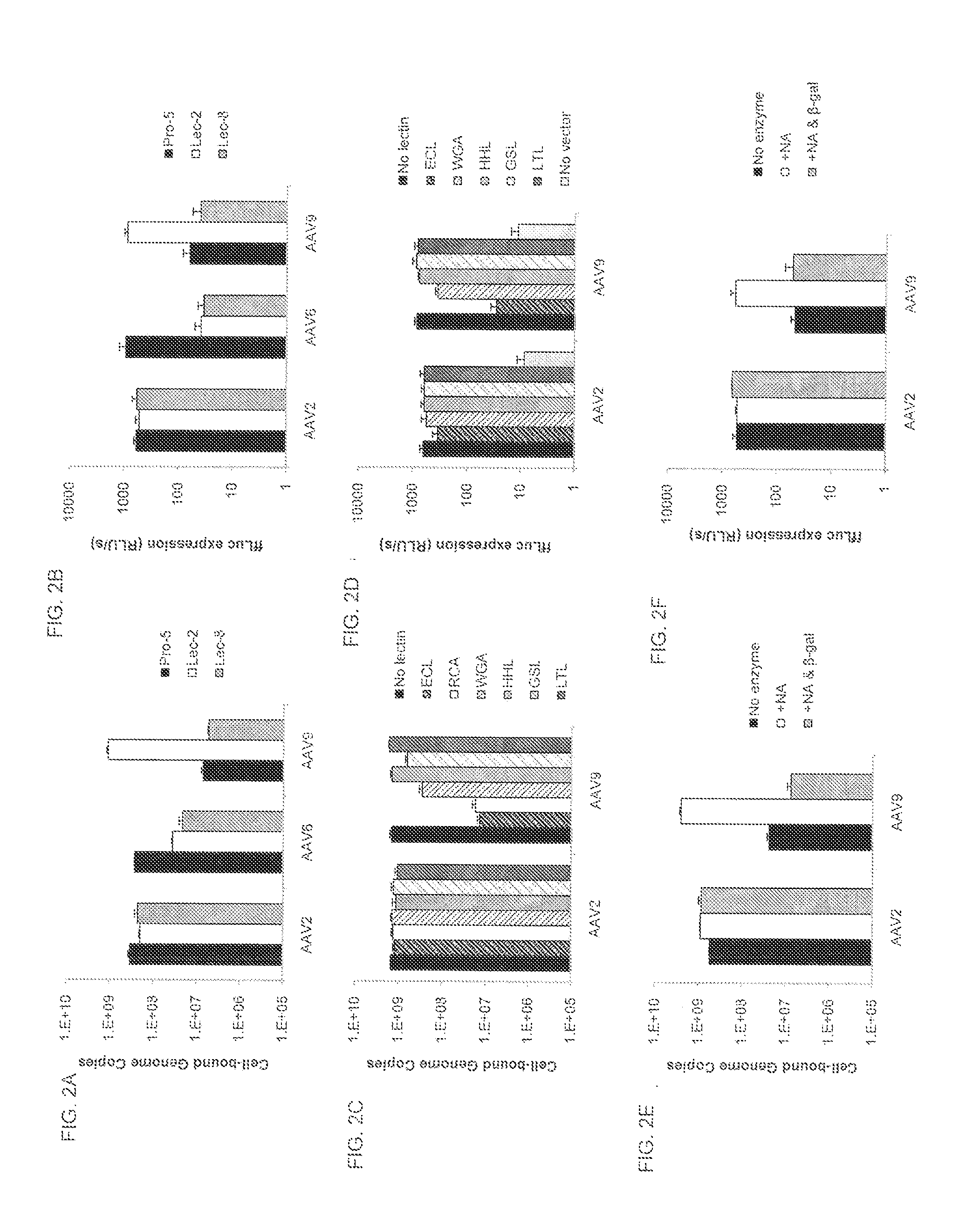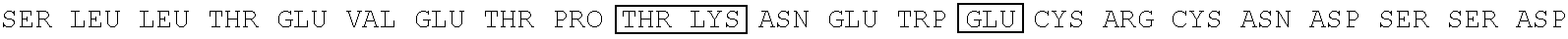Patents
Literature
1800 results about "Glycan" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
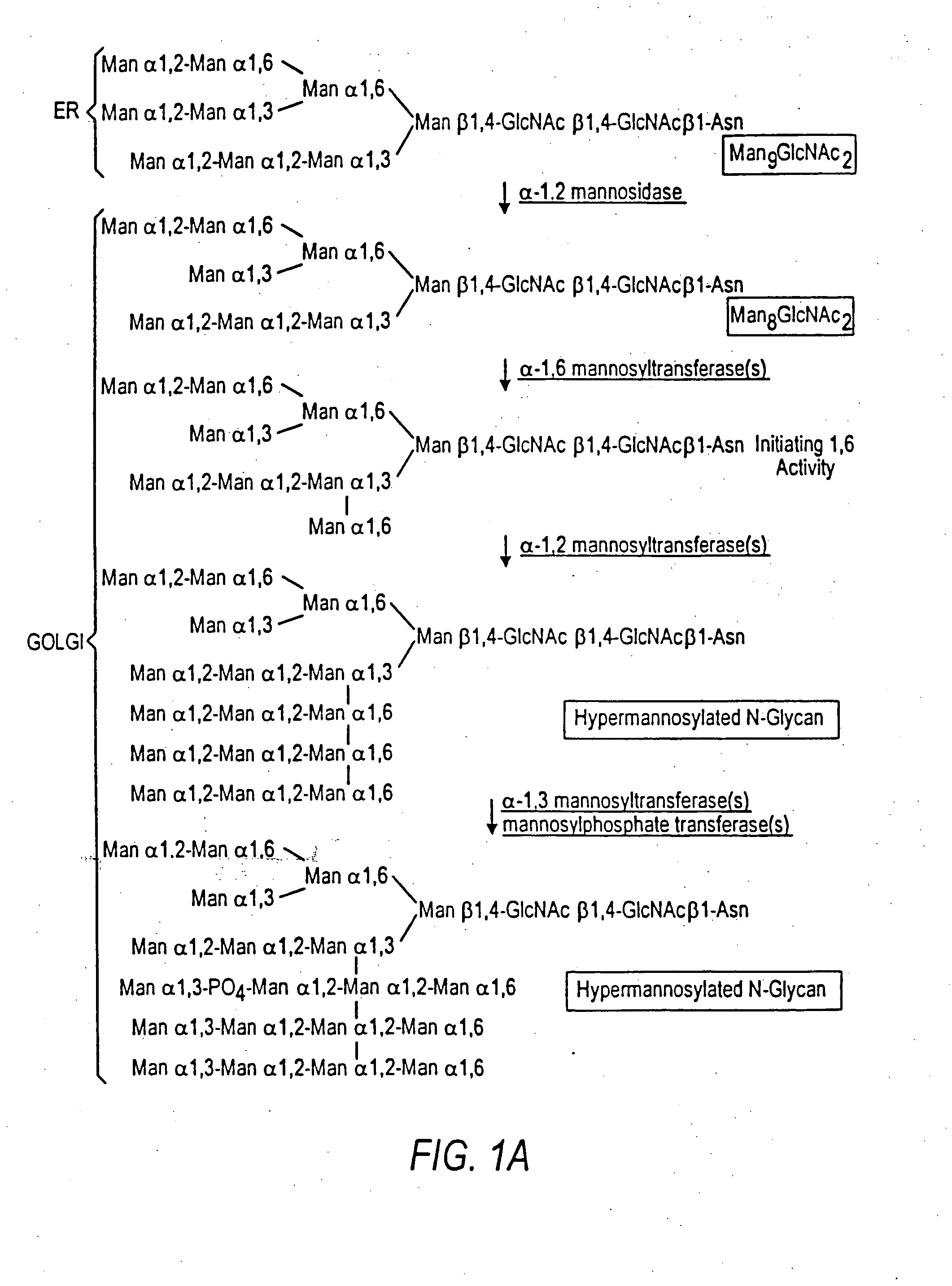
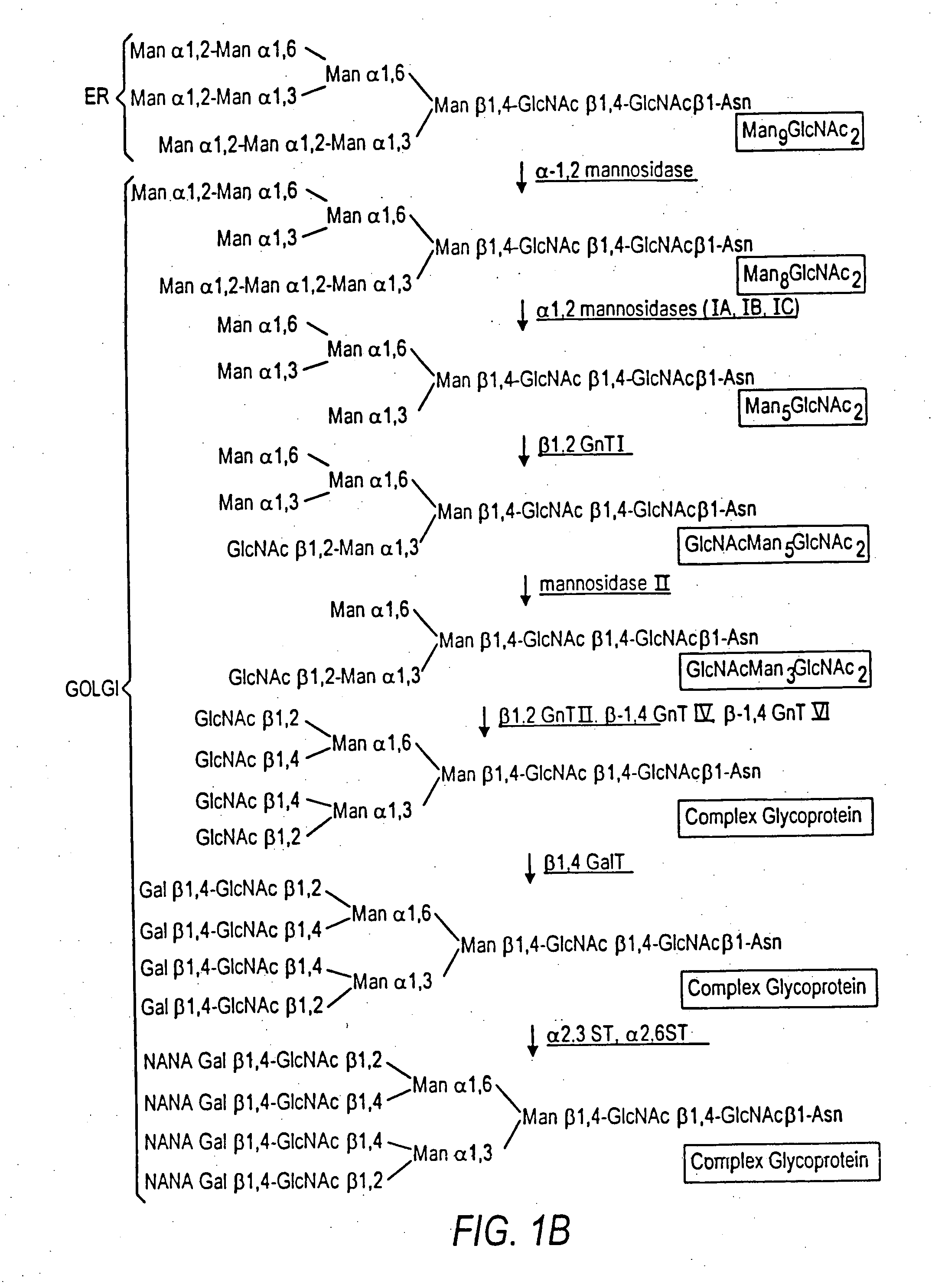
The terms glycan and polysaccharide are defined by IUPAC as synonyms meaning "compounds consisting of a large number of monosaccharides linked glycosidically". However, in practice the term glycan may also be used to refer to the carbohydrate portion of a glycoconjugate, such as a glycoprotein, glycolipid, or a proteoglycan, even if the carbohydrate is only an oligosaccharide. Glycans usually consist solely of O-glycosidic linkages of monosaccharides. For example, cellulose is a glycan (or, to be more specific, a glucan) composed of β-1,4-linked D-glucose, and chitin is a glycan composed of β-1,4-linked N-acetyl-D-glucosamine. Glycans can be homo- or heteropolymers of monosaccharide residues, and can be linear or branched.
