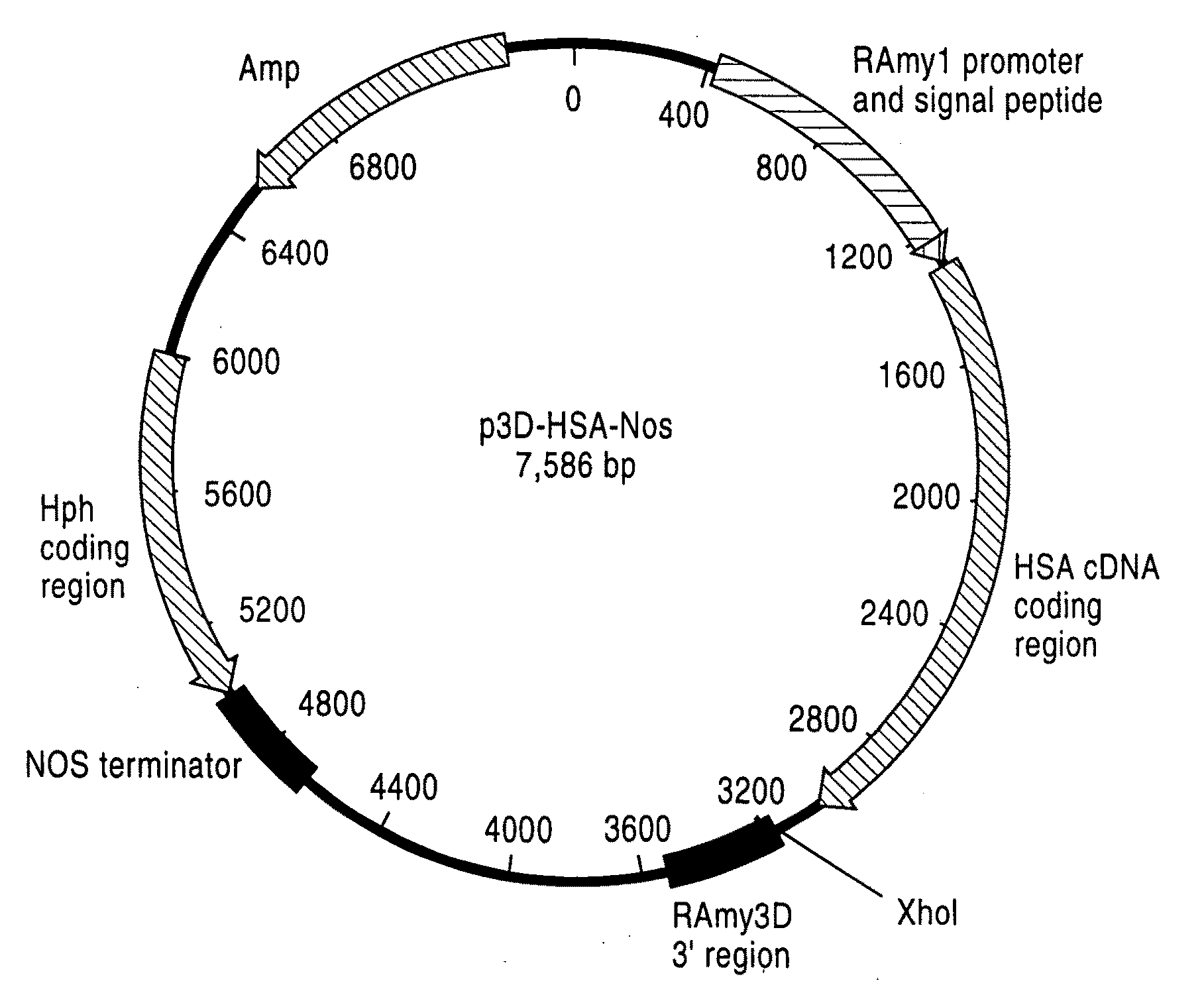
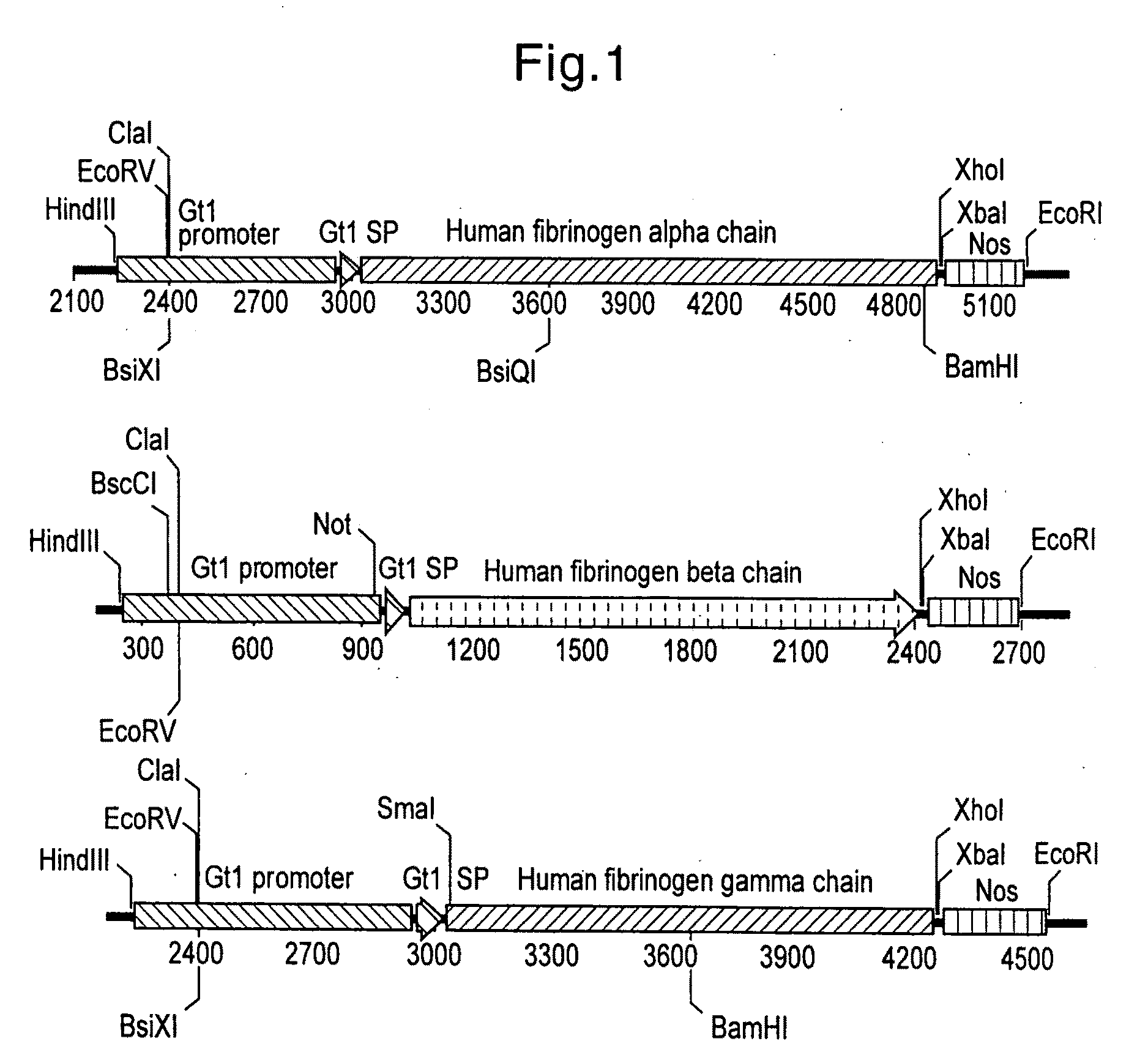
Human Blood Proteins Expressed in Monocot Seeds
a monocot seed and protein technology, applied in the field of human blood proteins produced, can solve the problems of short or limited supply of human blood proteins, and none of these patents or publications discloses the production of human blood proteins in monocot seeds in high yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 1
Production of Transgenic Rice Encoding AAT and Fibrinogen Polypeptides
[0106]The basic procedures of particle bombardment-mediated rice transformation and plant regeneration were carried out as described by Huang et al., 2001. Rice variety TP309 seeds were dehusked, sterilized in 50% (v / v) commercial bleach for 25 min and washed with sterile water. The sterilized seeds were placed on rice callus induction medium (RCI) plates containing [N6 salts (Sigma), B5 vitamins (Sigma), 2 mg / l 2,4-D and 3% sucrose]. The rice seeds were incubated for 10 days to induce callus formation. Primary callus was dissected from the seeds and placed on RCI for 3 weeks. This was done twice more to generate secondary and tertiary callus which was used for bombardment and continued subculture. A callus of 1-4 mm diameter was placed in a 4 cm circle on RCI with 0.3M mannitol 0.3M sorbitol for 5-24 hrs prior to bombardment. Microprojectile bombardment was carried out using the Biolistic PDC-1000 / He system (Bio-...
example 2
Production of Rice Extract Containing Recombinant Blood Proteins and its Use in Parenteral and Enteric Formulations
General Procedure for Production of Rice Extract
