Method for synthesizing Piberacine precursor
A synthetic method and compound technology, applied in the field of synthesis of piperacine precursors, can solve problems such as unestablished, and achieve the effects of safe use, saving labor, electricity and freight, and high yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0028] The purpose of the present invention is to provide a method beneficial to industrial production of MC.
[0029] 1. Preparation process
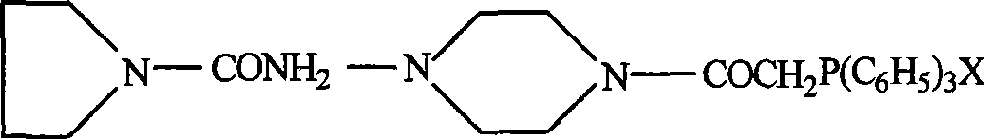
[0030] by and (X represents Cl or Br atom)
[0031] After the reaction, react with triphenylphosphine to obtain the following formula
[0032]
[0033] Reaction with 3.4.5-benzoic acid benzaldehyde to obtain the following formula
[0034]
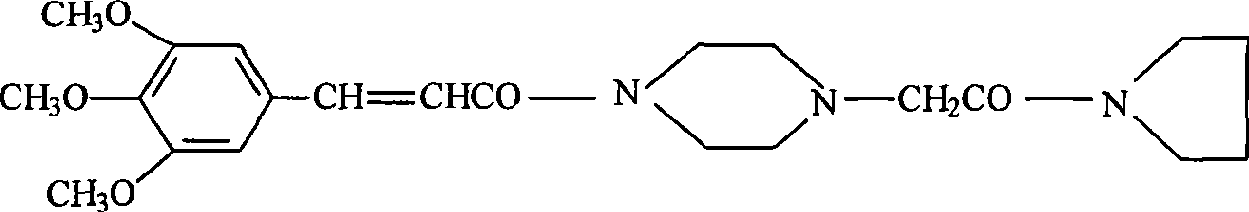
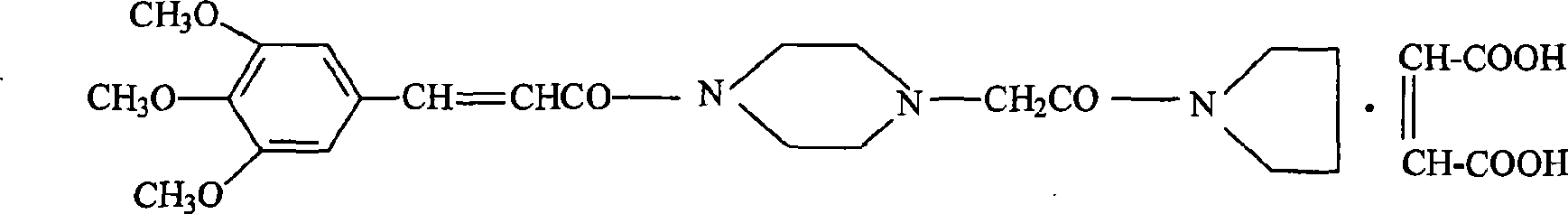
[0035] Maleic acid reacts with the piperacine precursor represented by formula ④ to generate
[0036]
[0037] According to the present invention, at first by the reaction of formula ⑦ and the compound represented by formula ⑧, then react with three phosphine.
[0038] The reaction of formula ⑦ and formula ⑧ can be carried out in the following solvents: chloroform, benzene, xylene, ether, cyclohexane and other solvents. The ratio of the reactants of formula ⑦ and formula ⑧ is not particularly limited, and can be selected within a wide range. Generally, the ratio of the former to th...
PUM
| Property | Measurement | Unit |
|---|---|---|
| melting point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More