Electrochromatic epoxy resin polymer and preparation method thereof
An epoxy resin and electrochromic technology, applied in the direction of color-changing fluorescent materials, chemical instruments and methods, etc., can solve the problems of easy peeling off of the film and difficulty in measuring the electrochromic performance, and achieve good bonding performance, good mechanical properties, Good effect of acid and alkali resistance and heat resistance
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
specific Embodiment approach 1
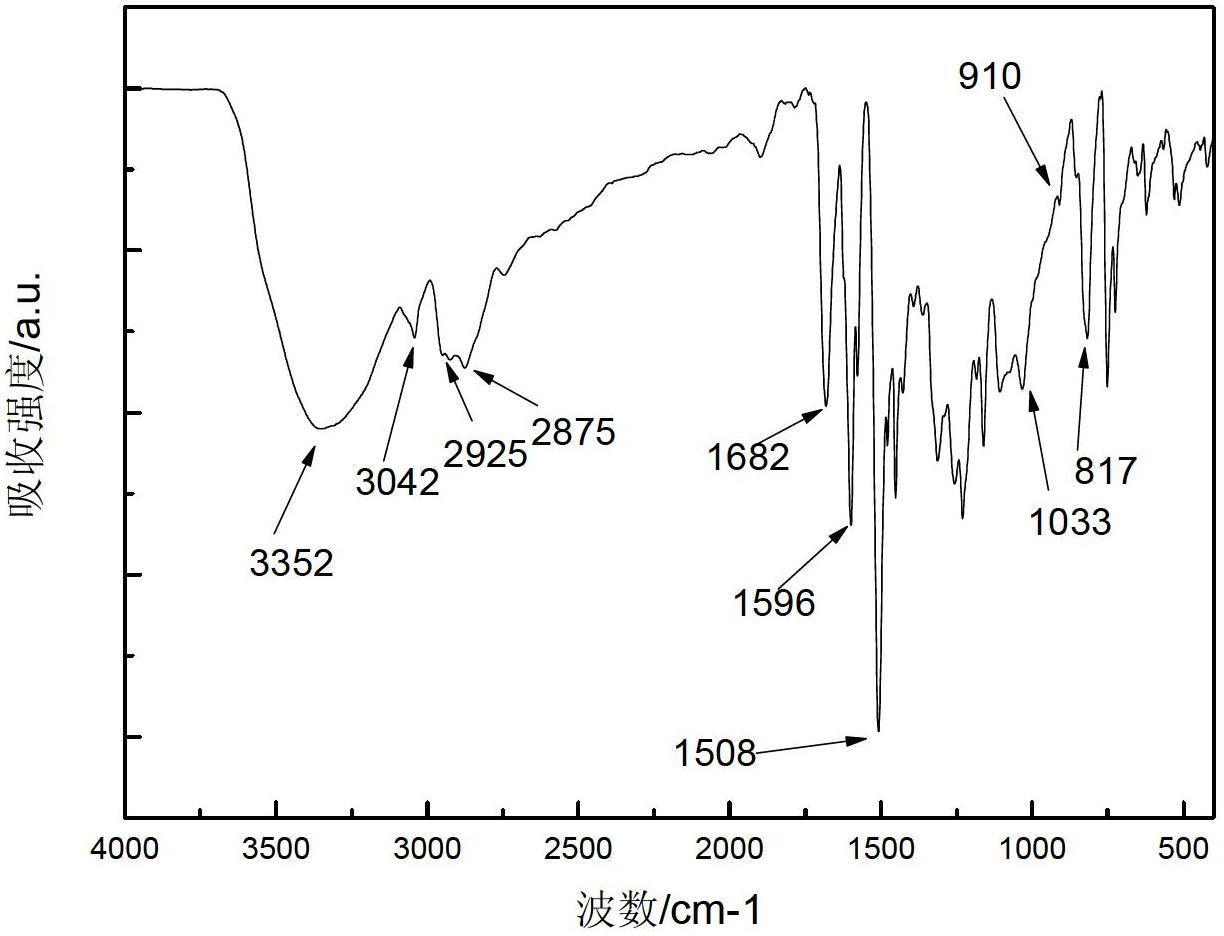
[0036] Embodiment 1: The chemical structural formula of a kind of epoxy resin polymer with electrochromic performance in this embodiment is as follows:
[0037]
[0038] Among them, n is 10~20, R is
[0039]
[0040]
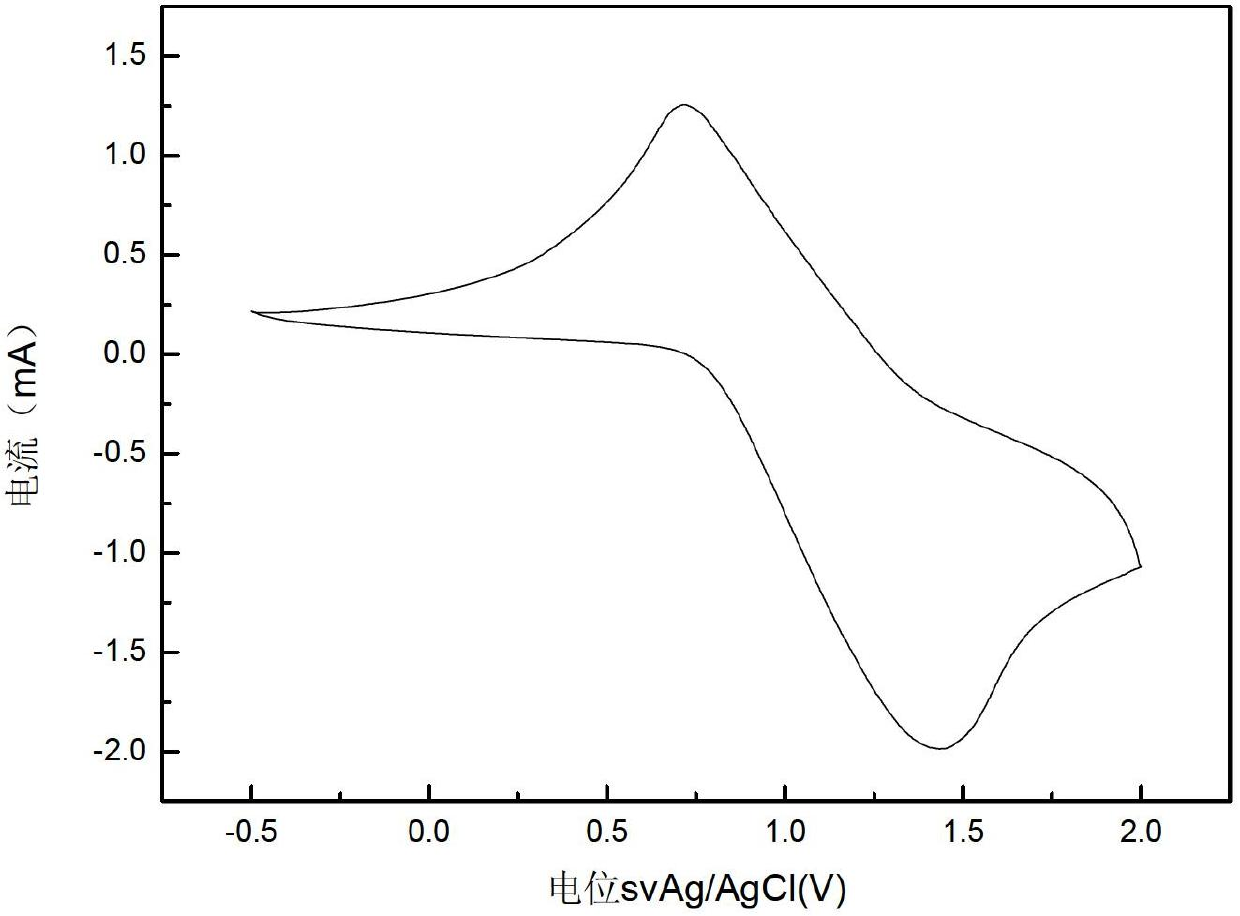
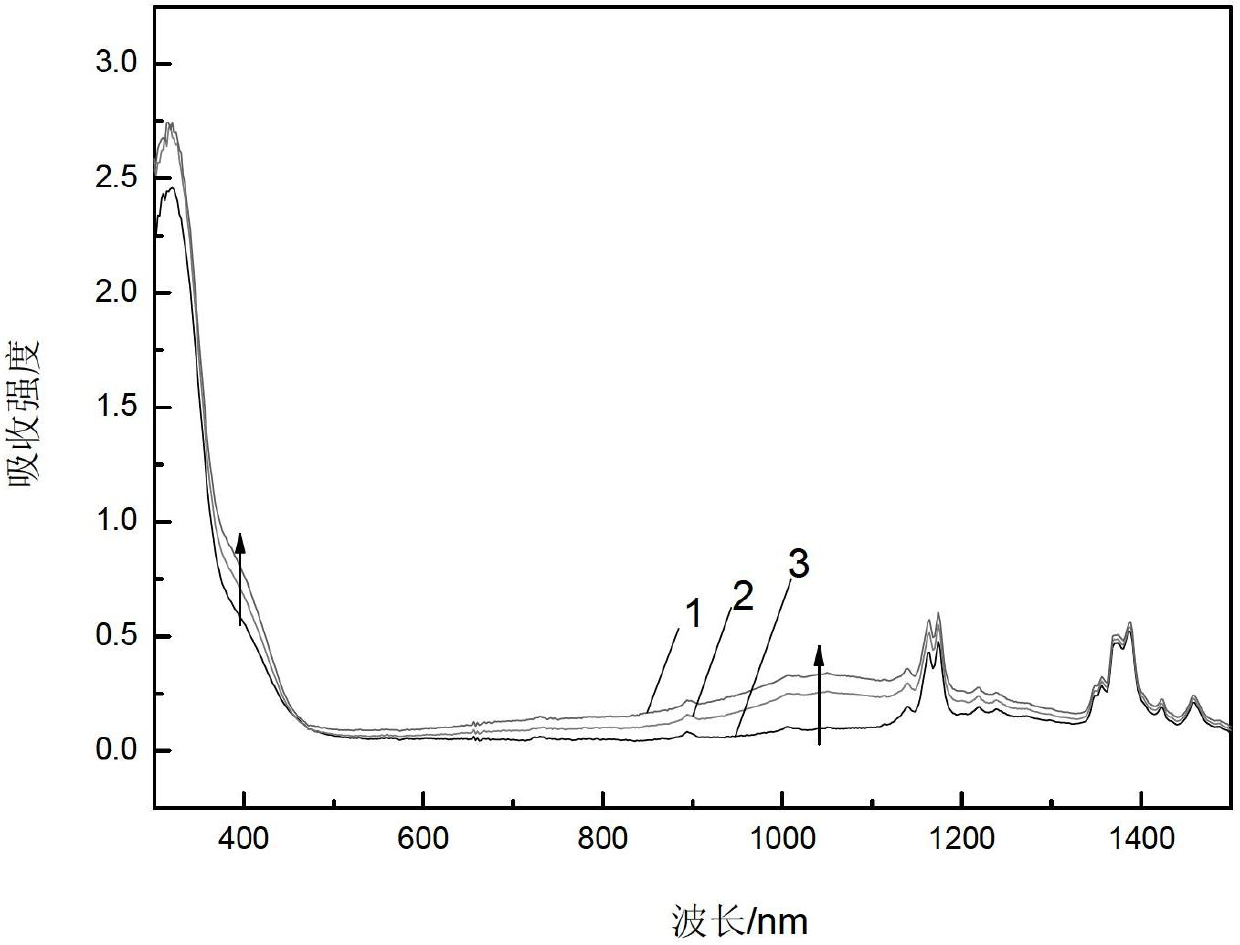
[0041] The epoxy resin polymer of this embodiment has good bonding performance, a shear strength of about 30MPa, good acid and alkali resistance and heat resistance, and the highest service temperature can reach 120°C. It has good mechanical properties, and