Polymer-based sustained release device
a technology of sustained release and polymer, which is applied in the field of polypeptides, can solve the problems of poor patient compliance, fluctuating levels of medicaments, serum drug levels outside the therapeutic window, etc., and achieves excellent release profile, minimized loss of activity, and improved bioavailability of incorporated drugs.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
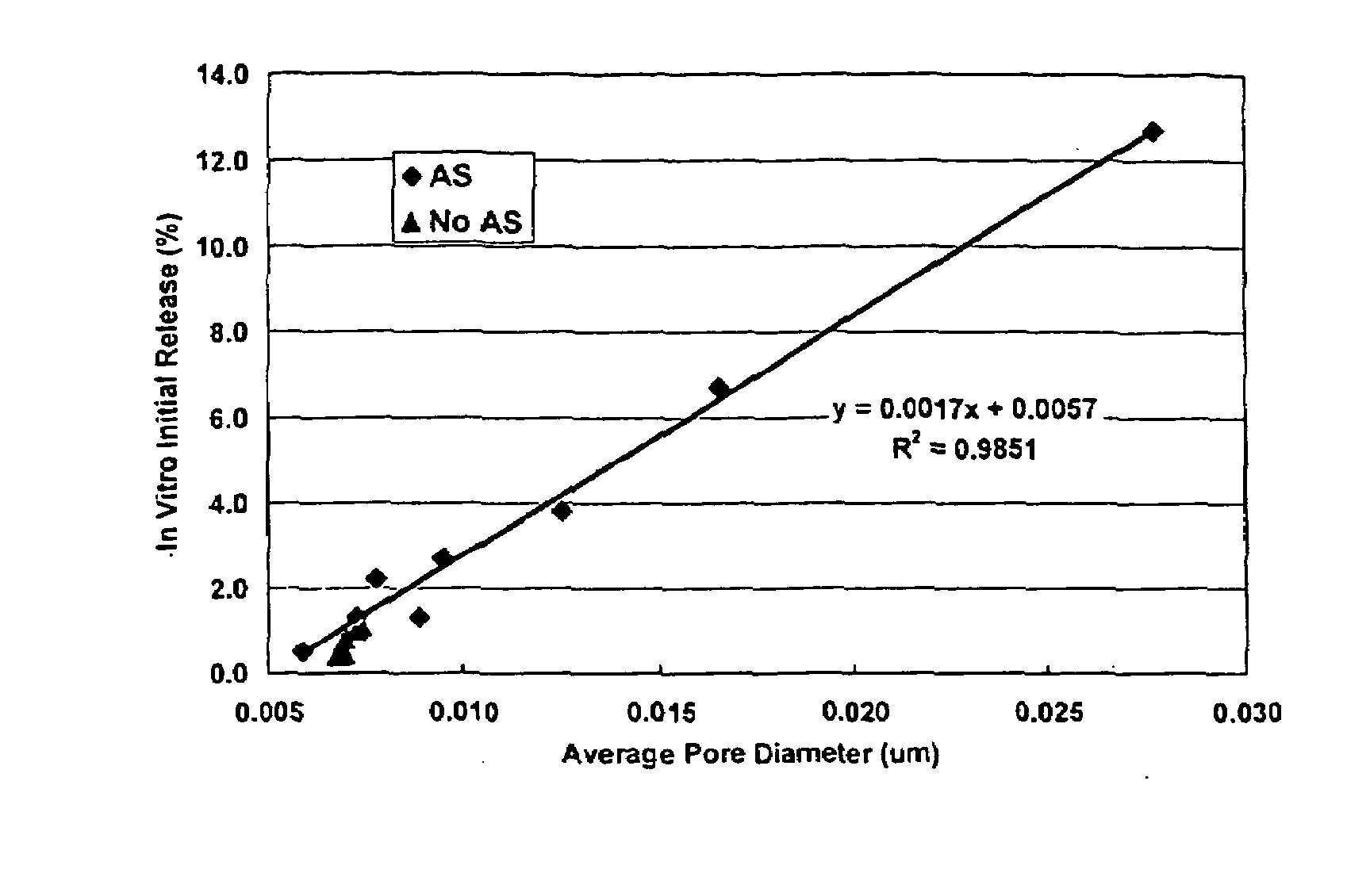
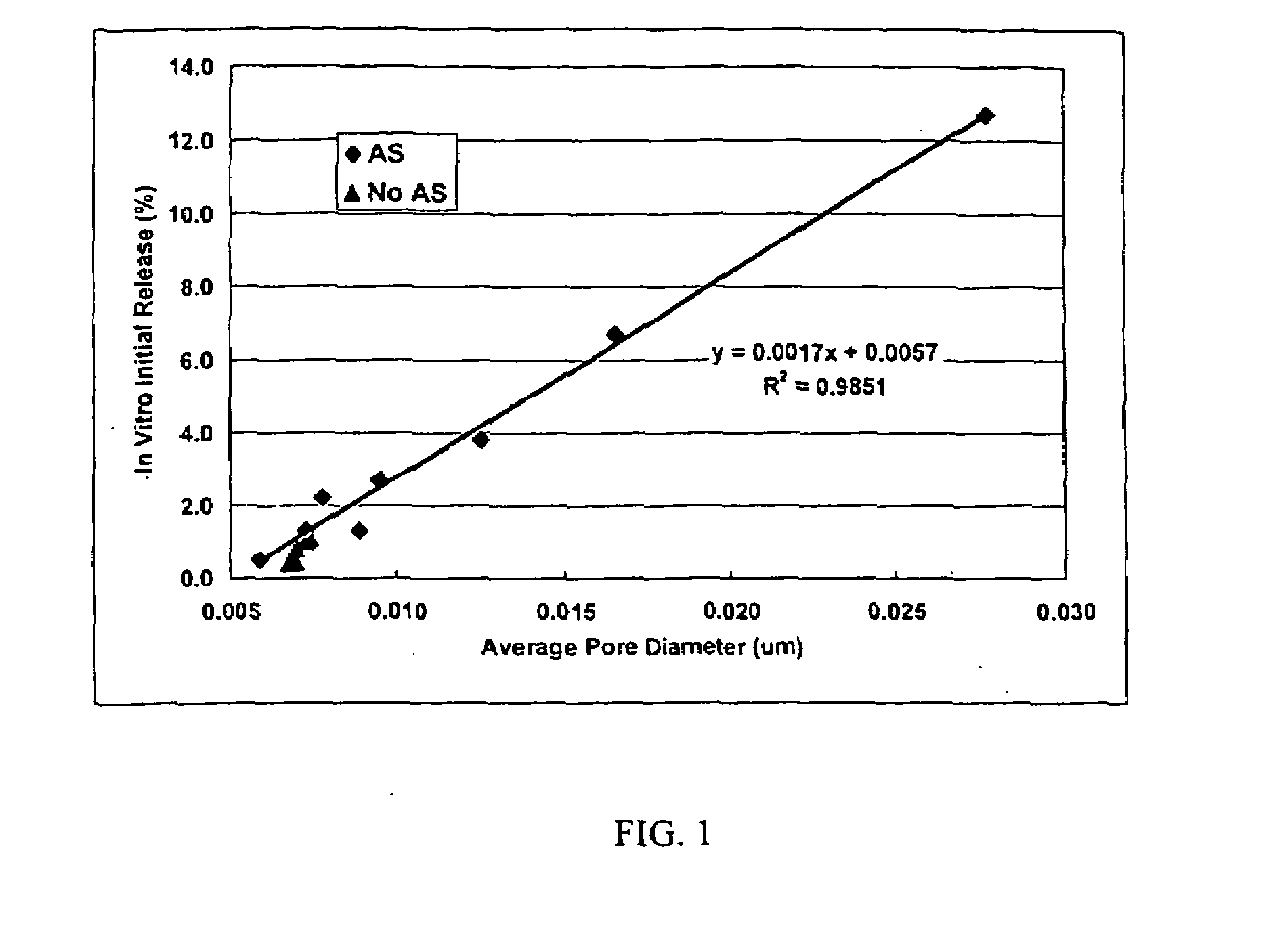
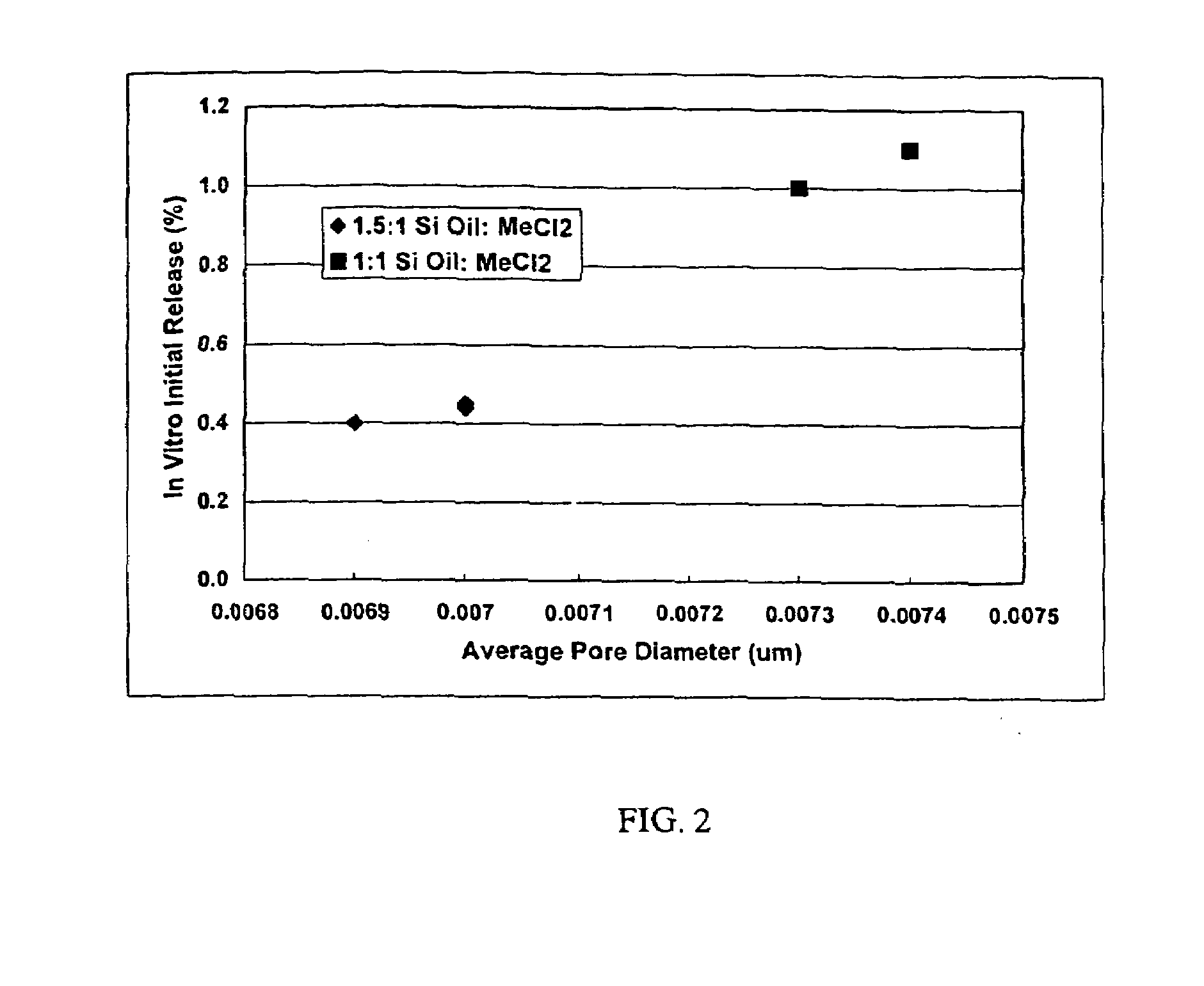
[0024]This invention relates to compositions for the sustained release of biologically active polypeptides, and methods of forming and using said compositions, for the sustained release of biologically active polypeptides. The sustained release compositions of this invention comprise a biocompatible polymer, and agent, such as a biologically active polypeptide, and a sugar. The agent and sugar are dispersed in the biocompatible polymer separately or, preferably, together. In a particular embodiment, the sustained release composition is characterized by a release profile having a ratio of maximum serum concentration (Cmax) to average serum concentration (Cave) of about 3 or less. As used herein, the terms a or an refer to one or more.
[0025]The Agent
[0026]In a preferred embodiment, the agent is a biologically active polypeptide such as an antidiabetic or glucoregulatory polypeptide, including GLP-1, GLP-2, exendin-3, exendin-4 or an analog, derivative or agonist thereof. Most specific...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
| molecular weight | aaaaa | aaaaa |
| molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More