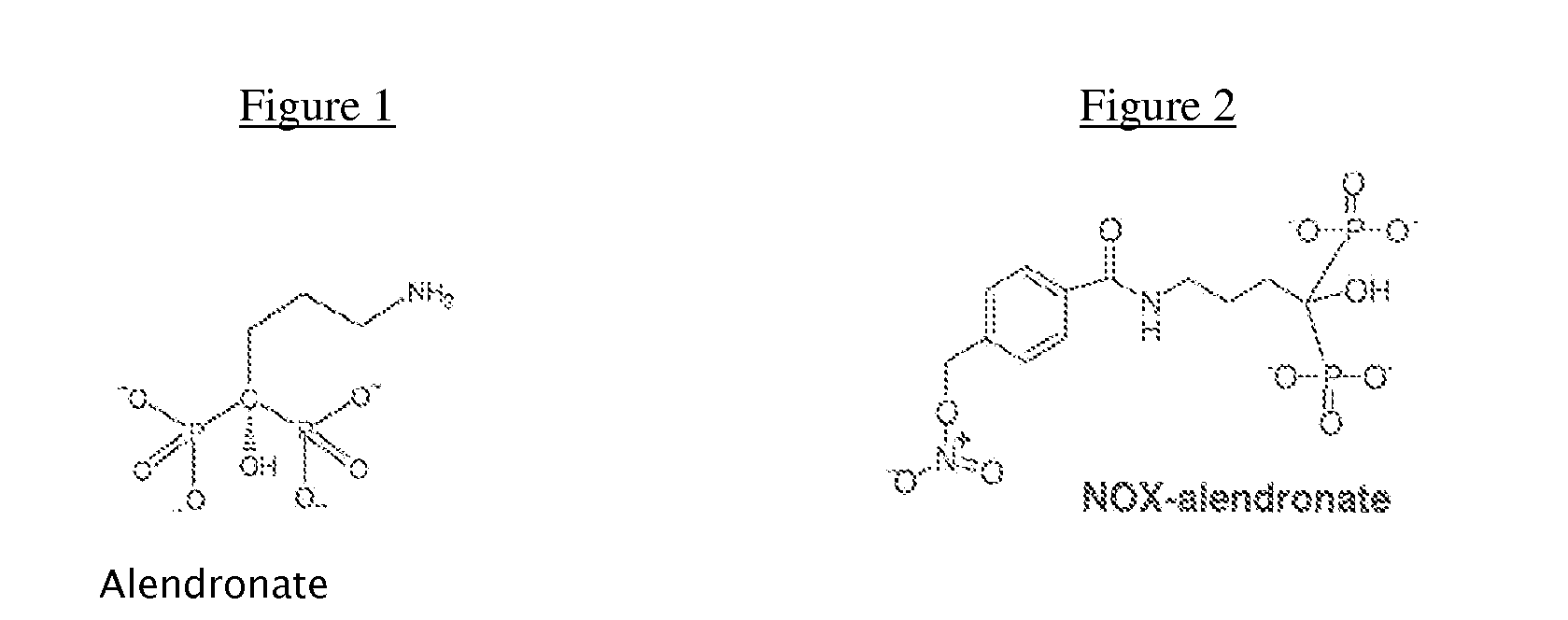
Methods and compositions for stimulating bone growth using nitric oxide releasing bisphosphonate conjugates (no-bisphosphonate)
a technology of nitric oxide and bisphosphonate, which is applied in the direction of phosphorous compound active ingredients, drug compositions, biocides, etc., can solve the problems of reducing bone growth, reducing bone density, and reducing bone density, so as to promote bone growth, and enhance bone formation or bone mineral density
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
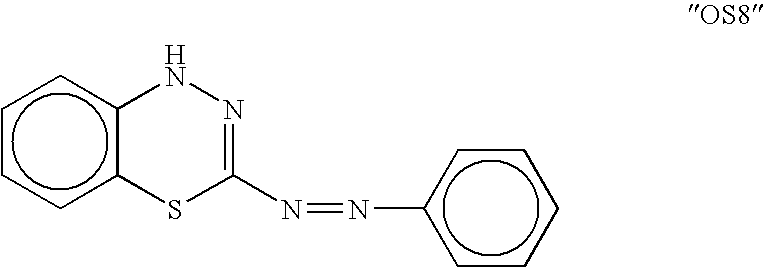
[0076] In this screen, the standard positive control was the compound 59-0008 (also denoted “OS8”), which is of the formula:
[0077] In more detail, the 2T3-BMP-2-LUC cells, a stably transformed osteoblast cell line described in Ghosh-Choudhury et al., Endocrinology (1996) 137:331-39, referenced above, was employed. The cells were cultured using α-MEM, 10% FCS with 1% penicillin / streptomycin and 1% glutamine (“plating medium”), and were split 1:5 once per week. For the assay, the cells were resuspended in a plating medium containing 4% FCS, plated in microtiter plates at a concentration of 5×103 cells (in 50 μl) / well, and incubated for 24 hours at 37° C. in 5% CO2. To initiate the assay, 50 μl of the test compound or the control in DMSO was added at 2× concentration to each well, so that the final volume was 100 μl. The final serum concentration was 2% FCS, and the final DMSO concentration was 1%. Compound 59-0008 (10 μM) was used as a positive control.
[00...
example 2
[0079] Selected compounds and appropriate controls were assayed in vitro (ex vivo) for bone formation activity (described above in “Techniques for Neonatal Mouse Calvaria Assay (in vitro)). Histomorphometrical assessments of ex vivo calvaria were carried out using an OsteoMetrics bone morphometry measurement program, according to the manufacturer's instructions. Measurements were determined using either a 10- or 20-fold objective with a standard point counting eyepiece graticule.
[0080] New bone formation was determined (using a 20× objective) by measuring the new bone area formed in one field in 3 representative sections of each bone (4 bones per group). Each measurement was carried out ½ field distance from the end of the suture. Both total bone and old bone area were measured. Data were expressed as new bone width in mm.
[0081] Osteoblast numbers are determined by point counting. The number of osteoblast cells lining the bone surface on both sides of the b...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More