Anti-cd70 antibody-drug conjugates and their use for the treatment of cancer and immune disorders
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Sequence Analysis of Anti-CD70 Monoclonal Antibodies 1F6 and 2F2
[0297] To determine the cDNA sequences encoding the light (VL) and heavy (VH) chain variable regions of 1F6 and 2F2 mAb, total RNA was isolated from the 1F6 and 2F2 hybridomas using the TRIzol® Reagent (Invitrogen, Carlsbad, Calif.) according to the manufacturer's instructions. The gene-specific primers mIgcK1 5′-CTT CCA CTT GAC ATT GAT GTC TTT G-3′ (SEQ ID NO:41) and mIgG1 5′-CAG GTC ACT GTC ACT GGC TCA G-3′ (SEQ ID NO:42) were applied to reverse transcribe the light chain variable (VL) and heavy chain variable (VH) first strand cDNAs from both RNA preparations, respectively. First strand cDNA reactions were run using the SuperScript™ First Strand Synthesis System for RT-PCR from Invitrogen (Carlsbad, Calif.). The VL and VH cDNAs were then poly-G tailed using terminal deoxynucleotidyl transferase (TdT) (Invitrogen) and the supplied TdT buffer in conditions specified by the manufacturer. Poly-G tailed VL and VH first s...
example 2
Synthesis of Anti-CD70 Drug Conjugates
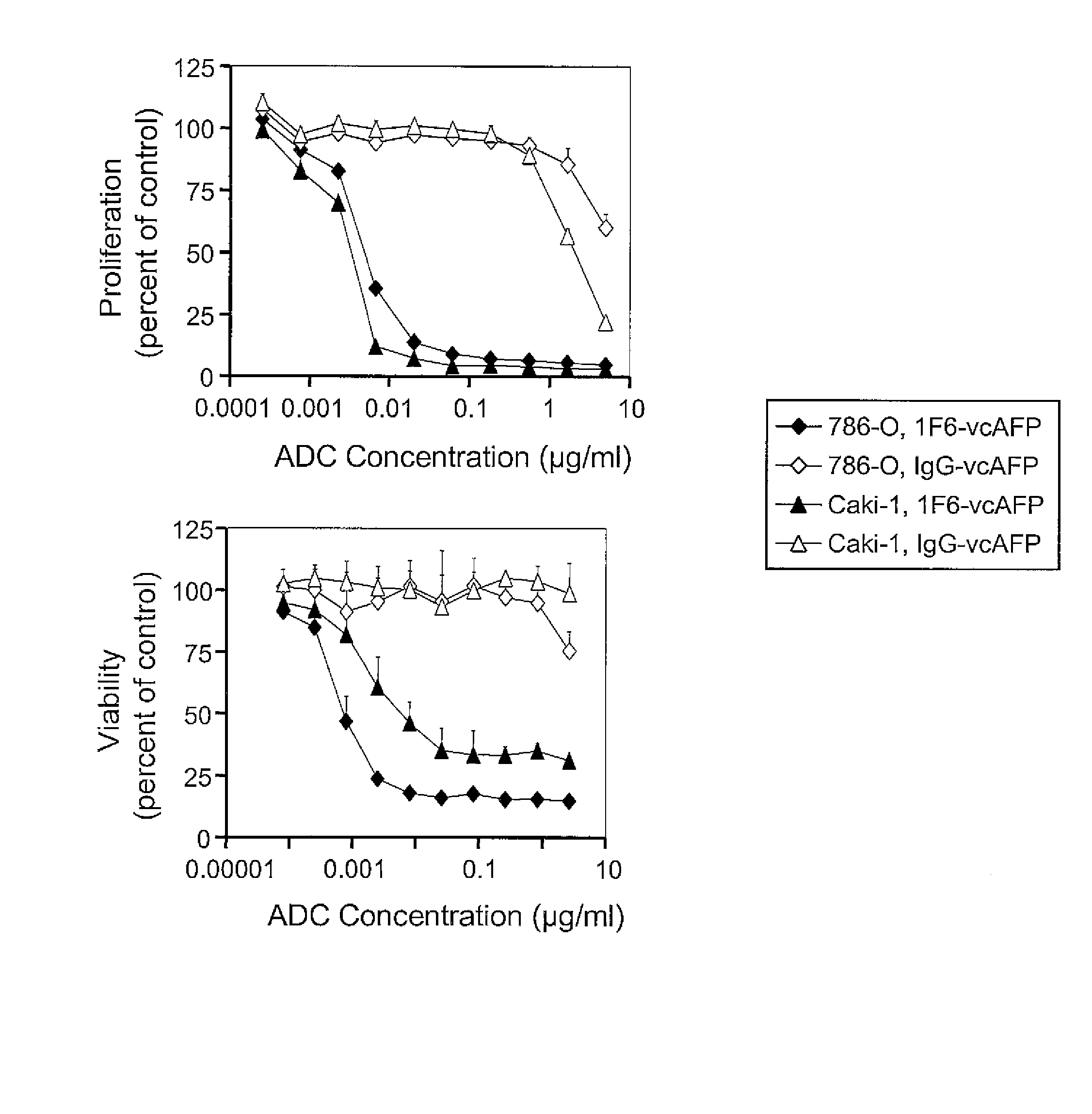
[0299] The ability of anti-CD70 to deliver a potent cytotoxic drug in the form of an antibody drug conjugate (ADC) to eliminate CD70-expressing cells was tested. Monomethyl auristatin E (MMAE), auristatin phenylalanine phenylenediamine (AFP), and monomethyl auristatin phenylalanine (MMAF) were used as the targeted cytotoxic drugs for this study. The drugs were linked to the anti-CD70 mAb 1F6 by the valine-citruline (vc) dipeptide linker to give the 1F6-vcMMAE, 1F6-vcAFP conjugates and 1F6-vcMMAF.
[0300] A. Synthesis of F6-vcMMAE
[0301] The synthesis of the ADC 1F6-vcMMAE, the general structure of which is depicted above, is described below.
[0302] 1. Drug-Linker Compound Synthesis
[0303] Synthesis of auristatin E has been previously described (U.S. Pat. No. 5,635,483; Pettit, 1999, Prog. Chem. Org. Nat. Prod. 70:1-79). The monomethyl derivative of Auristatin E (MMAE) was prepared by replacing a protected form of monomethylvaline for N,N-dimet...
example 3
Expression of CD70 on Hematologic Cell Lines
[0338] Surface expression of CD70 was examined using flow cytometry. In general, 0.2×106 cells were incubated with 50 μl of staining medium (RPMI-1640 supplemented with 5-10% FBS) containing a fluorochrome-conjugated mAb (10 μg / ml) for single-color flow cytometry or a cocktail of fluorochrome-conjugated mAbs for multiple-color flow cytometry. Incubations were carried out on ice for 20-30 minutes. Cells were then washed 3 times with the staining buffer and fixed in PBS containing 1% of paraformaldehyde. Flow cytometric analysis was performed with a FACScan (BD Immunocytometry, San Jose, Calif.) and data were analyzed by either the CellQuest (BD Immunocytometry) or the WinMDI software. The anti-human CD70 monoclonal antibodies (mAb) 1F6 and 2F2 were obtained from the Central Laboratory of the Netherlands Red Cross Blood Transfusion Service (Amsterdam, The Netherlands). 1F6 was conjugated to AlexaFluor488 (AF) (Molecular Probes, Eugene, Oreg...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Time | aaaaa | aaaaa |
| Dimensionless property | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More