[0108]Transgenic rice containing heterologous polypeptides can be converted to rice extracts by either a dry milling or wet milling process. In the dry milling process, transgenic paddy rice seeds containing the heterologous polypeptides were dehusked with a dehusker. The rice was grounded into a fine flour though a dry milling process, for example, in one experiment, at speed 3 of a model 91 Kitchen Mill from K-TEC. Phosphate buffered saline (“PBS”), containing 0.135 N NaCl, 2.7 mM KCl, 10 mM Na2HPO4, 1.7 mM KH2PO4, at pH 7.4, with or without additional NaCl, such as 0.35 N NaCl, was added to the rice flour. In some experiments, approximately 10 ml of extraction buffer was used for each 1 g of flour. In other experiments, the initial flour / buffer ratio varied over a range such as 1 g / 40 ml to 1 g / 10 ml. The mixture was in...
example 3
Concentration and Diafiltration of Recombinant Blood Protein and Control Rice Extracts
[0116]The conditions used in concentration and diafiltration vary depending on volume, speed, cost, etc. These conditions are standard in the art based on the description herein. The frozen initial extract was thawed in the coldroom (about 2-8° C.) for six hours. The thawed material was clarified though a 0.45 μm filter and concentrated using a 5000 Nominal Molecular Weight Cutoff membrane of polyethersulfone.
[0117]90 ml of the filtrate of control extract was concentrated to 10 ml and additional 10 ml of deionized water can be added to the concentrated filtrate. The diluted filtrate can be diafiltrated one more time using water. The precipitate starts forming at 16 mS and increases as the ionic strength decreases. A solution of 1.0M ammonium bicarbonate was added to the retentate to add ionic strength. The haze decreases although does not disappear completely. The material was diafiltered multiple ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More