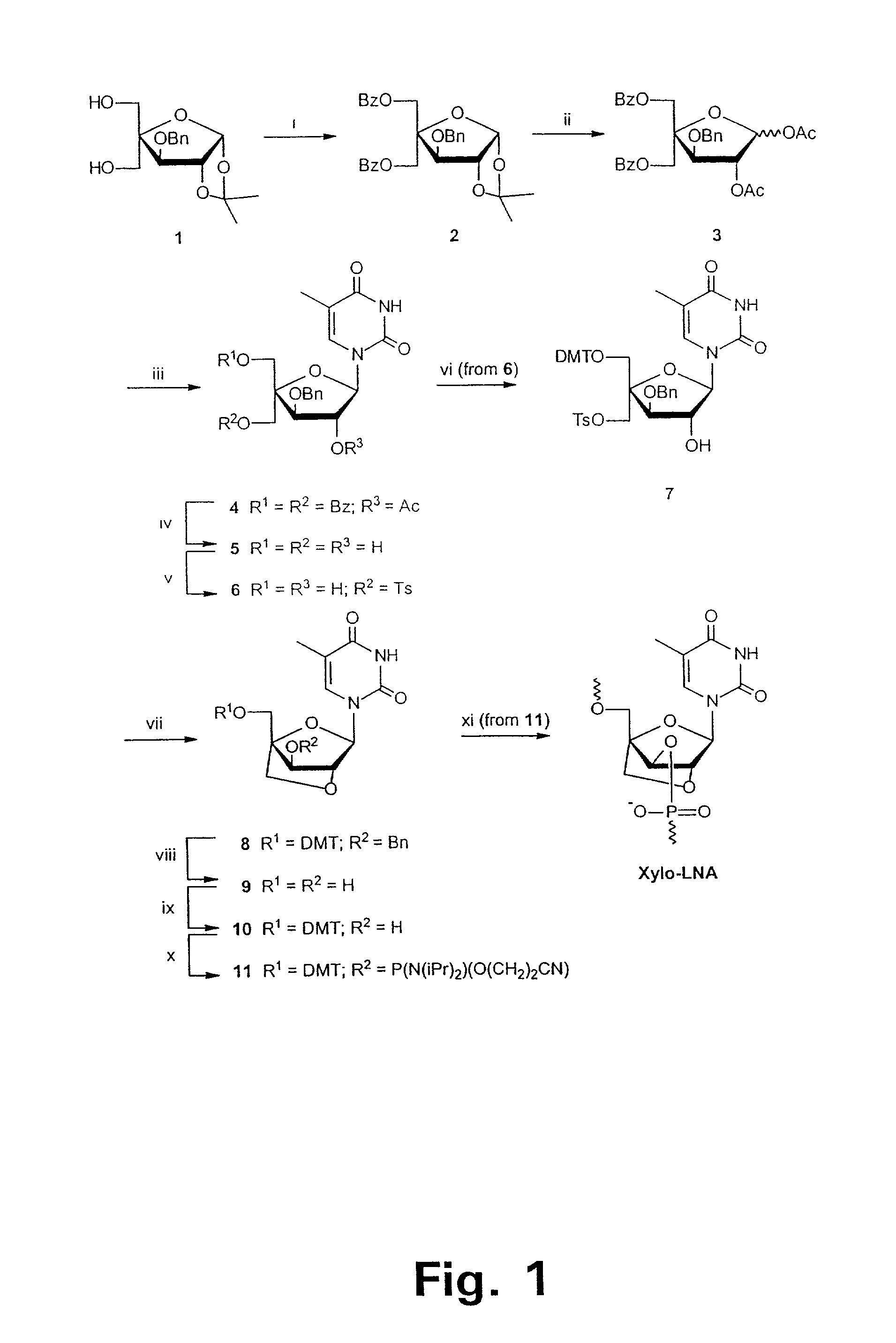
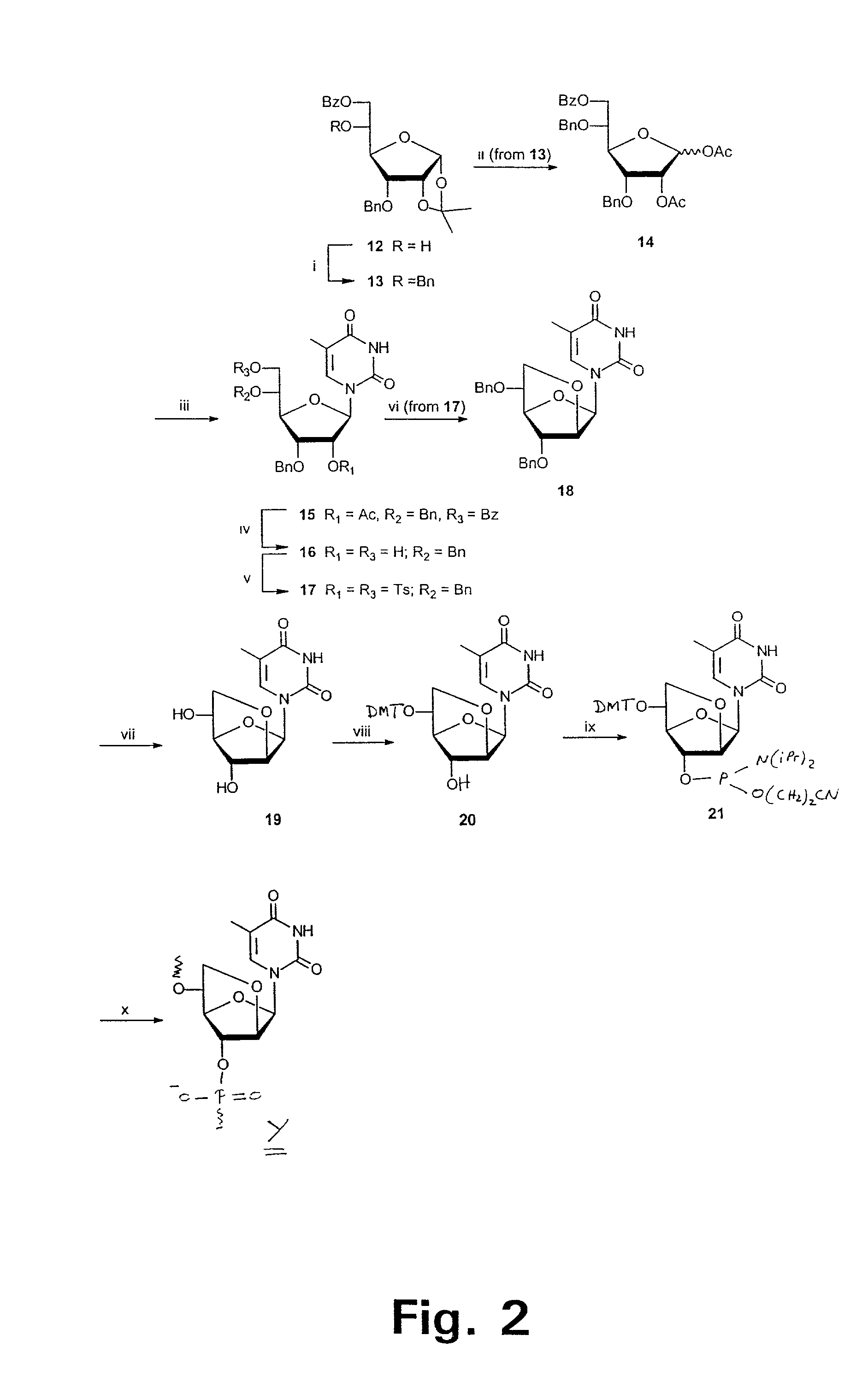
Xylo-LNA analogues
Based on the above and on the remarkable properties of the 2′-O,4′-C-methylene bridged LNA monomers it was decided to synthesise oligonucleotides comprising one or more 2′-O,4′-C-methylene-β-D-xylofuranosyl nucleotide monomer(s) as the first stereoisomer of LNA modified oligonucleotides. Modelling clearly indicated the xylo-LNA monomers to be locked in an N-type furanose conformation. Whereas the parent 2′-deoxy-β-D-xylofuranosyl nucleosides were shown to adopt mainly an N-type furanose conformation, the furanose ring of the 2′-deoxy-β-D-xylofuranosyl monomers present in xylo-DNA were shown by conformational analysis and computer modelling to prefer an S-type conformation thereby minimising steric repulsion between the nucleobase and the 3′-O-phopshate group (Seela, F.; Wömer, Rosemeyer, H. Helv. Chem. Acta 1994, 77, 883). As no report on the hybridisation properties and binding mode of xylo-configurated oligonucleotides in an RNA context was believed to exist, it was the aim to synthesise 2′-O,4′-C-methylene-β-D-xylofuranosyl nucleotide monomer and to study the thermal stability of oligonucleotides comprising this monomer. The results showed that fully modified or almost fully modified Xylo-LNA is useful for high-affinity targeting of complementary nucleic acids. When taking into consideration the inverted stereochemistry at C-3′ this is a surprising fact. It is likely that Xylo-LNA monomers, in a sequence context of Xylo-DNA monomers, should have an affinity-increasing effect.
Owner:QIAGEN GMBH
