Drug-loaded polyelectrolyte capsules in response to phosphatase concentration and preparation method thereof
A polyelectrolyte and phosphatase technology, applied in capsule delivery, pharmaceutical formulations, medical preparations of non-active ingredients, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0020] Example 1 Preparation of Polyelectrolyte (PAH / PSS-β-Glycerophosphate) Empty Shell Capsules
[0021] (1), preparation of gelatin microspheres
[0022] Dissolve 0.5g of gelatin in 10ml of water, swell for 10 minutes, heat and dissolve in a water bath at 55°C, prepare a 5% solution, quickly pour it into 8-9ml of acetone, let it stand for layers, and remove the upper white turbidity solution, the remaining part was redissolved in a water bath, and the pH value was adjusted between 3.2-3.8 with acetic acid, and the acetone solution was added dropwise until the liquid was milky white, with one or two drops in excess, quickly transferred to a round bottom flask, and placed in an ice bath After stirring with a magnetic stirrer for 10 minutes, 200 μl of 8% glutaraldehyde was added to cure for 30 minutes. Stop stirring, remove gelatin, and place in a small beaker.
[0023] (2), preparation of PSS-β-glycerophosphate complex
[0024] Prepare 1000 mL of anionic polyelectrolyte...
Embodiment 2
[0033] Example 2 Preparation of Doxorubicin Polyelectrolyte (PAH / PSS-β-Glycerophosphate) Capsules
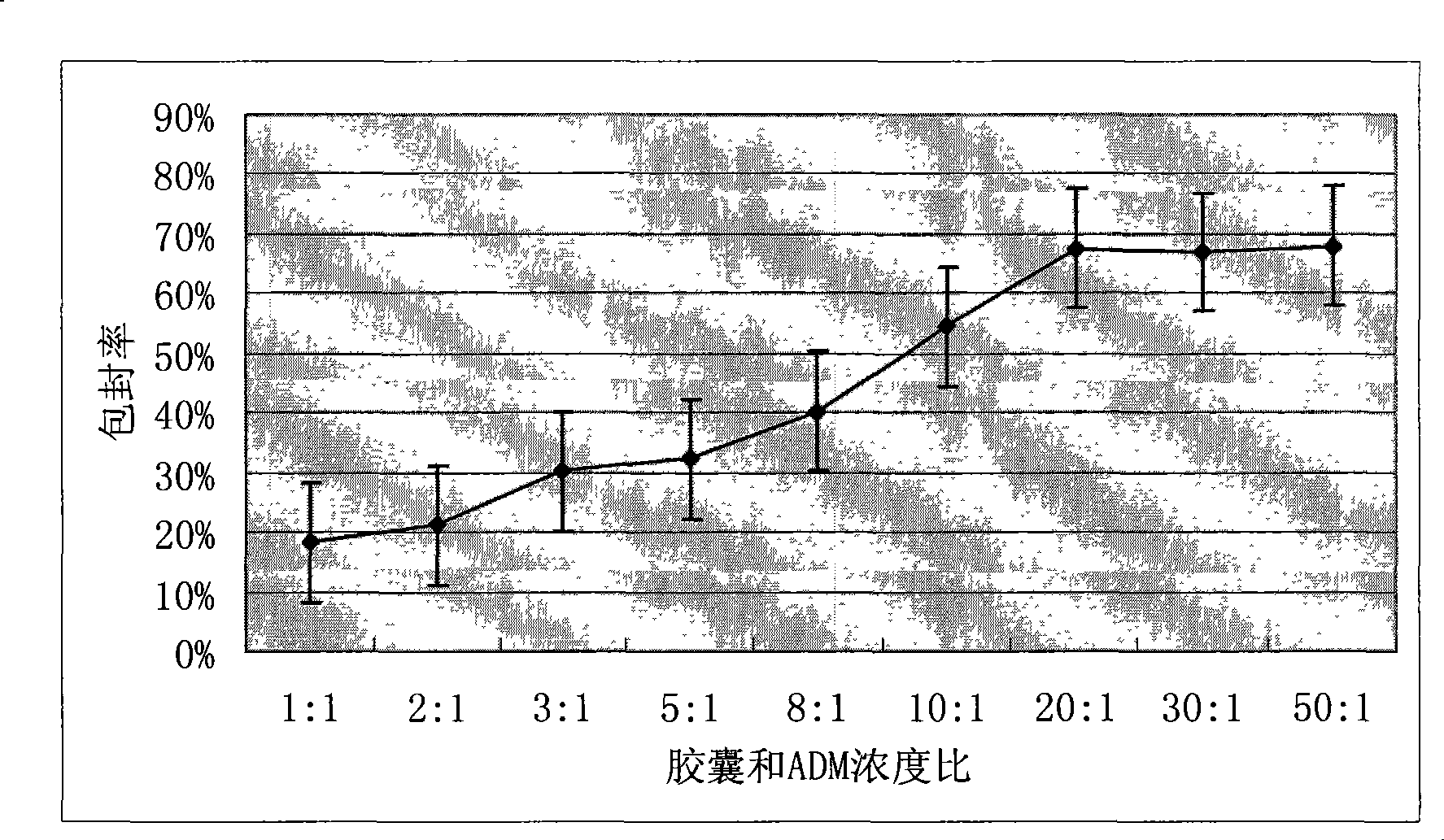
[0034] Get the doxorubicin solution 10mL of 100ug / mL and add in the cavity capsule that 20mg makes among the embodiment 1, after adding 0.7mol / L NaCl to adjust the salt concentration, place in the shaker 20 ℃ of constant temperature oscillations 24h, high-speed centrifugation (5000r / min×5min) the above solution, discard the supernatant, and the precipitate is a polyelectrolyte capsule carrying doxorubicin.
Embodiment 3
[0035] Example 3 Physicochemical Properties of Polyelectrolyte (PAH / PSS-β-Glycerophosphate) Empty Shell Capsules
[0036] TEM pictures of the capsules are shown in figure 1 shown. It can be seen from the figure that the prepared capsules have better sphericity and better dispersibility in the solution, and the particle size measured under electron microscope is mostly 200-300nm.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More