Freeze-dried protein renaturation method
A protein refolding and protein denaturation technology, applied in the field of biopharmaceuticals, can solve problems such as excessive dilution of samples
Inactive Publication Date: 2009-10-28
张鹏
View PDF0 Cites 7 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Most of the current optimization is to change the formula of the refolding solution mixed in to improve the refolding rate, but the excessive dilution of the sample is unavoidable
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment Construction
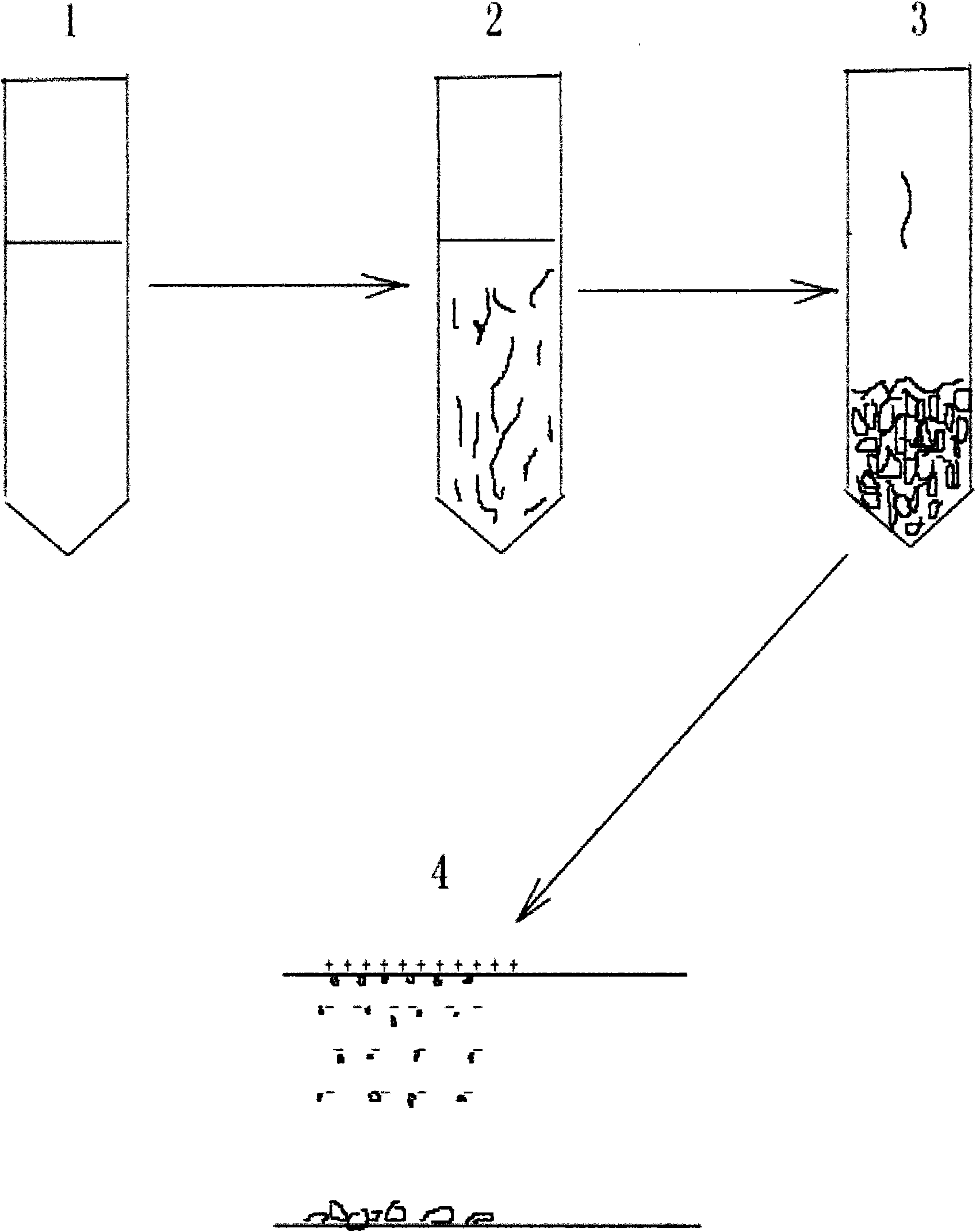
[0011] The present invention will be described in further detail in conjunction with the accompanying drawings. Step 1 Dissolve the target protein with reagents such as 6M guanidine hydrochloride or 8M urea according to the optimized biological operation formula. Step 2 needs to optimize the temperature drop rate and solution dosage for different protein pairs, and find a suitable freezing method. Step 3: sublimate water and the like in a freeze-drying vacuum dryer. Step 4 Appropriately crush the solid particle components obtained after freeze-drying, and separate the active protein powder by high-voltage electrostatic adsorption.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention relates to a freeze-dried protein renaturation method based on a natural phenomenon that solute is naturally separated from solvent in the solution freezing process. The solubility of solute salt in a solution is influenced by temperature, denaturation salt (urea, guanidine hydrochloride, and the like) is gradually crystallized and separated from the solution because the solubility of the denaturation salt lowers at a low temperature, and two vital denaturation factors, namely molecular heat movement and the denaturation salt concentration of the solution, are reduced. The crystallization speed can be controlled by the temperature drop speed so as to establish a wonderful condition for protein renaturation, namely the low temperature as well as a smooth downward gradient of the denaturation salt concentration. Meanwhile, a renaturation solution is aided with antifreezing agents (DMSO, fucose, glucose, and the like) with a certain concentration, renaturation activators (cyclodextrin, glycin, molecular chaperones, and the like), reductant-oxidant, buffer salts, and the like for preventing freezing damage to the protein activity and improving the renaturation rate. The frozen dry protein is dried by a freeze drier to obtain solid particles which are suitably vibrated to break by utilizing the difference between the denaturation salt and dry protein powder in the crystallization proportion and the crystallization form and separated by adopting the methods of pneumatic separation, sieving, electrostatic adherence and the like to obtain target protein.
Description
Technical field: [0001] The invention relates to a protein renaturation method, which is applied to the fields of biological protein engineering, especially bacterial inclusion body protein engineering, molecular biology downstream research, genetic engineering and protein technology research, biopharmaceuticals and the like. Background technique: [0002] Protein renaturation is the process of qualitative change from the primary structure of inactive peptide chains to active protein tertiary or quaternary structures in the central dogma at the core of the biological field. It is of great significance both theoretically and practically. With the rapid development of molecular biology and genetic engineering protein technology, more and more protein and polypeptide products are provided for clinical, scientific research and industrial production through heterologous expression of proteins. So far, bacteria have become the preferred expression system for the production of reco...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): C07K1/14
Inventor 张鹏张志滨
Owner 张鹏