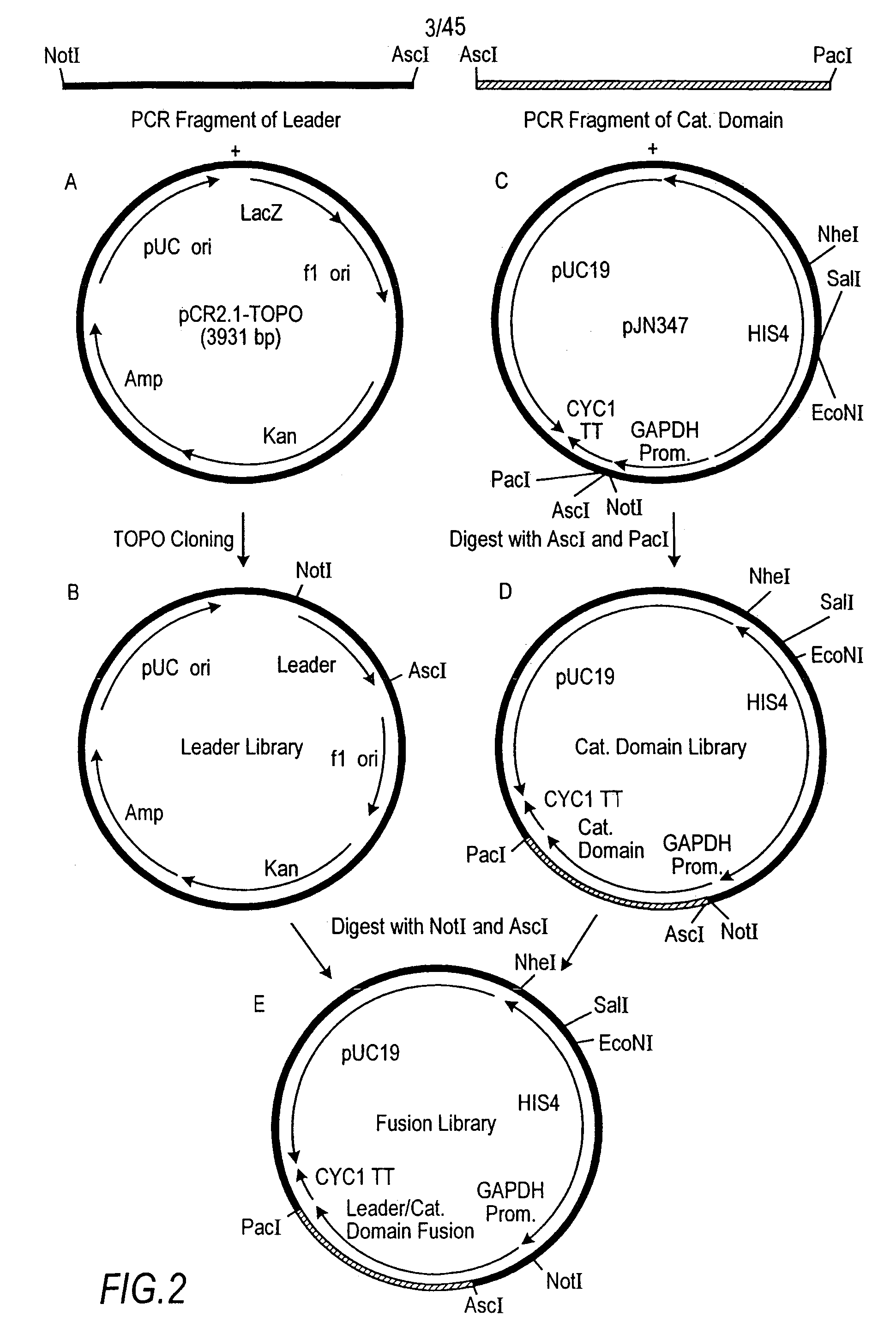
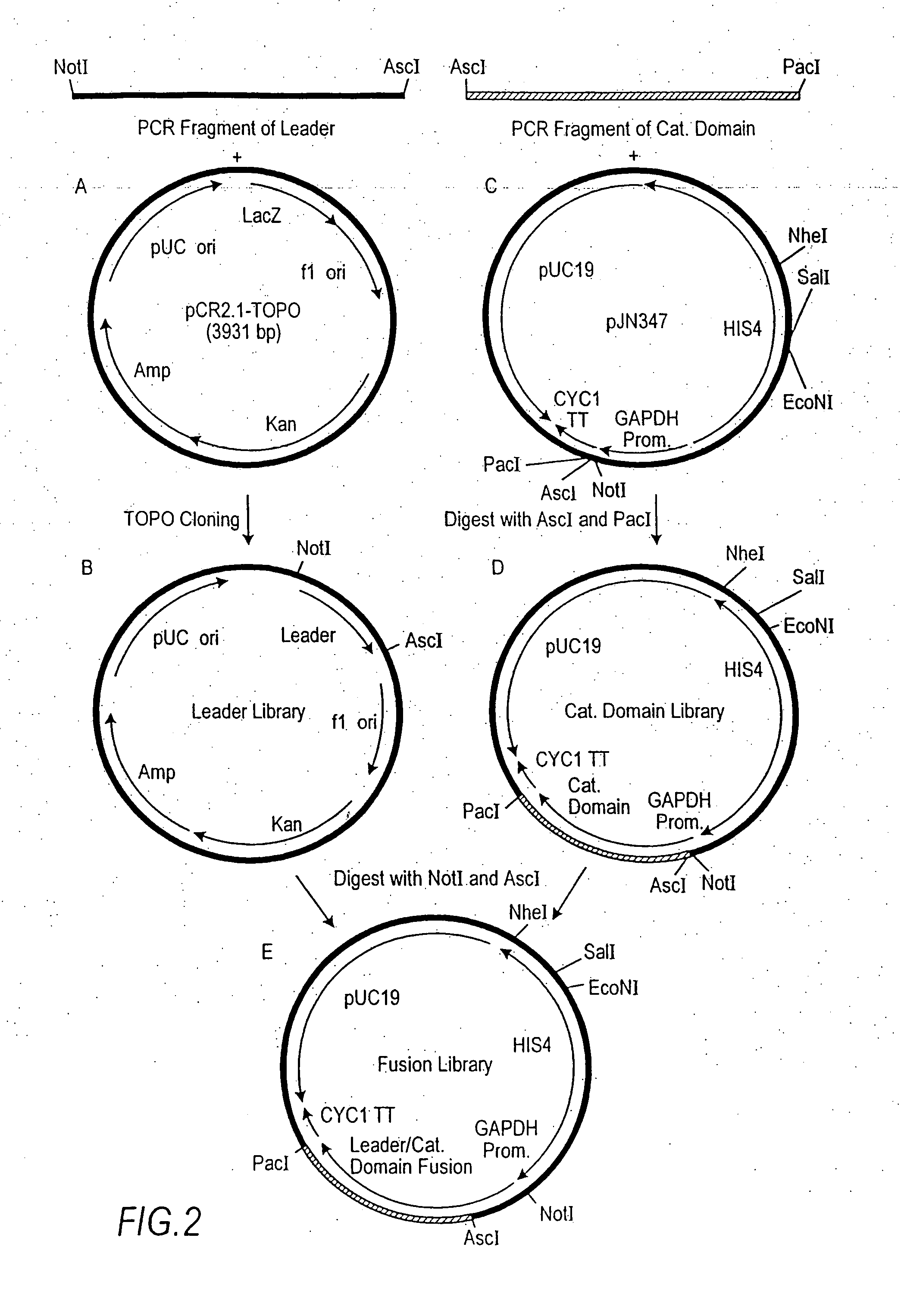
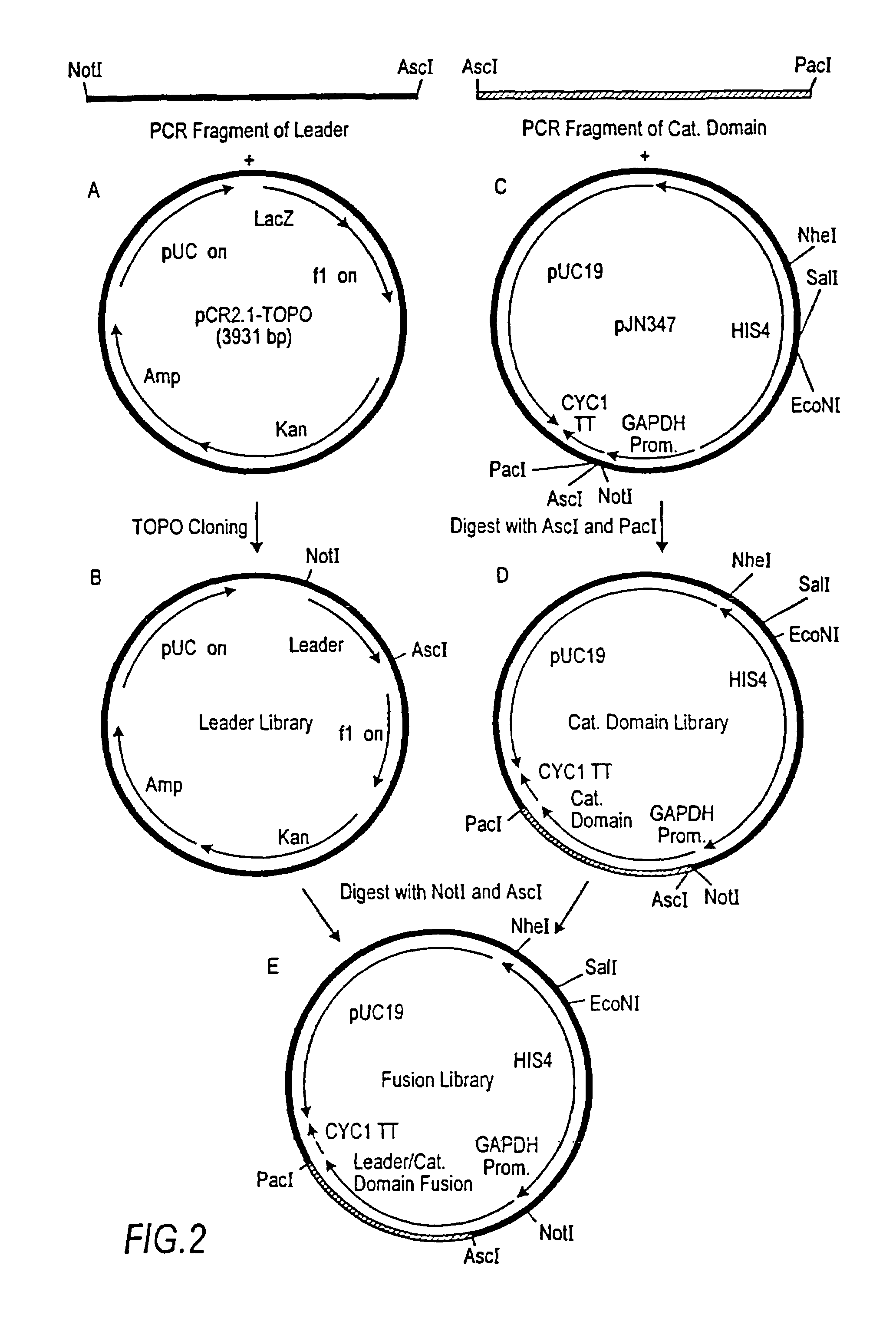
Combinatorial DNA library for producing modified N-glycans in lower eukaryotes
InactiveUS7449308B2The overall structure is closedHigh yieldSugar derivativesMicroorganismsHeterologousTherapeutic protein
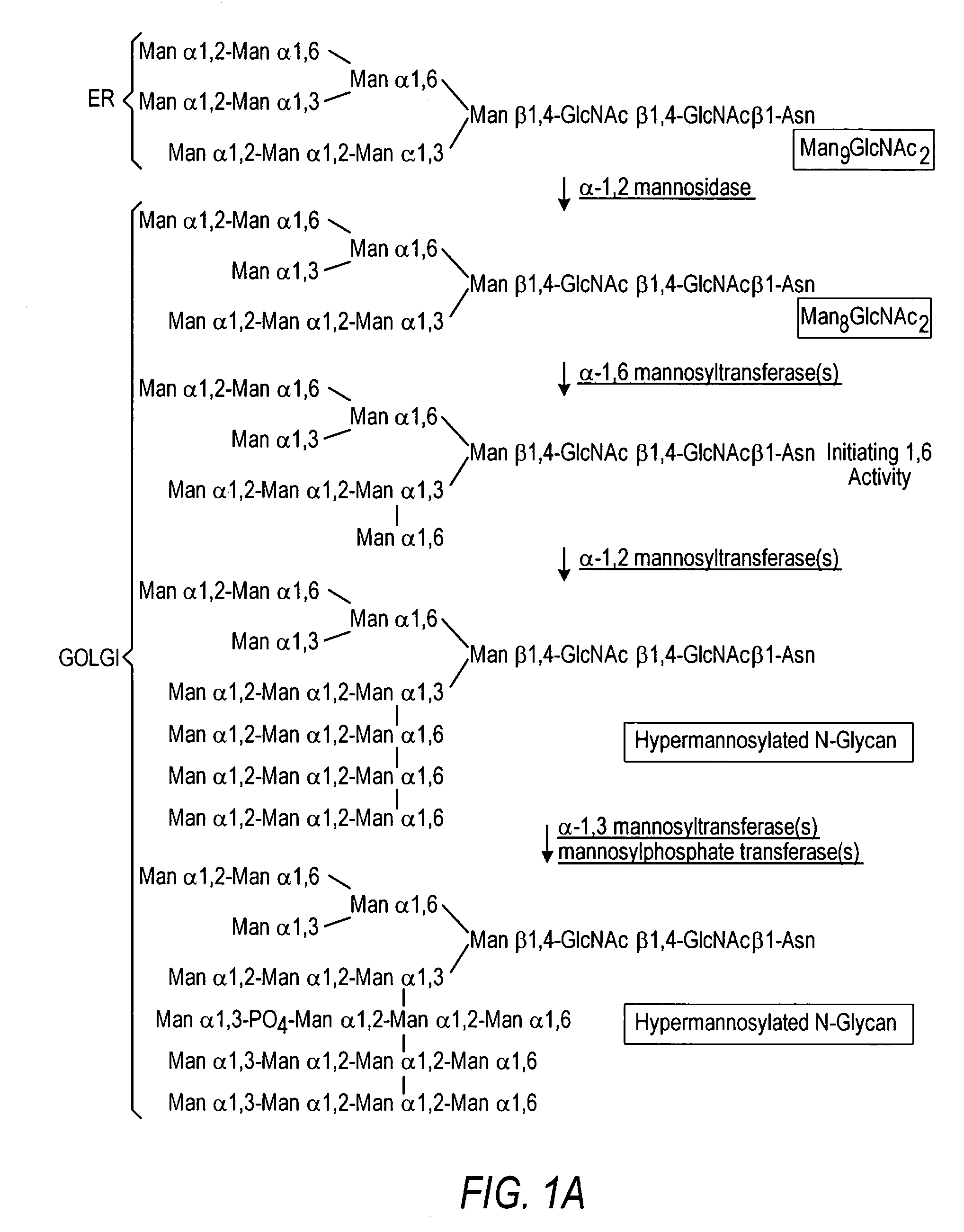
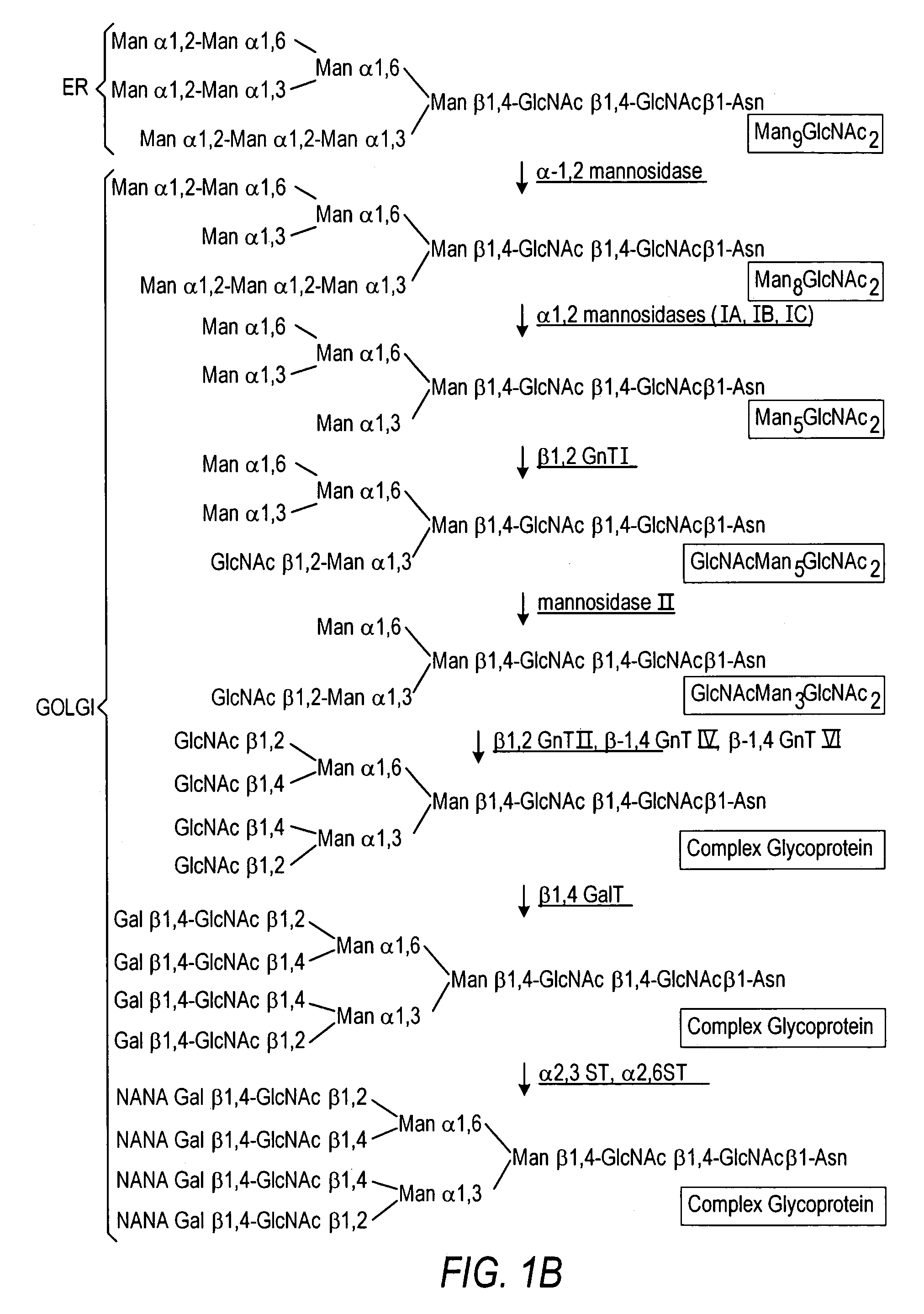
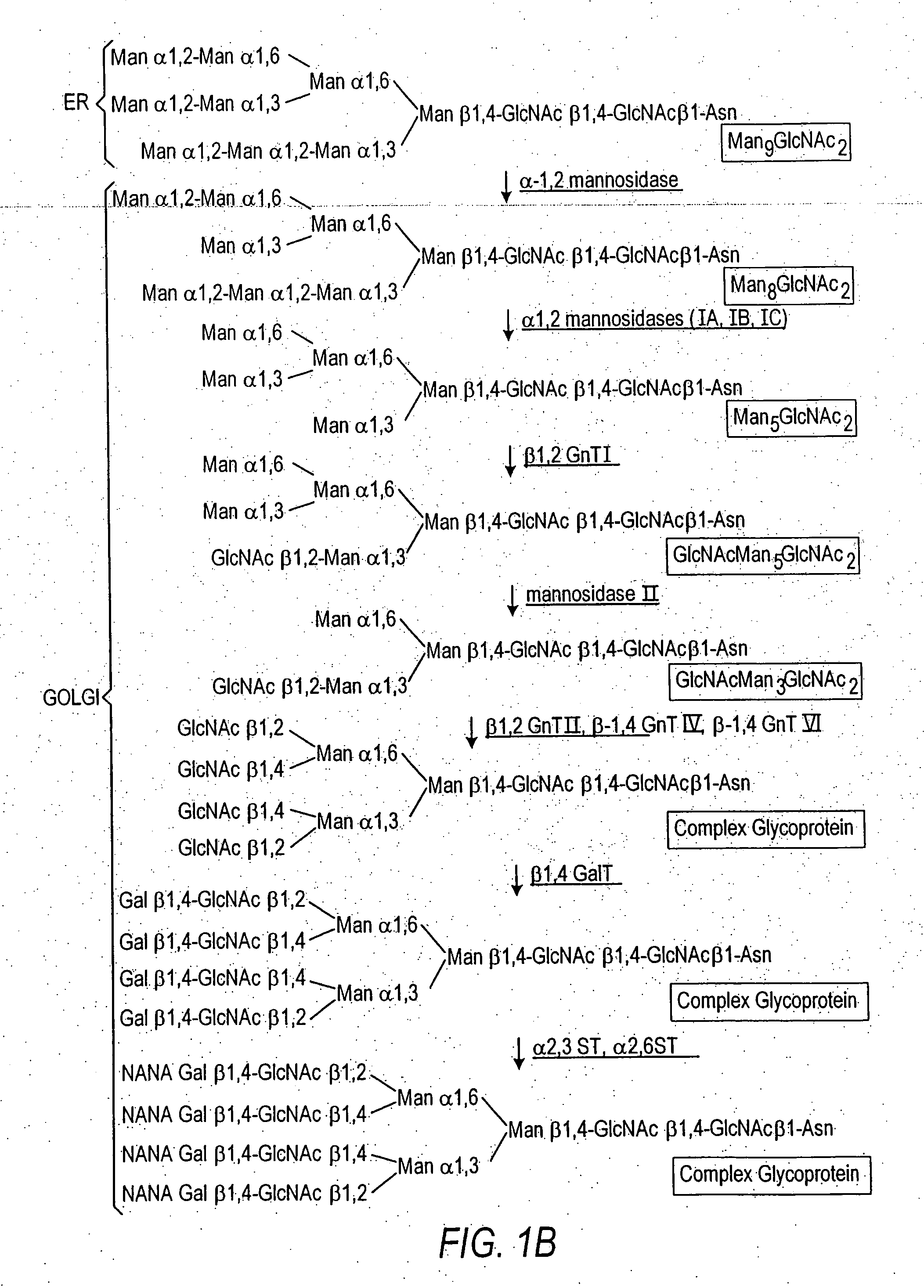
The present invention relates to eukaryotic host cells having modified oligosaccharides which may be modified further by heterologous expression of a set of glycosyltransferases, sugar transporters and mannosidases to become host-strains for the production of mammalian, e.g., human therapeutic glycoproteins. The invention provides nucleic acid molecules and combinatorial libraries which can be used to successfully target and express mammalian enzymatic activities such as those involved in glycosylation to intracellular compartments in a eukaryotic host cell. The process provides an engineered host cell which can be used to express and target any desirable gene(s) involved in glycosylation. Host cells with modified oligosaccharides are created or selected. N-glycans made in the engineered host cells have a Man5GlcNAc2 core structure which may then be modified further by heterologous expression of one or more enzymes, e.g., glycosyltransferases, sugar transporters and mannosidases, to yield human-like glycoproteins. For the production of therapeutic proteins, this method may be adapted to engineer cell lines in which any desired glycosylation structure may be obtained.
