Laccase gene, engineering bacteria and application
A technology of genetically engineered bacteria and engineered bacteria, applied in the field of environmental biology, can solve the problem of discovering endogenous laccase, and achieve the effect of improving expression activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0021] Codon Optimization of Laccase Gene
[0022] According to the cotA gene sequence of Bacillus subtilis (GENEID: 936023), combined with the codon preference expressed by Pichia pastoris, the codon optimization of the cotA gene sequence was carried out, and restriction sites were introduced into the upstream and downstream of the full-length gene: EcoRI and NotI. At the same time, in order to prevent the optimized gene sequence from containing EcoRI restriction site and subsequent DNA linearization BglII restriction site, and adjust the GC content of the optimized gene, the 855th base A was mutated to G, and the 1098th base was mutated. The base T is mutated to C. The full length of the gene is 1542 bases, encoding 514 amino acids. The optimized laccase gene was named Tcot.
[0023] Design restriction sites and protective bases at both ends of the gene, and design primers as follows:
[0024] P1: ACGGAATTCATGACTTTGGAAAAGTTTGTTGATGCTTTGCCAATTCCAGATACTTTGAAGCCAGTTCAAC
...
Embodiment 2
[0056] Expression of Laccase Gene in Yeast Cells
[0057] 1. Construction of expression vector
[0058] The codon-optimized PCR product was digested with EcoRI / NotⅠ, the fragment was recovered, inserted into the same double digested Pichia constitutive expression vector pGAPZaA (Invitrogen, USA) with the correct reading frame, ligated, and transformed E. coli DH5α, the recombinant plasmid pGAPZaA-Tcot was obtained. The recombinant plasmid was verified by EcoRI / NotI double enzyme digestion.
[0059] 2. Preparation of competent cells for electroporation
[0060] In a 50ml centrifuge tube containing 5ml YPD, culture yeast cells overnight at 30°C; take 30~50ul of the overnight culture, inoculate a 250ml shaker flask containing 50ml of fresh medium, and grow overnight until OD600=1.3~1.5; at 4 degrees, 1500g Collect the cells by centrifugation for 5 minutes, and suspend the cells with 50ml pre-cooled sterilized water; centrifuge as above, suspend the cells with 25ml pre-cooled ...
Embodiment 3
[0076] Study on the Properties of Laccase
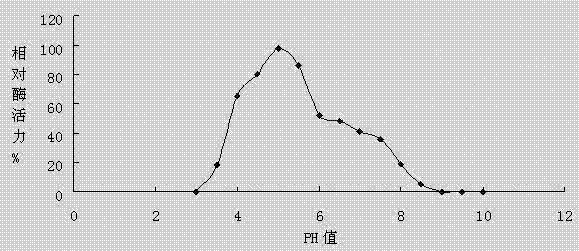
[0077] 1. Effect of pH value on laccase activity
[0078] Laccase was added to the buffer system with different pH values to measure the enzyme activity, the pH was set from 2.0 to 10.0, and points were taken every 0.5 for measurement. The enzyme activity assay reaction was carried out at 25° C., and the reaction time was 15 minutes. The result is as figure 1 It was shown that the optimum pH value of laccase is 4.5~5.5, and in the range of pH4.0 to pH6.5, the enzyme activity of laccase can maintain more than 50%.
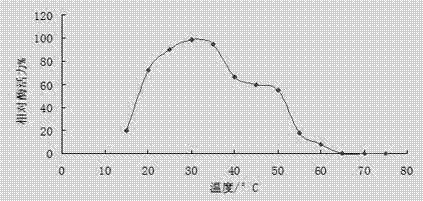
[0079] 2. Effect of temperature on laccase activity
[0080] The optimum reaction temperature of laccase was determined at the optimum pH value, the reaction time was 15min, the temperature was set from 15°C to 75°C, and points were taken every 5°C for measurement. The result is as figure 2 It shows that the optimum temperature of laccase is 25~35℃, and the enzyme activity can keep more than 60% at 20~45℃.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com