A primary byproduct is water, but conventional fuel
cell designs inevitably yield several more undesired byproducts such as
carbon monoxide,
sulfur, and the like, particularly when employing a carbon-based fuel.
The current design as exemplified by
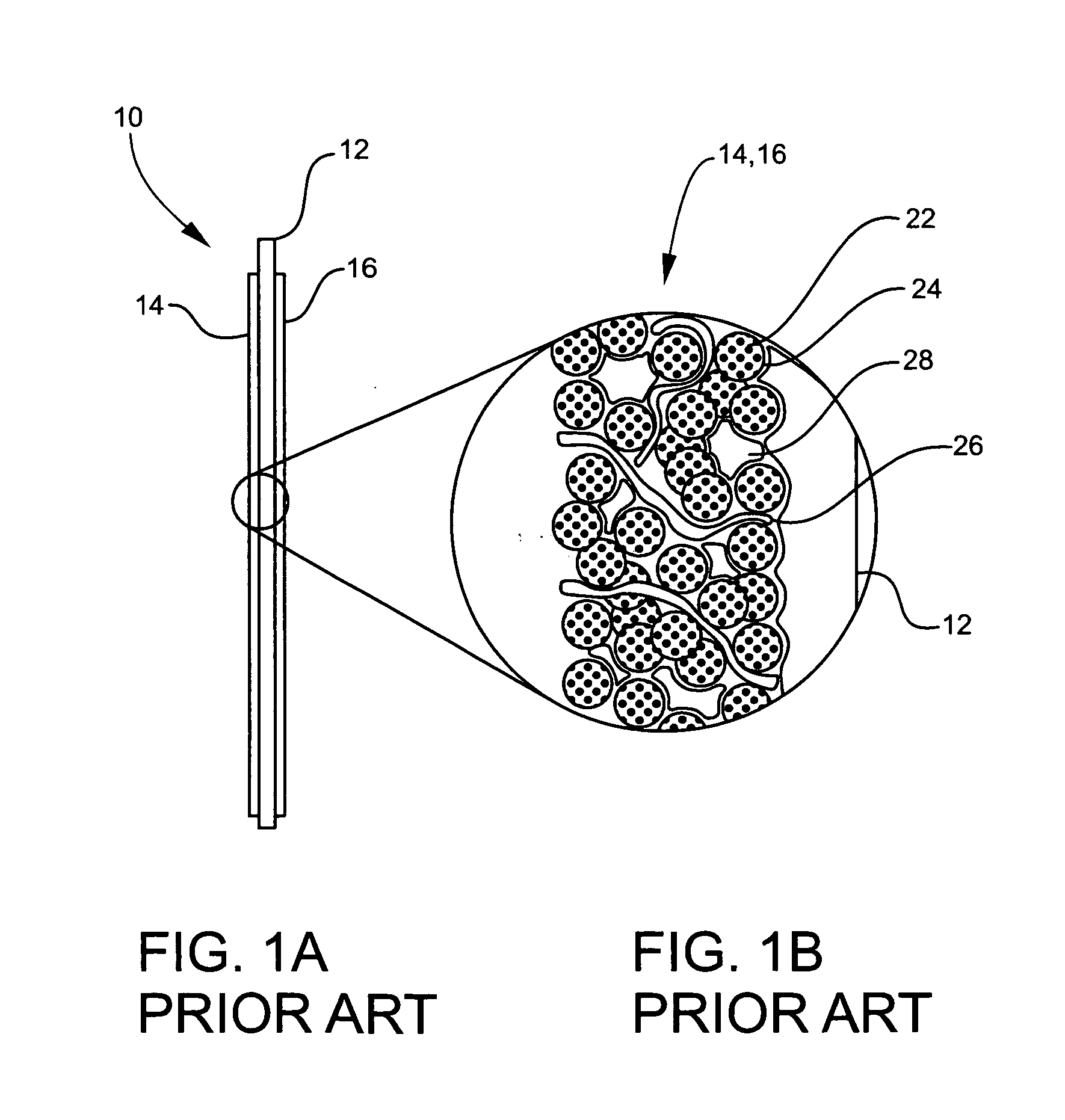
membrane electrode assembly 10 illustrated in FIGS. 1A and 1B is functional and an improvement over earlier designs, but it is still embodies significant limitations.
It follows that only about ⅙th or 17% of the
platinum is potentially available as a catalyst, such that roughly 83% of the
mass is wasted.
The degree of waste is much worse in direct
methanol fuel cells, which require larger particles and heavier catalyst loadings.
The remaining portion of the catalyst is potentially wasted, for instance, by being locked inside the particle.
This contact area excludes contact with the reactants and thus further limits the
usable, catalytic surface area.
Additionally, a yet additional loss of catalytic utilization occurs because the reaction can only take place at the triple-interface of the fuel, electrolyte, and catalyst.
Furthermore, those particles that do experience a favorable interface produce or use water, thereby changing the fuel / water ratio in their immediate microenvironment, often decreasing
catalytic efficiency.
Another problem is that the materials and mixtures utilized in the prior art are fairly electrically resistant.
This
internal resistance substantially decreases electrical production efficiency and necessitates the use of conductive current-collecting “field flow” plates that add sizeable cost and volume to the fuel
cell or fuel cell stack.
In addition, the carbon-
platinum mixture utilized in prior art approaches is essentially a brittle composition of dust or a composite of powders.
Over time the mixture tends to disintegrate and thereby limit lifespan and efficiency.