Owner:GLYCOFI
Production of sialylated N-glycans in lower eukaryotes
InactiveUS20060286637A1Improve efficiencyReducing competitive product inhibitionAntibody mimetics/scaffoldsTransferasesHeterologousSialic acid aldolase
The present invention relates to eukaryotic host cells which have been modified to produce sialylated glycoproteins by the heterologous expression of a set of glycosyltransferases, including sialyltransferase and / or trans-sialidase, to become host-strains for the production of mammalian, e.g., human therapeutic glycoproteins. Novel eukaryotic host cells expressing a CMP-sialic acid biosynthetic pathway for the production of sialylated glycoproteins are also provided. The invention provides nucleic acid molecules and combinatorial libraries which can be used to successfully target and express mammalian enzymatic activities (such as those involved in sialylation) to intracellular compartments in a eukaryotic host cell. The process provides an engineered host cell which can be used to express and target any desirable gene(s) involved in glycosylation.
Owner:GLYCOFI
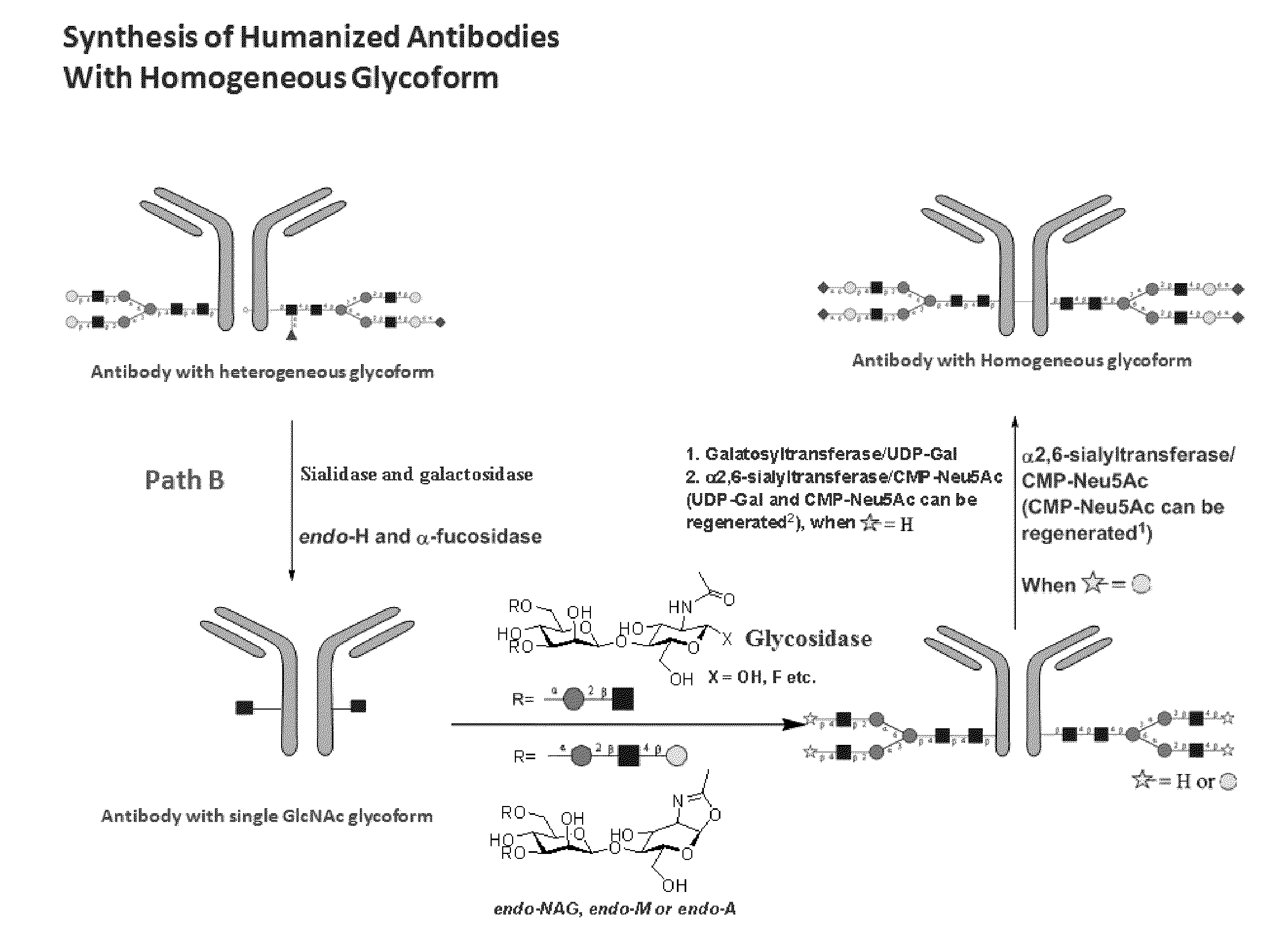
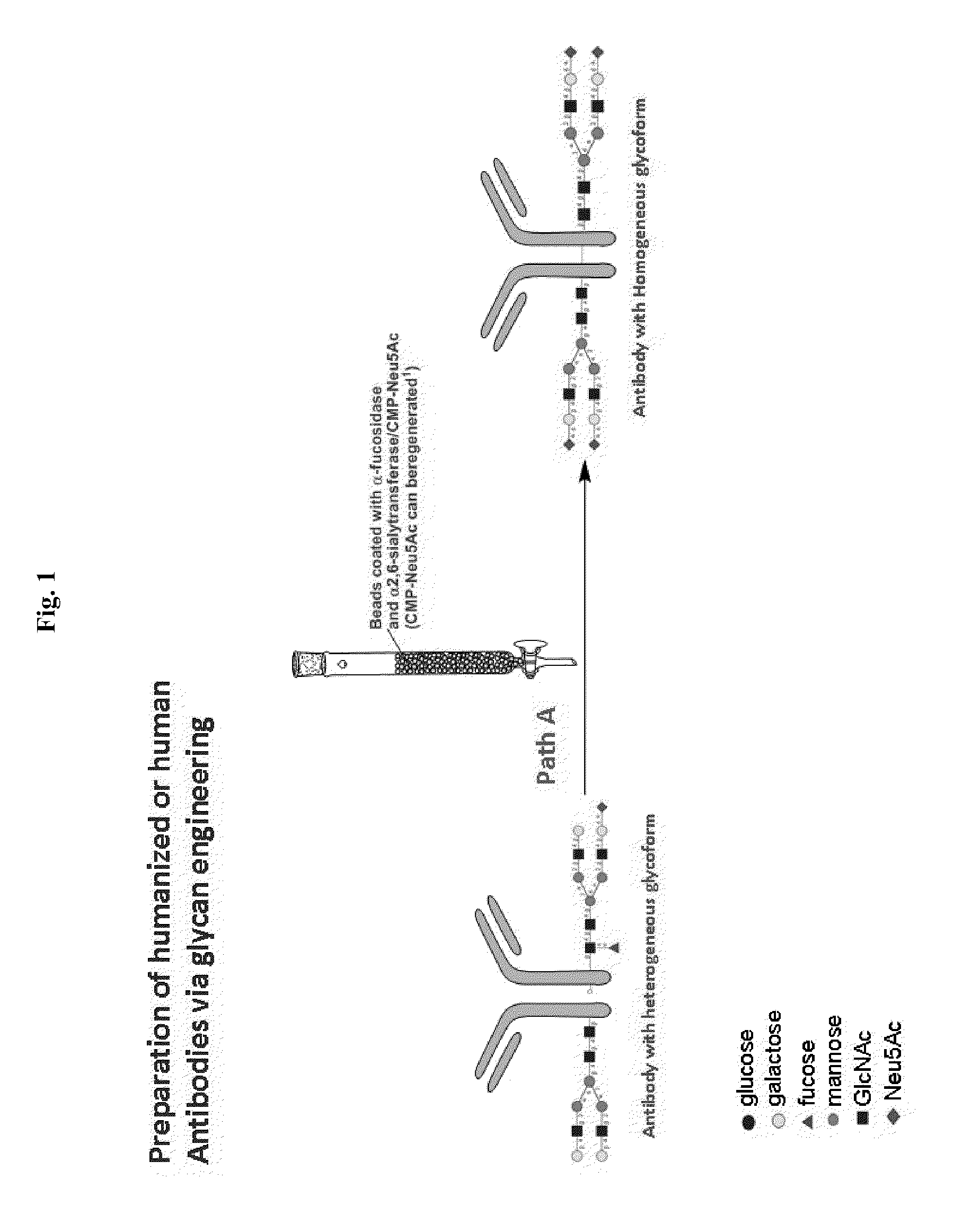
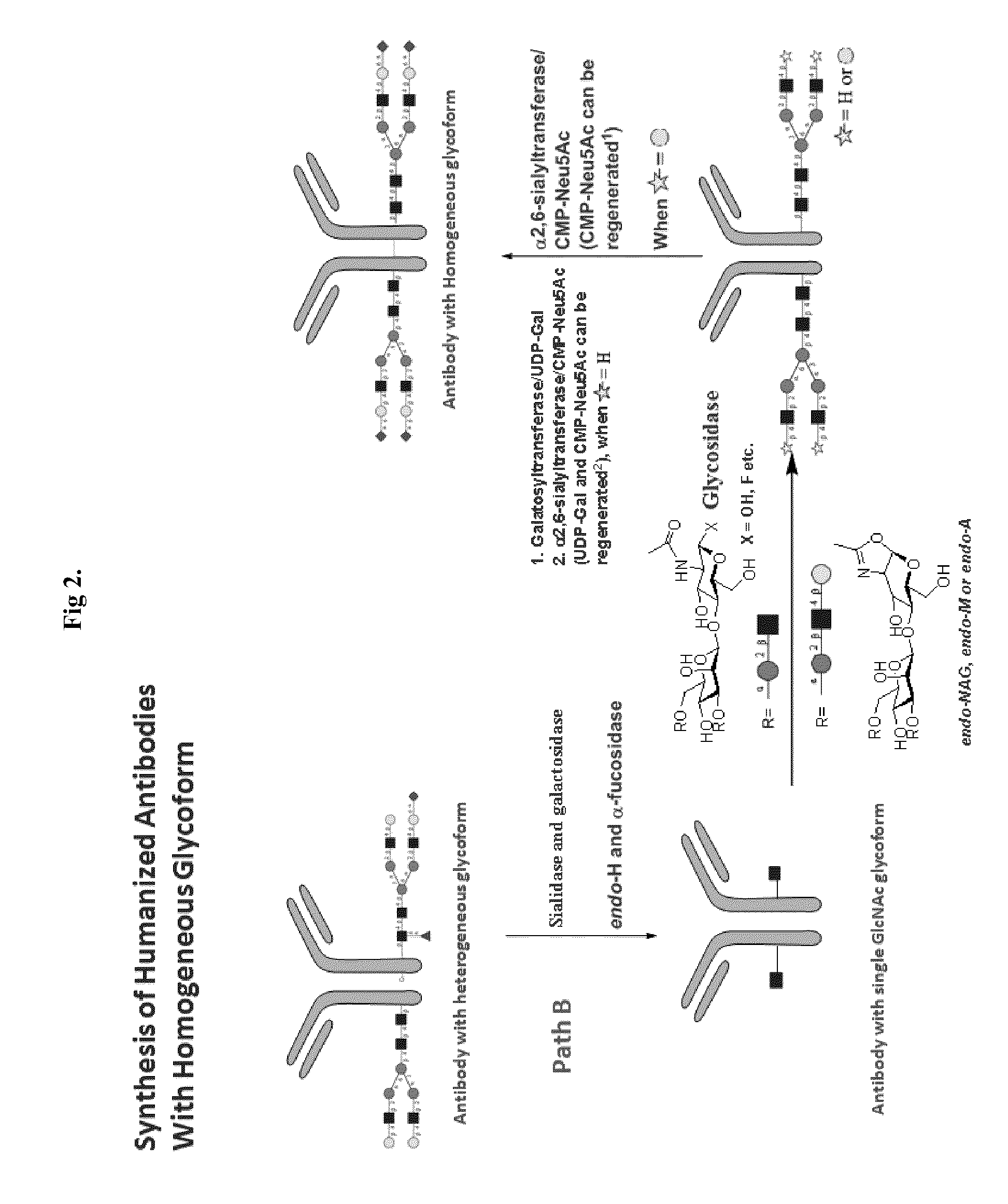
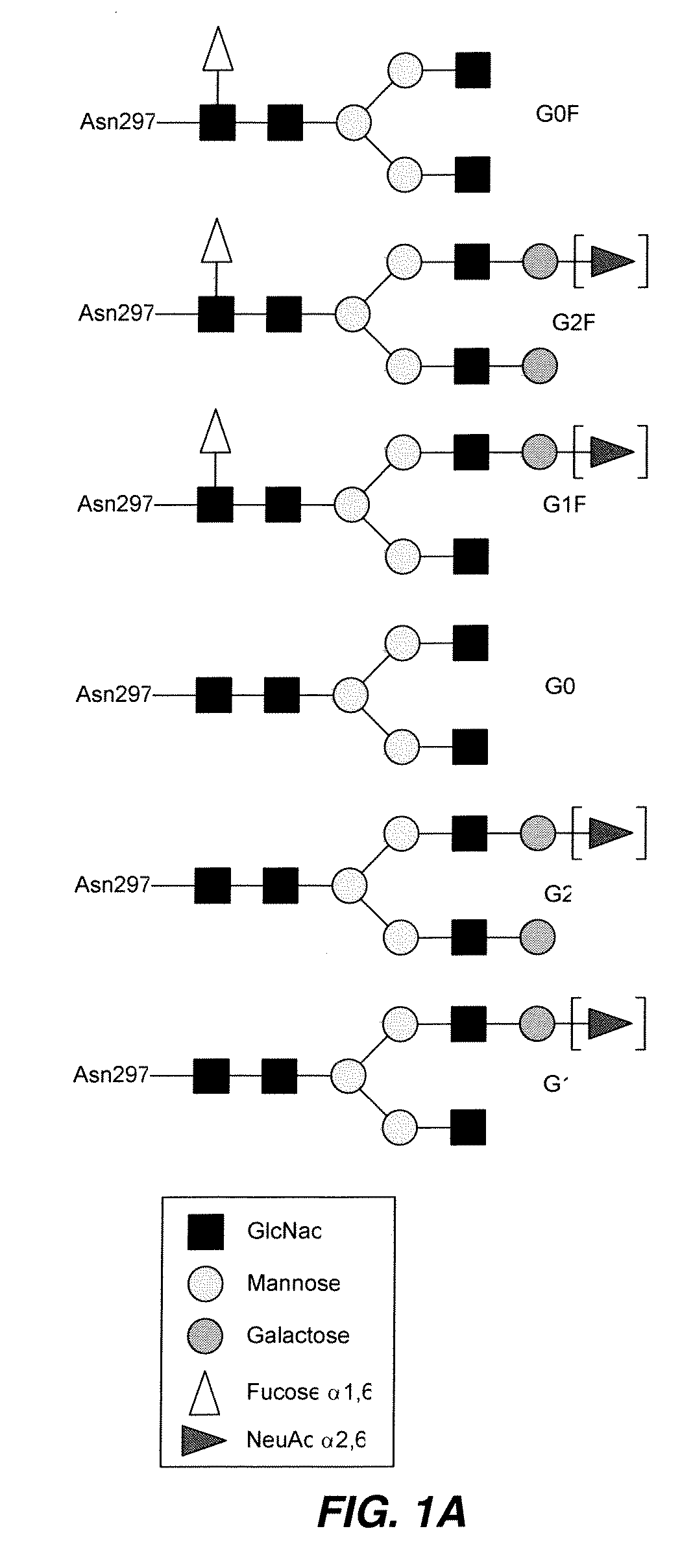
Methods for modifying human antibodies by glycan engineering
ActiveUS20110263828A1Improve efficacyImprove stabilityImmunoglobulinsFermentationGlycanAntibody fragments
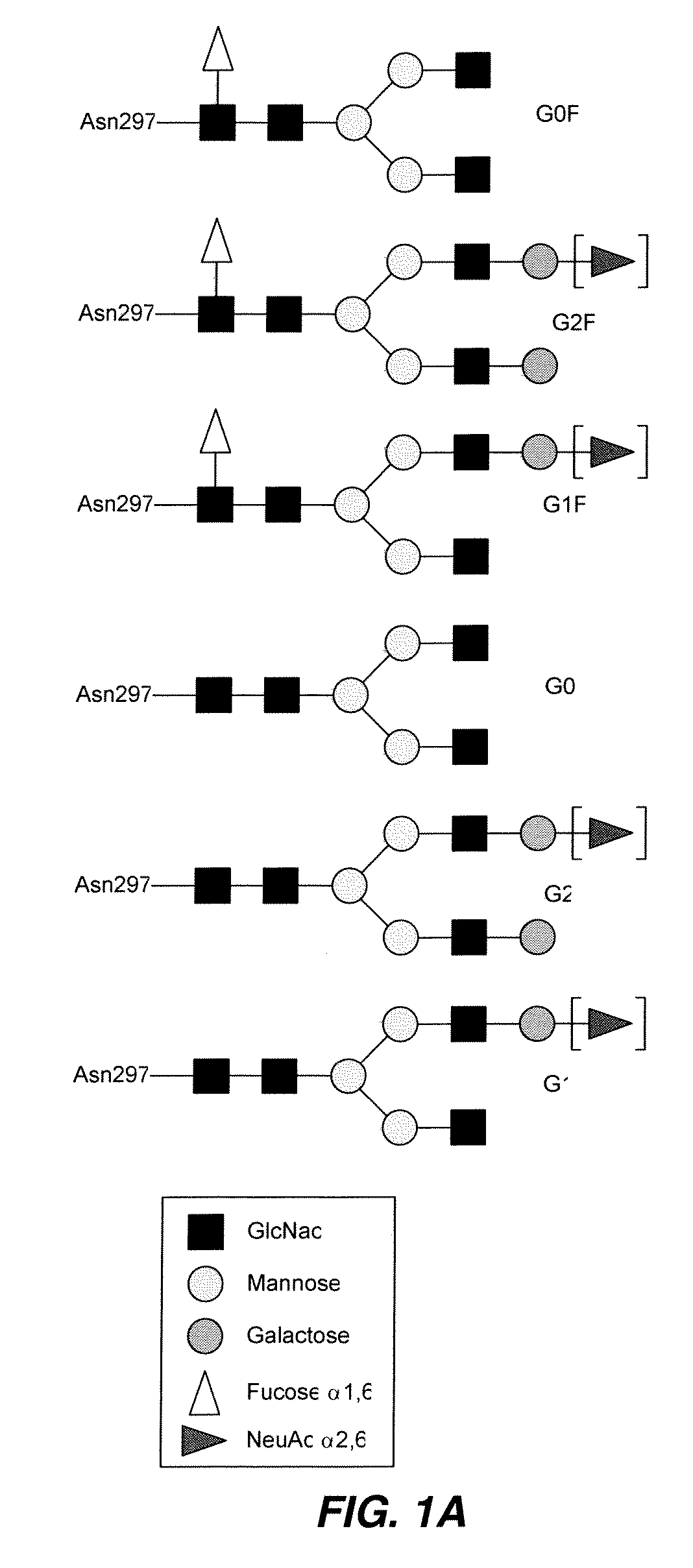
