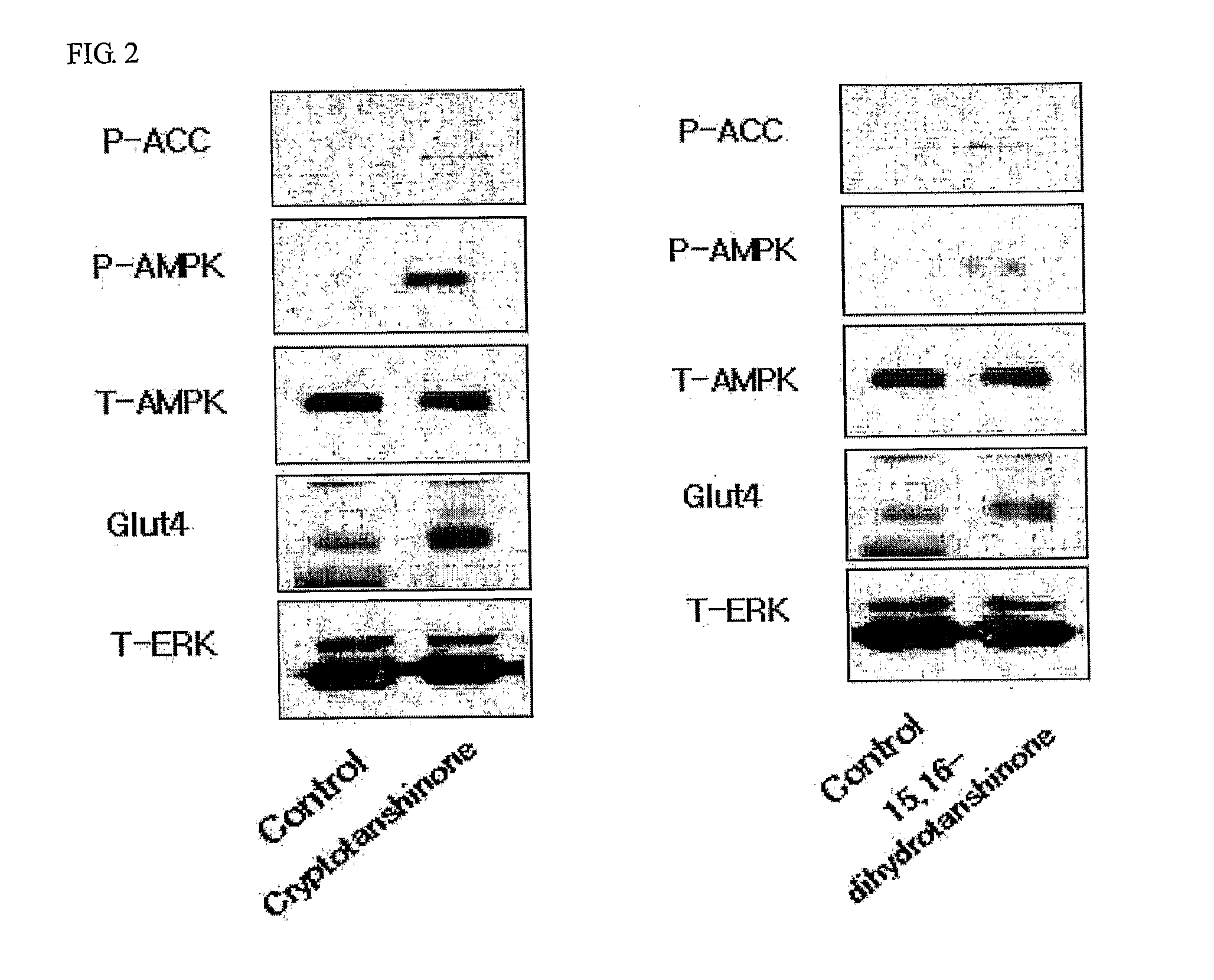
Determination of AMPK Activity
[0107] Myoblast cells, C2C12, were cell cultured in DMEM containing 10% bovine calf serum. When cell density reached a range of about 85% to 90%, the culture medium was replaced with 1% bovine calf serum medium to induce differentiation of cells. Enzymatic activity of AMPK was determined as follows. C2C12 cells were lysed to obtain protein extracts and then ammonium sulfate was added to a final concentration of 30%, followed by precipitation of proteins. Protein precipitates were dissolved in a buffer (62.5 mM Hepes, pH 7.2, 62.5 mM NaCl, 62.5 mM NaF, 1.25 mM Na pyrophosphate, 1.25 mM EDTA, 1 mM DTT, 0.1 mM PMSF, and 200 μM AMP). Thereafter, 200 μM SAMS peptide (HMRSAMSGLHLVKRR: the underlined serine residue is a phosphorylation site, as an AMPK phosphorylation site of acetyl-CoA carboxylase) and [γ-32P]ATP were added thereto and reactants were reacted for 10 minutes at 30° C. This was followed by spotting of the resulting reaction solution on p81 phos...
PUM
| Property | Measurement | Unit |
|---|---|---|
| density | aaaaa | aaaaa |
| density | aaaaa | aaaaa |
| density | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More