at the same time has aniline Electrochromic properties. The self-luminous response speed of the epoxy resin polymer in this embodiment is 1000 times that of liquid crystals, and has potential value in the development of molecular designers. The epoxy resin polymer synthesized in this embodiment has an epoxy value of 0.12-0.30 equivalent / 100g, and has adhesive and coating properties.
specific Embodiment approach 2
[0042] Specific embodiment two: a kind of preparation method of epoxy resin polymer with electrochromic performance of the present embodiment is carried out according to the following steps:
[0043] 1. Synthesis of monomers
[0044] a. Mix diaminotriphenylamine derivatives and p-hydroxybenzaldehyde in a molar ratio of 1:2~3 to obtain a mixture; b. Mix the mixture obtained in step a and the solvent in a mass-volume ratio of 0.8-2.1g : Mix 100mL evenly to obtain a mixture, then raise the temperature to 120°C~140°C under nitrogen protection, stir at a speed of 20~50rpm for 12h, when the solvent in the mixture evaporates until the volume of the mixture is the original volume At 1 / 4, stop stirring and collect the remaining 1 / 4 volume of liquid; c. Put the remaining 1 / 4 volume of liquid collected in step b into distilled water, let it stand for 6~12h, and then put it under the condition of vacuum degree of 0.09MPa Suction filtration under low temperature to collect the solid phase...
specific Embodiment approach 3
[0048] Specific embodiment 3: The difference between this embodiment and specific embodiment 2 is that the diaminotriphenylamine derivative described in step 1 is 4,4′-diamino-4″-N-carbazolyltriphenylamine, N, N-bis(4-aminophenyl)-N′,N′-diphenyl-1,4-phenylenediamine, N′-(4-aminophenyl)-N′-(4-ethoxy) Phenyl-1,4-phenylenediamine, N,N′-bis(β-naphthyl)-N,N′-bis(4-aniline)-p-phenylenediamine or N′-(4-aminophenyl) -N'-phenyl-1,4-phenylenediamine, N',N'-diphenyl-N,N'-bis(4-aniline)-p-phenylenediamine, N',N'-diphenyl -N,N'-bis(4-phenylamine)biphenylenediamine or N,N'-bis(α-naphthyl)-N,N'-bis(4-phenylamine)biphenylenediamine.Others are the same as step 2 same.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Shear strength | aaaaa | aaaaa |
| Epoxy value | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More