Modified Fc regions of antibodies and antibody fragments, both human and humanized, and having enhanced stability and efficacy, are provided. Fc regions with core fucose residues removed, and attached to oligosaccharides comprising terminal sialyl residues, are provided. Antibodies comprising homogeneous glycosylation of Fc regions with specific oligosaccharides are provided. Fc regions conjugated with homogeneous glycoforms of monosaccharides and trisaccharides, are provided. Methods of preparing human antibodies with modified Fc using glycan engineering, are provided.
Owner:ACAD SINIC
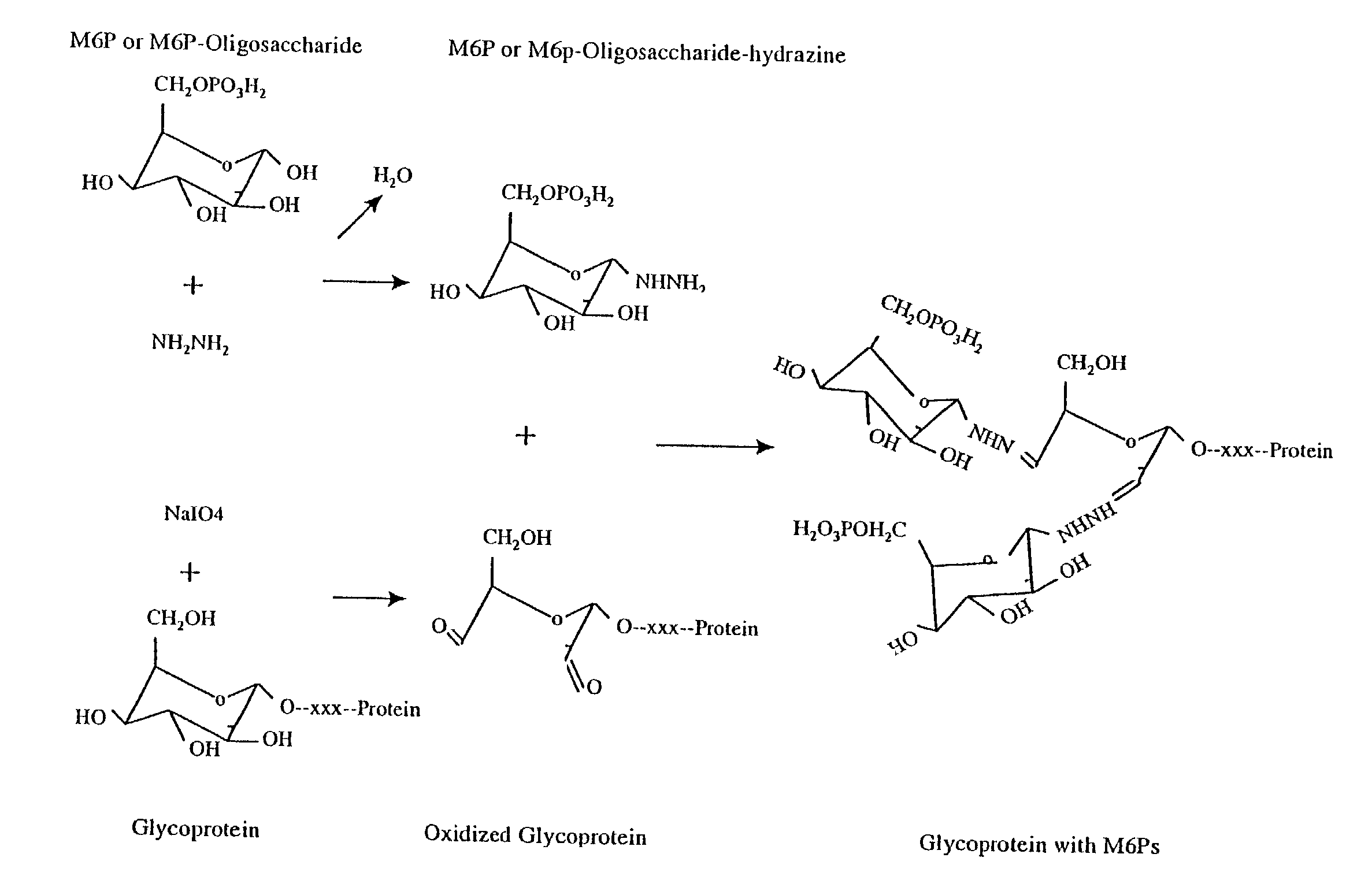
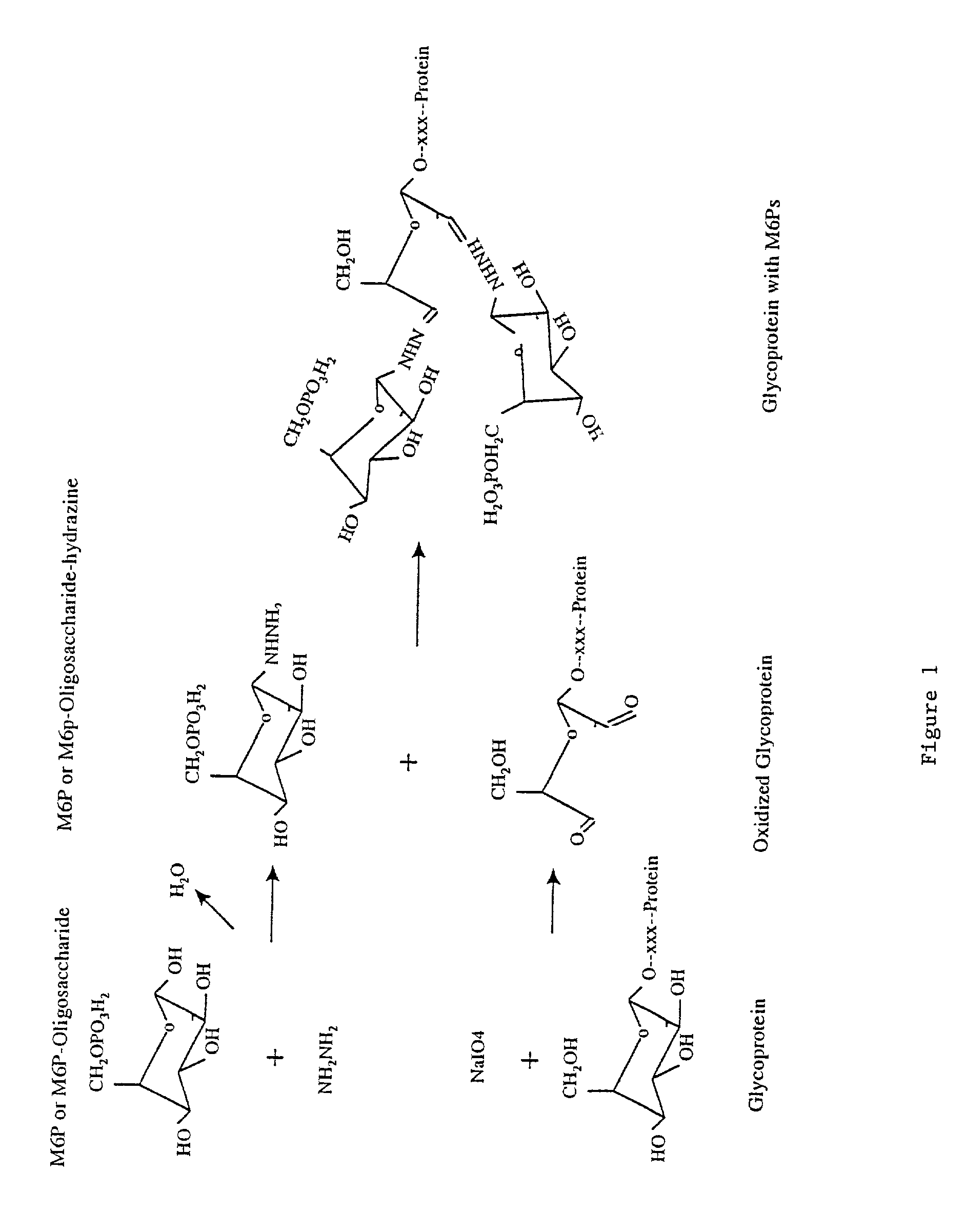
Methods for introducing mannose 6-phosphate and other oligosaccharides onto glycoproteins
InactiveUS7001994B2Well formedIncrease the cellular uptake of lysosomal enzymesHydrolasesPeptide/protein ingredientsPhosphorylationPhosphoric acid
Methods to introduce highly phosphorylated mannopyranosyl oligosaccharide derivatives containing mannose 6-phosphate (M6P), or other oligosaccharides bearing other terminal hexoses, to carbonyl groups on oxidized glycans of glycoproteins while retaining their biological activity are described. The methods are useful for modifying glycoproteins, including those produced by recombinant protein expression systems, to increase uptake by cell surface receptor-mediated mechanisms, thus improving their therapeutic efficacy in a variety of applications.