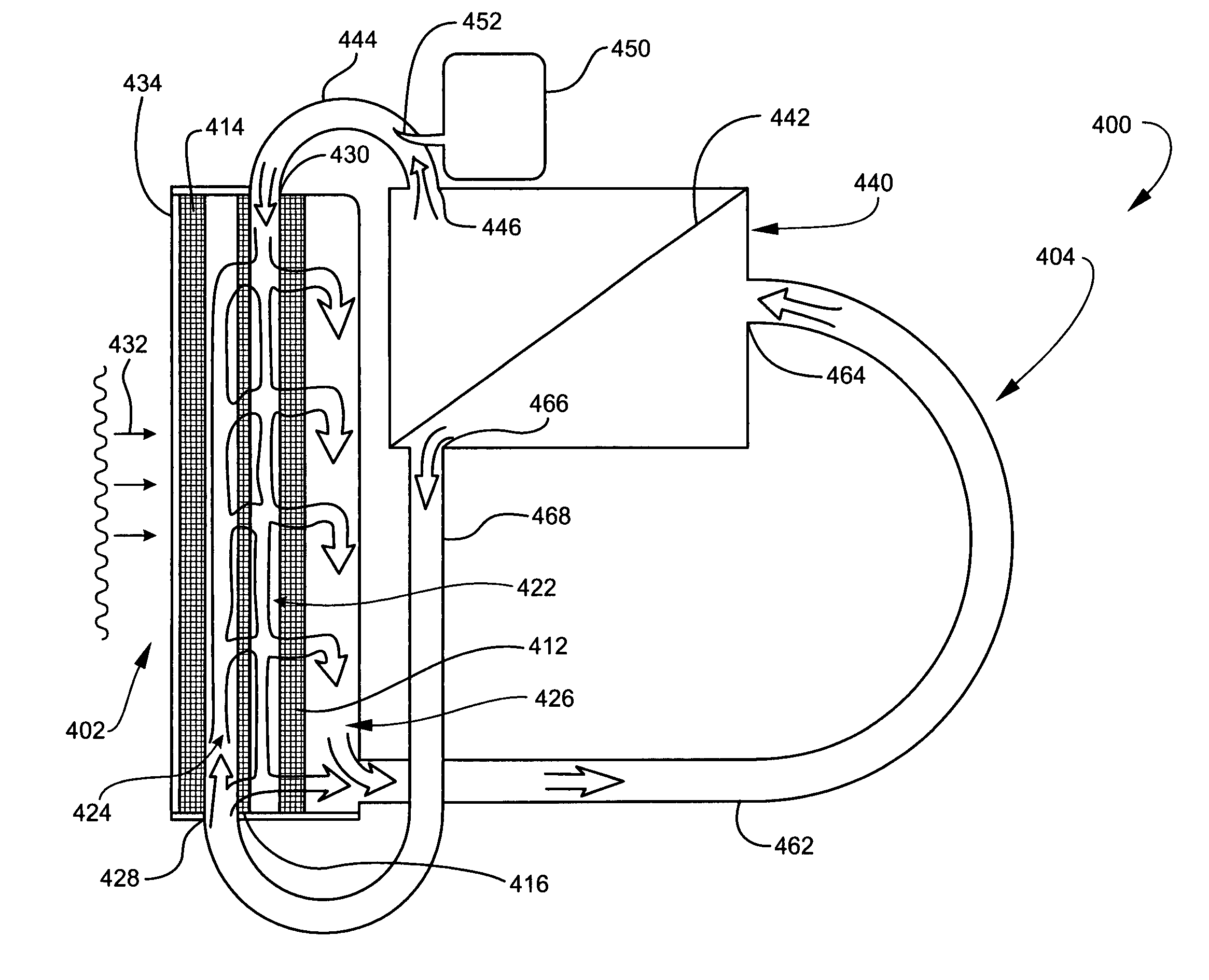
Moreover, in current fuel cell designs, the fuel flows by and diffuses into the
anode but not though it.
Consequently,
inert compounds can build up in the pores and physically block the fuel from reaching the catalyst, hence further limiting efficiency.
Water is produced at one electrode and thus can potentially flood the electrode.
To ameliorate or compensate for these flooding and
drying events, current fuel cell designs must resort to the addition of extensive, costly and power-
robbing balance-of-
plant apparatus.
Much of the prohibitive cost of fuel cell production can be attributed to the balance-of-
plant and not the fuel cell stack itself.
Unfortunately, even with the use of balance-of-
plant apparatus, the simultaneous ideal humidification for each electrode (
anode and
cathode, as well as the electrolyte) is never quite uniformly achieved.
Poisoning and
contamination remain a pervasive problem in many fuel cell designs.
Catalysts suffer from poisoning by common contaminants found in many fuel stocks.
Over time, these substances adhere to the catalytic particles of conventional electrodes, degrading their performance and limiting their lifespan.
Sensitivity to poisoning seriously limits the feasibility and commercial viability of the currently existing technologies.
It is especially a problem in fuel cells that use currently available fossil fuels and
natural gas derivatives.
These fuels have relatively high amounts of
sulfur compounds and complex hydrocarbons that form a variety of toxic intermediary compounds.
To partially atone for this problem, manufacturers are forced to incorporate expensive, additional balance-of-plant apparatus such as those noted above, particularly fuel stock scrubbers, reformers, shift reactors, and advanced filters, all of which can be bulky and / or expensive and escalate inefficiency, maintenance requirements, and
pollution.
The extra equipment also requires energy to run that is parasitically drawn from the output of fuel cell.
 Login to View More
Login to View More