Owner:GENZYME CORP
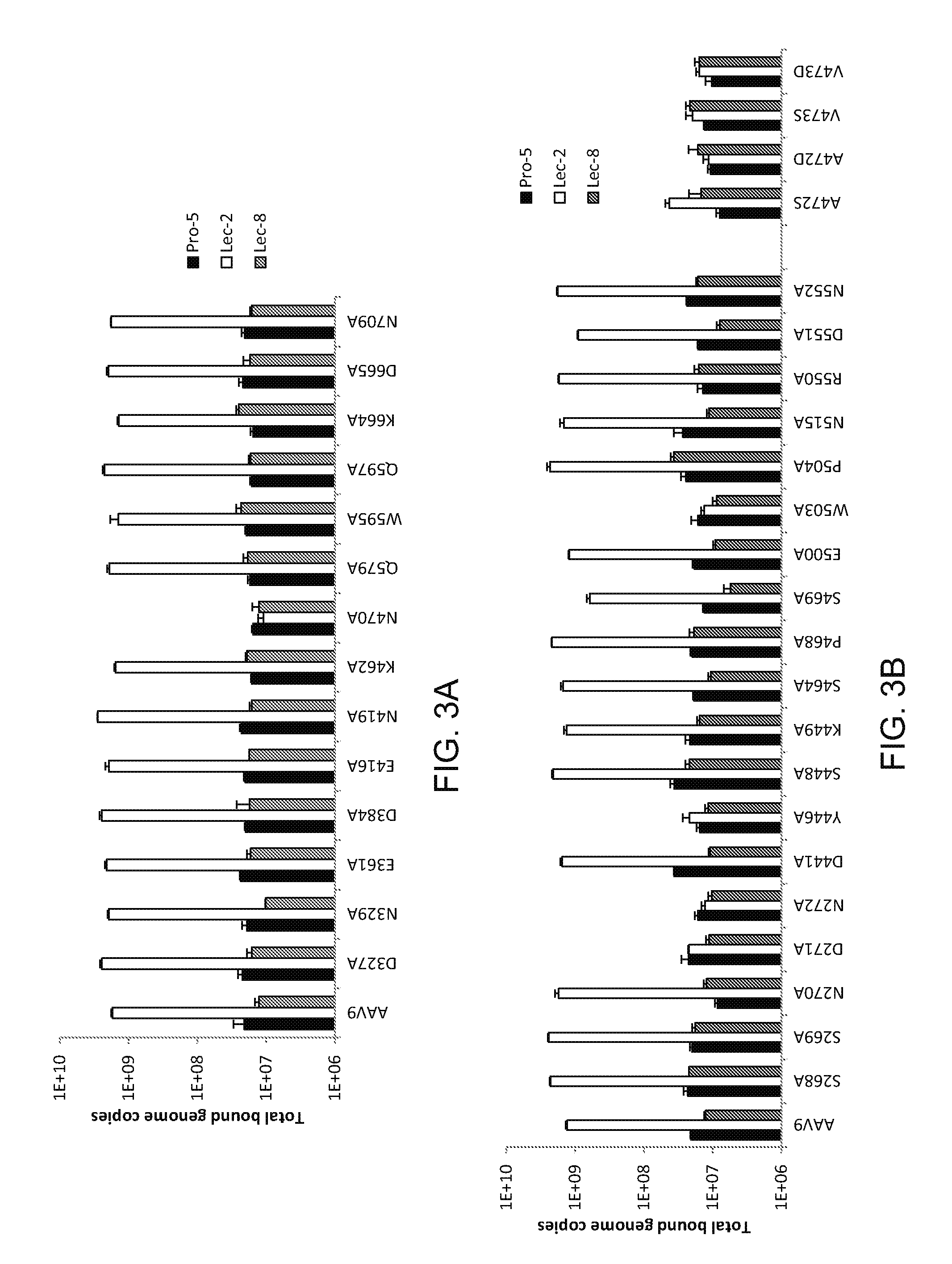
Compositions and Methods for Altering Tissue Specificity and Improving AAV9-Mediated Gene Transfer
ActiveUS20130323226A1Improve efficiencyImprove usabilityOrganic active ingredientsVectorsViral vectorFhit gene
A method of altering the targeting and / or cellular uptake efficiency of an adeno-associated virus (AAV) viral vector having a capsid containing an AAV9 cell surface binding domain is described. The method involves modifying a clade F cell surface receptor which comprises a glycan having a terminal sialic acid residue and a penultimate β-galactose residue. The modification may involve retargeting the vector by temporarily functionally ablate AAV9 binding in a subset of cells, thereby redirecting the vector to another subset of cells. Alternatively, the modification may involve increasing cellular update efficiency by treating the cells with a neuraminidase to expose cell surface β-galactose. Also provided are compositions containing the AAV9 vector and a neuraminidase. Also provided is a method for purifying AAV9 using β-galactose linked to solid support. Also provided are mutant vectors which have been modified to alter their targeting specificity, including mutant AAV9 in which the galactose binding domain is mutated and AAV in which an AAV9 galactose binding domain is engineered.
Owner:THE TRUSTEES OF THE UNIV OF PENNSYLVANIA
Human milk oligosaccharides to promote growth of beneficial bacteria
ActiveUS20120171165A1Improve immune system systemImprove system enteric nervous systemBiocideNervous disorderPremature thelarcheBacteroides
Disclosed are nutritional compositions including human milk oligosaccharides that can be administered to individuals including preterm infants, infants, toddlers, and children for improving gastrointestinal function and tolerance, as well as the growth of beneficial bacteria. Additional suitable methods of using the nutritional compositions including the human milk oligosaccharides are also disclosed.
Owner:ABBOTT LAB INC
Production of modified glycoproteins having multiple antennary structures
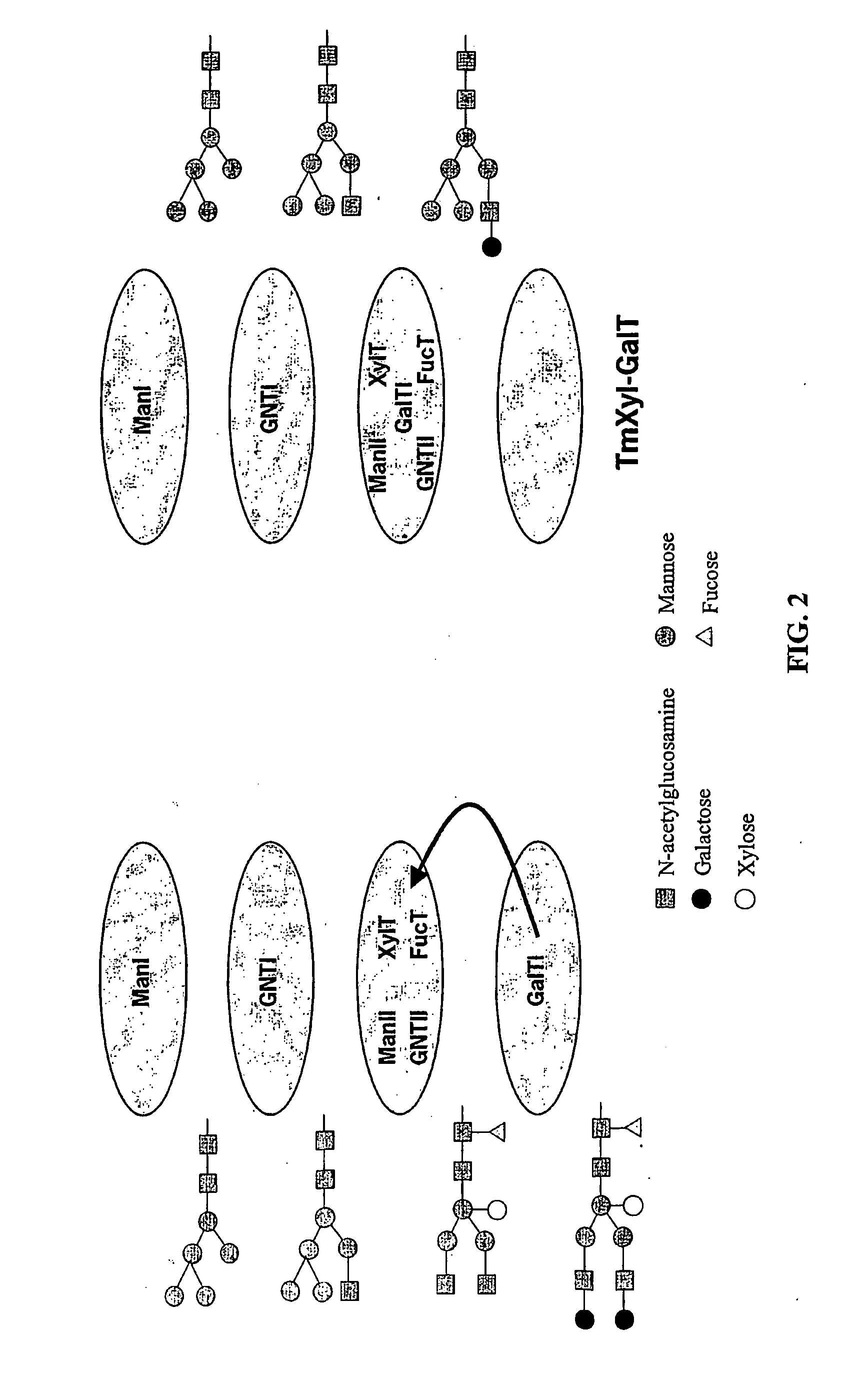
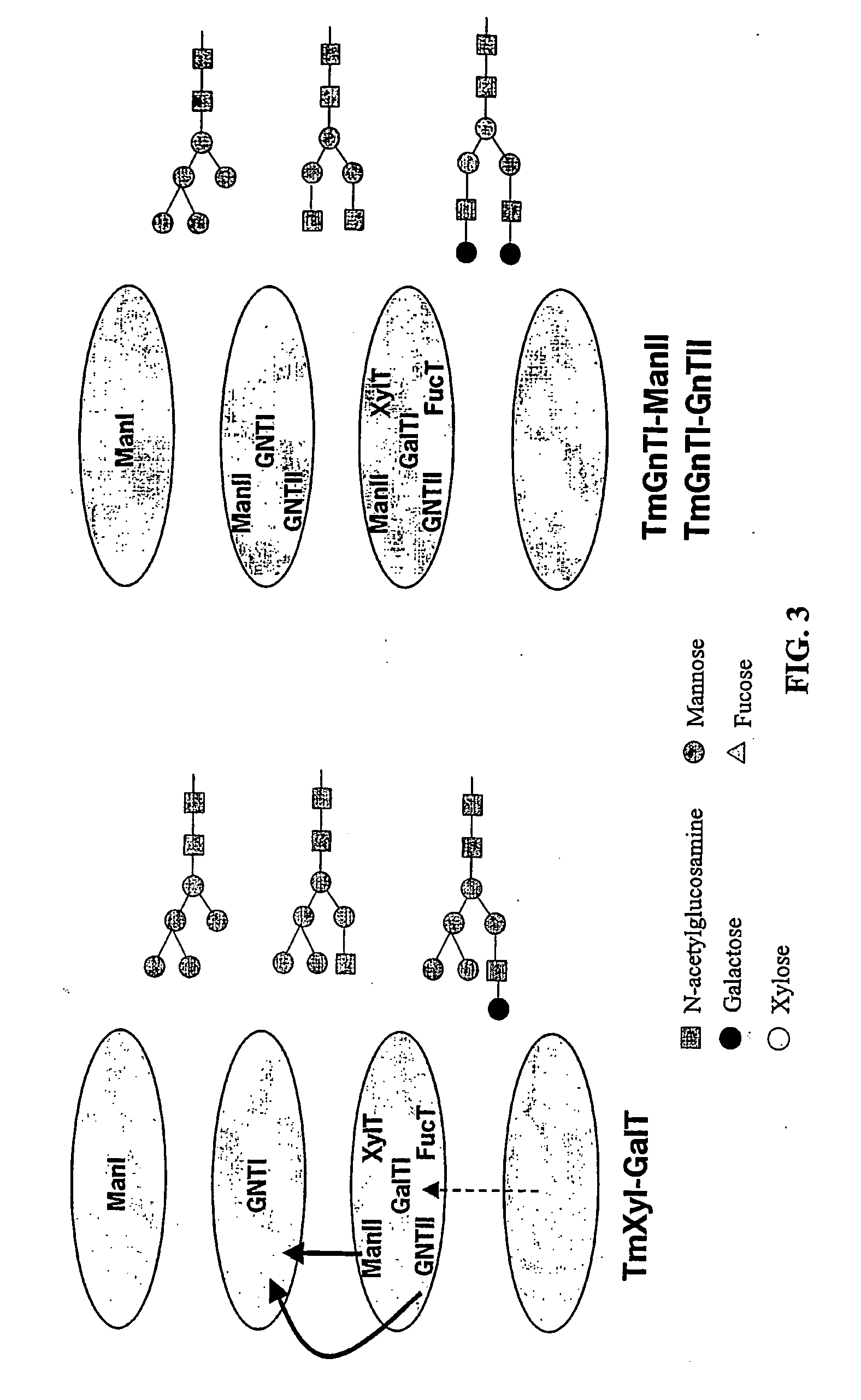
The present invention relates to eukaryotic host cells, especially lower eukaryotic host cells, having modified oligosaccharides which may be modified further by heterologous expression of a set of glycosyltransferases, sugar and sugar nucleotide transporters to become host-strains for the production of mammalian, e.g., human therapeutic glycoproteins. The process provides an engineered host cell which can be used to express and target any desirable gene(s) involved in glycosylation. Host cells with modified lipid-linked oligosaccharides are created or selected. N-glycans made in the engineered host cells exhibit GnTIII, GnTIV, GnTV, GnT VI or GnTIX activity, which produce bisected and / or multiantennary N-glycan structures and may be modified further by heterologous expression of one or more enzymes, e.g., glycosyltransferases, sugar, sugar nucleotide transporters, to yield human-like glycoproteins. For the production of therapeutic proteins, this method may be adapted to engineer cell lines in which any desired glycosylation structure may be obtained.
Owner:GLYCOFI
Nucleic acids of aspergillus fumigatus encoding industrial enzymes and methods of use
The present invention provides nucleotide sequences of Aspegillus fumigatus that encode proteins which exhibit enzyme activities. Vectors, expression constructs, and host cells comprising the nucleotide sequences of the enzyme genes are also provided. The invention further provides methods for producing the enzymes, and methods for modifying the enzymes in order to improve their desirable characteristics. The activities displayed by the enzymes of the invention include those of a tannase, cellulase, glucose oxidase, glucoamylase, phytase, beta-galactosidases, invertase, lipase, alpha-amylase, laccase, polygalacturonase or xylanase. The enzymes of the invention can be used in a variety of industrial processes. Enzymatically active compositions in various forms as well as antibodies to the enzymes and fragments thereof, are also provided.
Owner:MERCK & CO INC
MONOCLONAL ANTIBODIES WITH ALTERED AFFINITIES FOR HUMAN FCyRI, FCyRIIIa, AND C1q PROTEINS
InactiveUS20130149300A1Good curative effectLow toxicitySugar derivativesImmunoglobulins against cytokines/lymphokines/interferonsDiseaseFc-Gamma Receptor
Disclosed herein are GNGN and G1 / G2 antibodies that recognize and bind various FcRs and C1q. Also disclosed herein are glycan-optiminzed antibodies, predominantly of the GNGN or G1 / G2 glycoform, with enhanced Fcγ receptor binding achieved through CHO, Nicotiana benthamiana and yeast manufacturing systems. Nucleic acids encoding these antibodies, as well as expression vectors and host cells including these nucleic acids are also disclosed herein. Methods and pharmaceutical compositions including the monoclonal antibodies are provided herein for the prevention and / or therapeutic treatment of viral infections, cancers and inflammatory diseases.
Owner:MAPP BIOPHARM +1
N-acetylglucosamintransferase III expression in lower eukaryotes
InactiveUS20050208617A1The overall structure is closedFungiPeptide/protein ingredientsLipid formationHeterologous
The present invention relates to eukaryotic host cells having modified oligosaccharides which may be modified further by heterologous expression of a set of glycosyltransferases, sugar transporters and mannosidases to become host-strains for the production of mammalian, e.g., human therapeutic glycoproteins. The process provides an engineered host cell which can be used to express and target any desirable gene(s) involved in glycosylation. Host cells with modified lipid-linked oligosaccharides are created or selected. N-glycans made in the engineered host cells exhibit GnTIII activity, which produce bisected N-glycan structures and may be modified further by heterologous expression of one or more enzymes, e.g., glycosyltransferases, sugar transporters and mannosidases, to yield human-like glycoproteins. For the production of therapeutic proteins, this method may be adapted to engineer cell lines in which any desired glycosylation structure may be obtained.
Owner:GLYCOFI
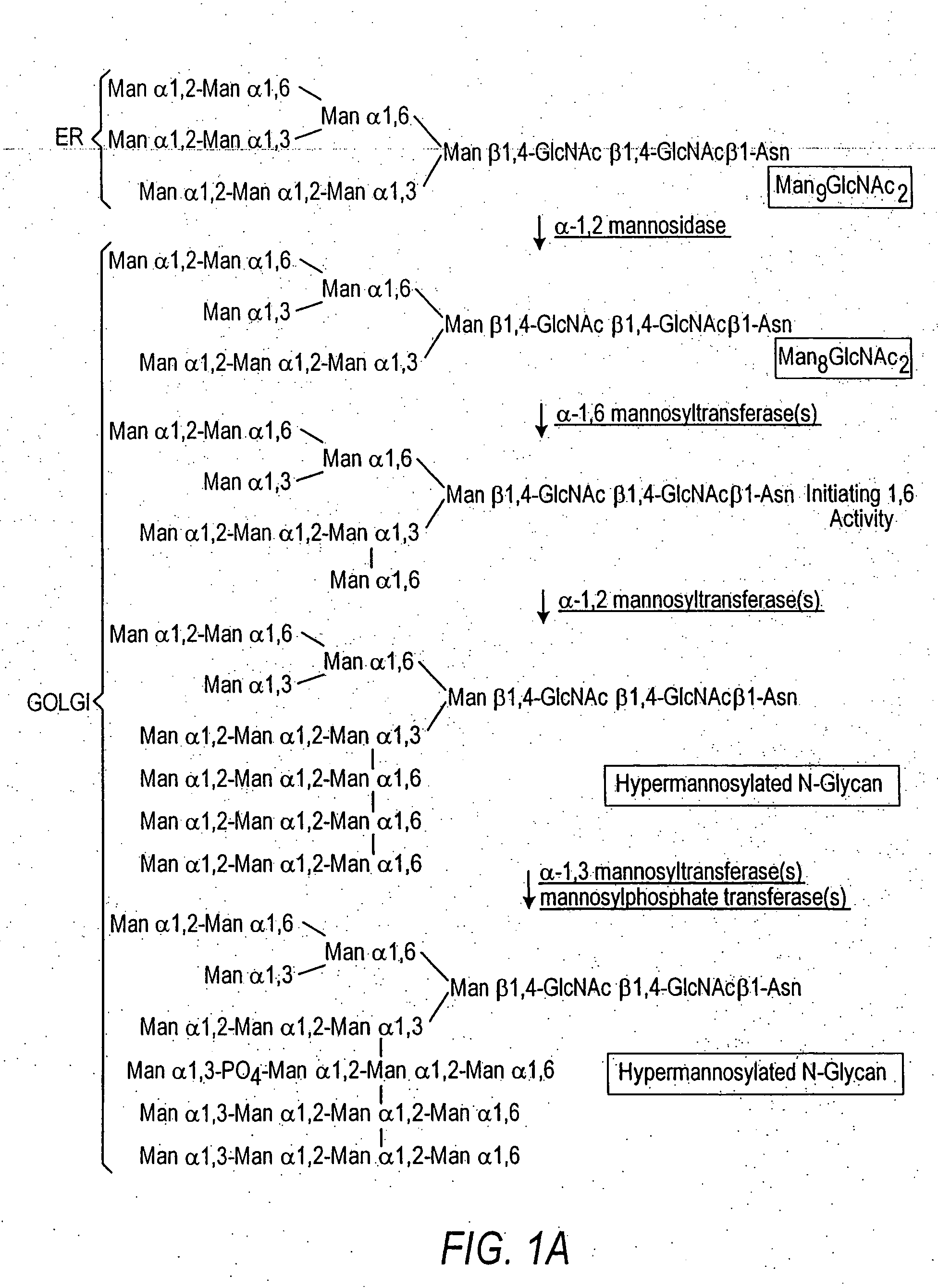
Methods for eliminating mannosylphosphorylation of glycans in the production of glycoproteins
The present invention relates to the elimination of mannosylphosphorylation on the glycans of glycoproteins in the yeast genus Pichia. The elimination of mannosylphosphorylated glycoproteins results from the disruption of the PNO1 gene and the newly isolated P. pastoris MNN4B gene. The present invention further relates to methods for producing modified glycan structures in host cells that are free of glycan mannosylphosphorylation.
Owner:GLYCOFI
Preparation and use of sulfated oligosaccharides
PCT No. PCT / AU96 / 00238 Sec. 371 Date Oct. 28, 1997 Sec. 102(e) Date Oct. 28, 1997 PCT Filed Apr. 24, 1996 PCT Pub. No. WO96 / 33726 PCT Pub. Date Oct. 31, 1996Sulfated oligosaccharides, wherein the oligosaccharide has the general formula I:R1-(Rx)n-R2(I)wherein R1 and R2 and each Rx represents a monosaccharide unit, all of which may be the same or different, adjacent monosaccharide units being linked by 1->2, 1->3, 1->4 and / or 1->6 glycosidic bonds and n is an integer of from 1 to 6, and use thereof as anti-angiogenic, anti-metastatic and / or anti-inflammatory agents.
Owner:AUSTRALIEN NAT UNIV
Methods for eliminating mannosylphosphorylation of glycans in the production of glycoproteins
The present invention relates to the elimination of mannosylphosphorylation on the glycans of glycoproteins in the yeast genus Pichia. The elimination of mannosylphosphorylated glycoproteins results from the disruption of the PNO1 gene and the newly isolated P. pastoris MNN4B gene. The present invention further relates to methods for producing modified glycan structures in host cells that are free of glycan mannosylphosphorylation.
Owner:GLYCOFI
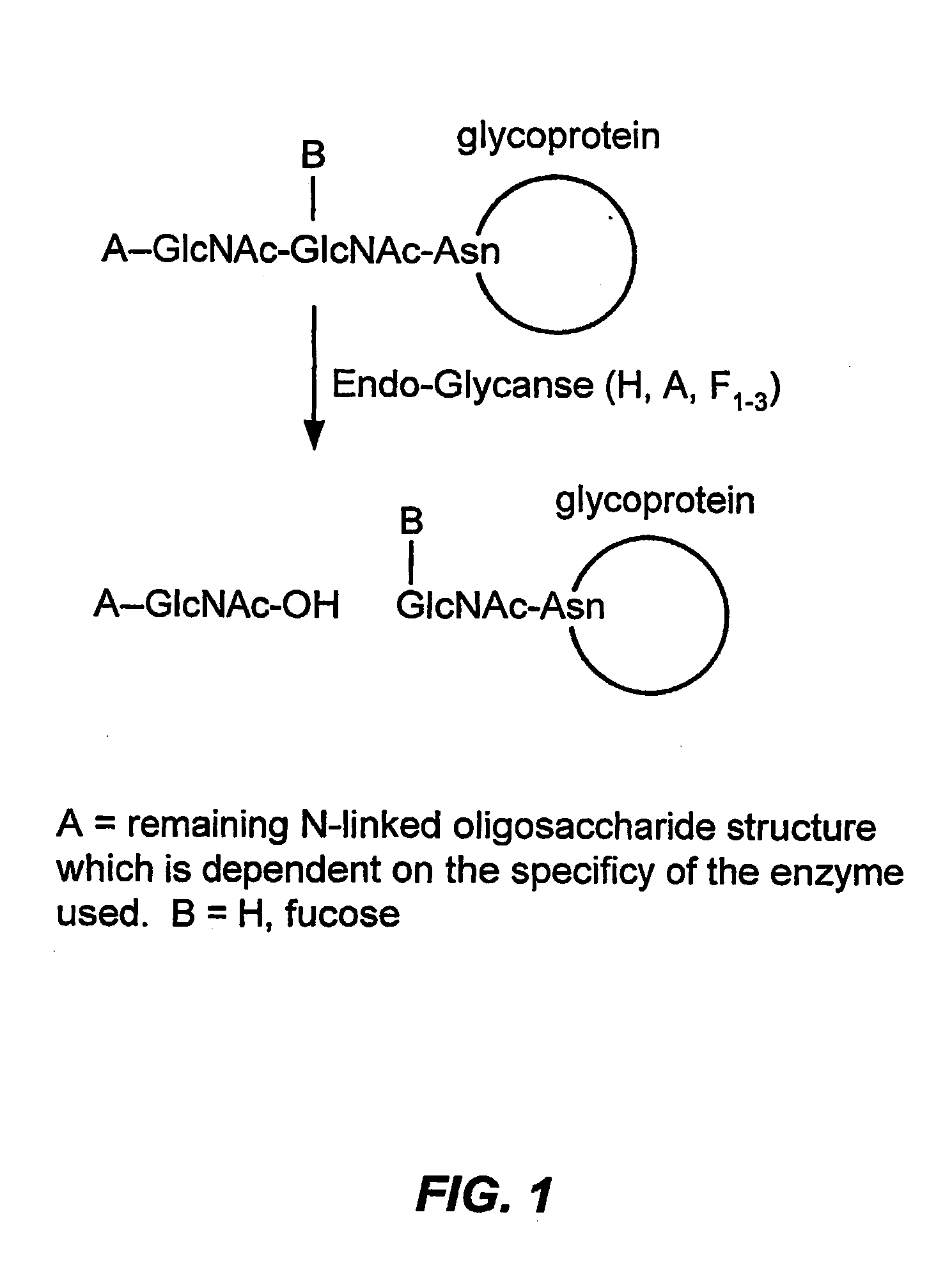
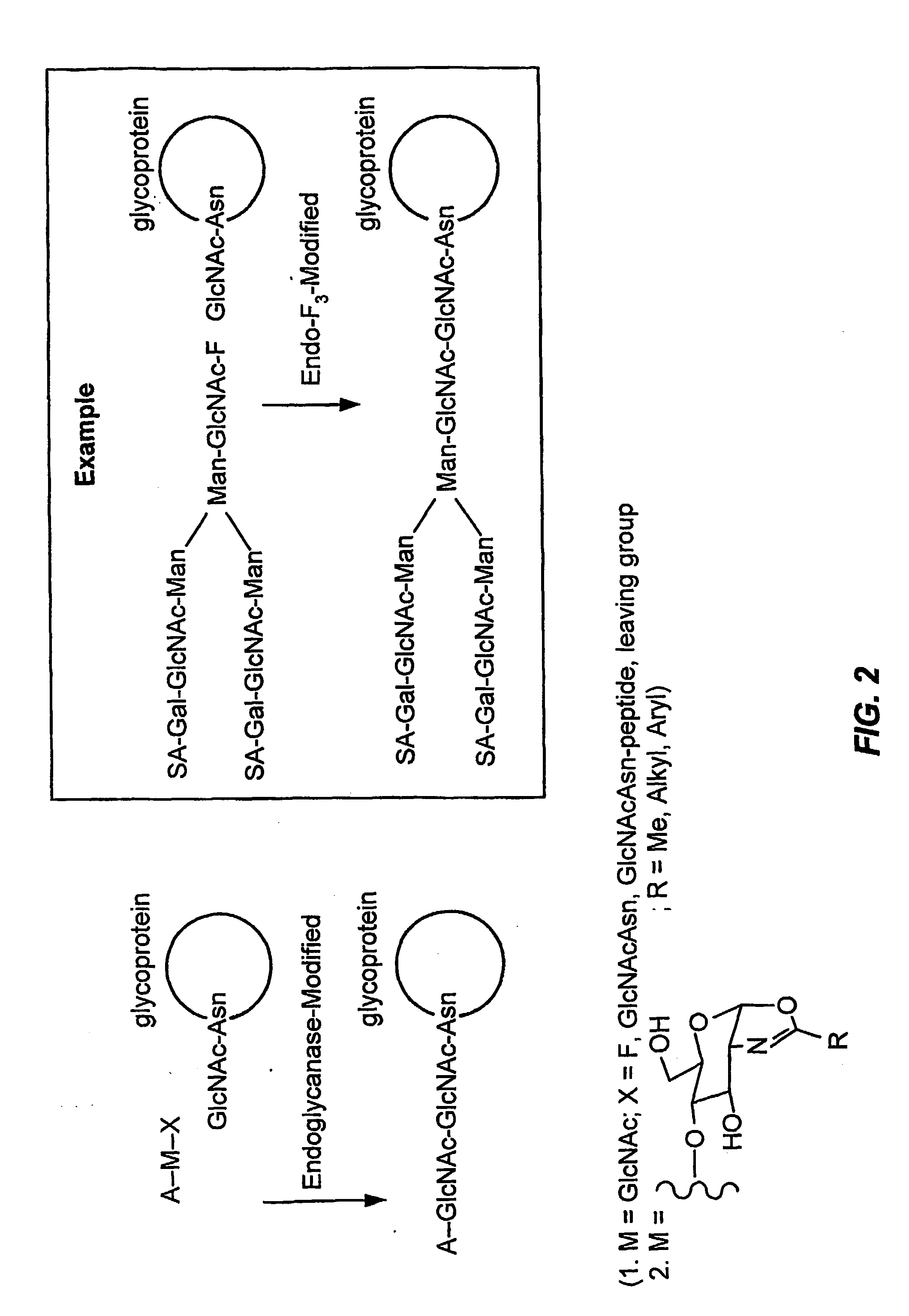
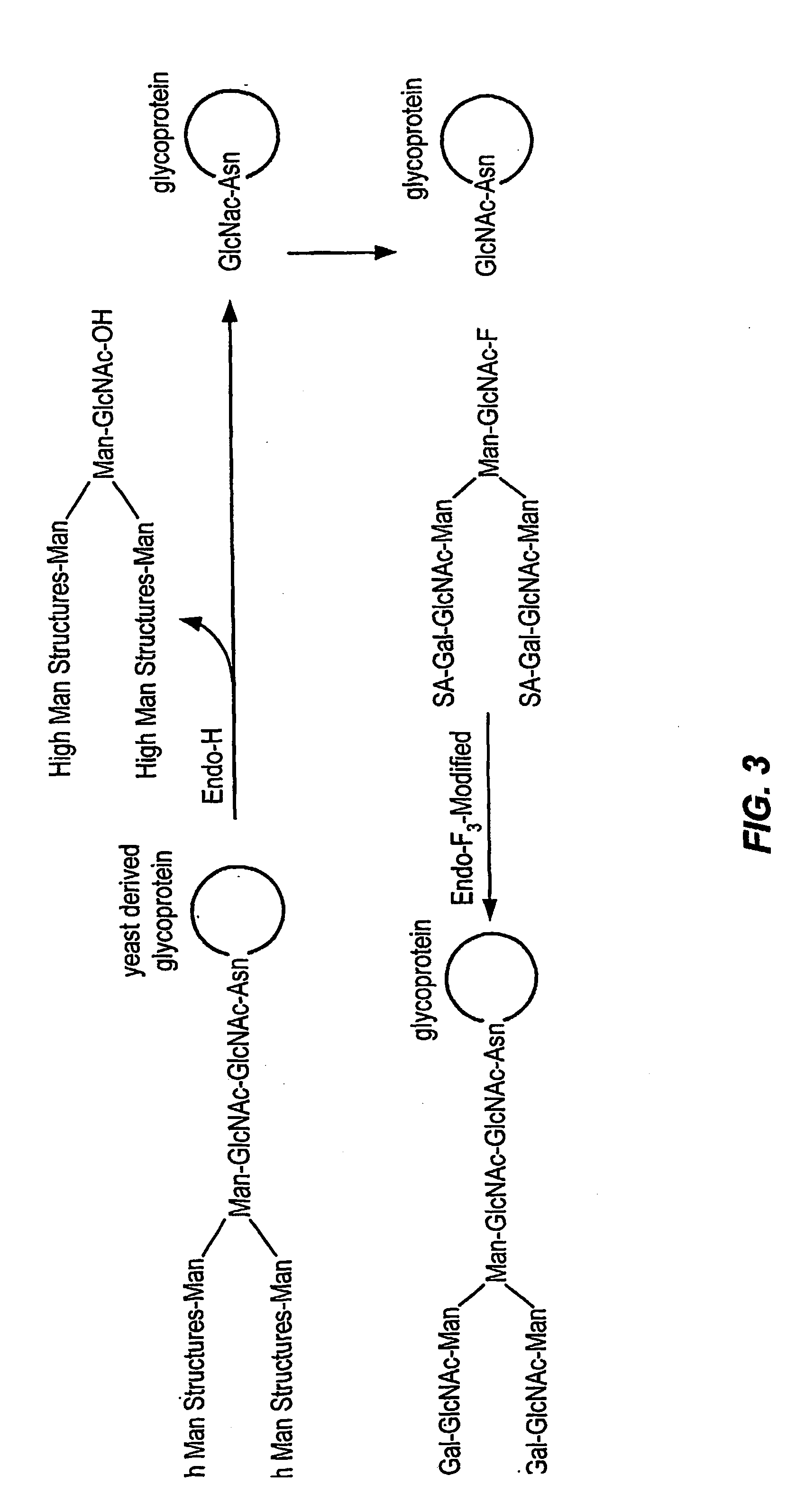
Glycoprotein remodeling using endoglycanases
This invention provides methods for modifying glycosylation patterns of glycoproteins, including recombinantly produced glycoproteins. Also provided are glycoprotein compositions in which the glycoproteins have a homogeneous glycosylation pattern.
Owner:RATIOPHARM GMBH
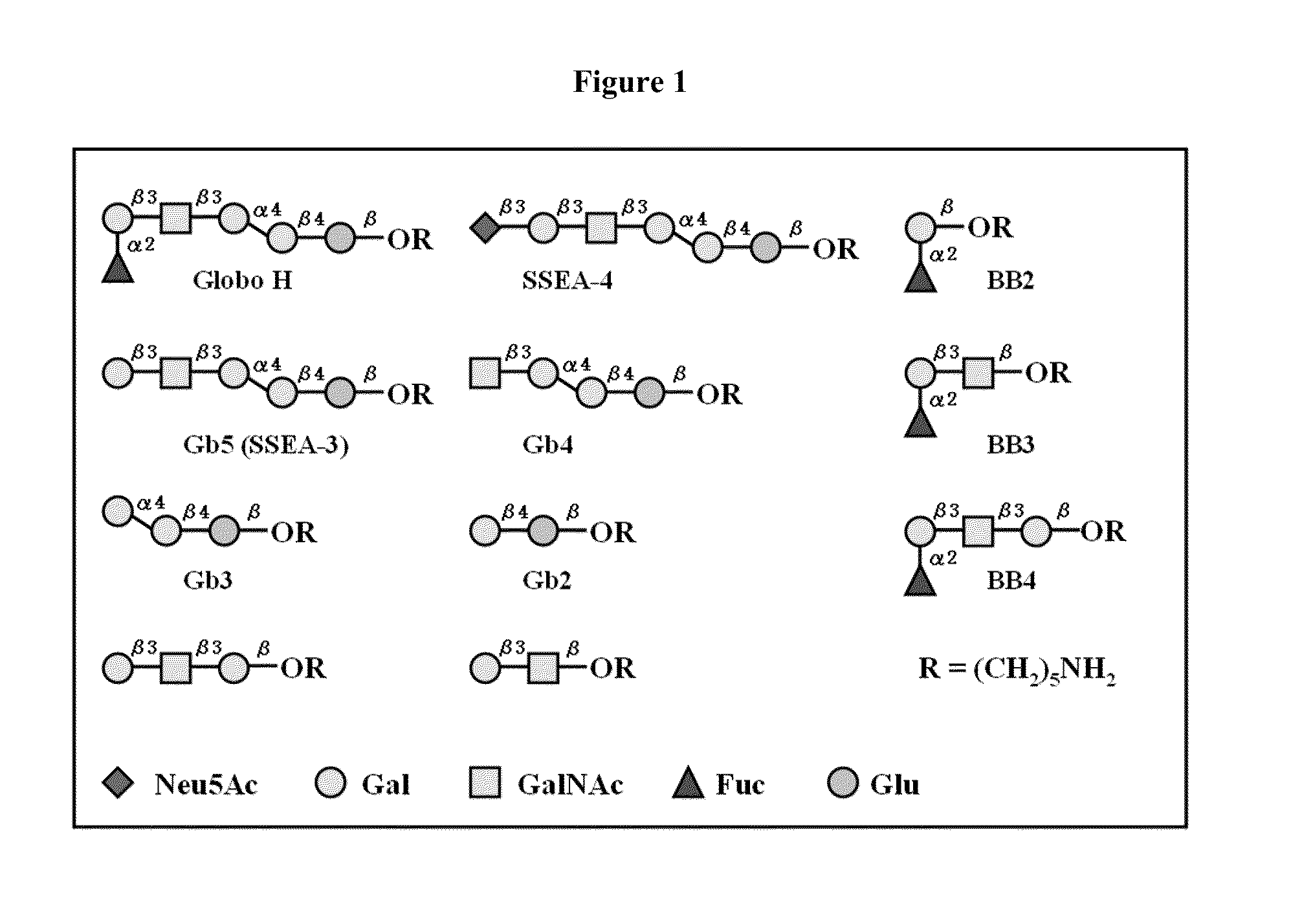
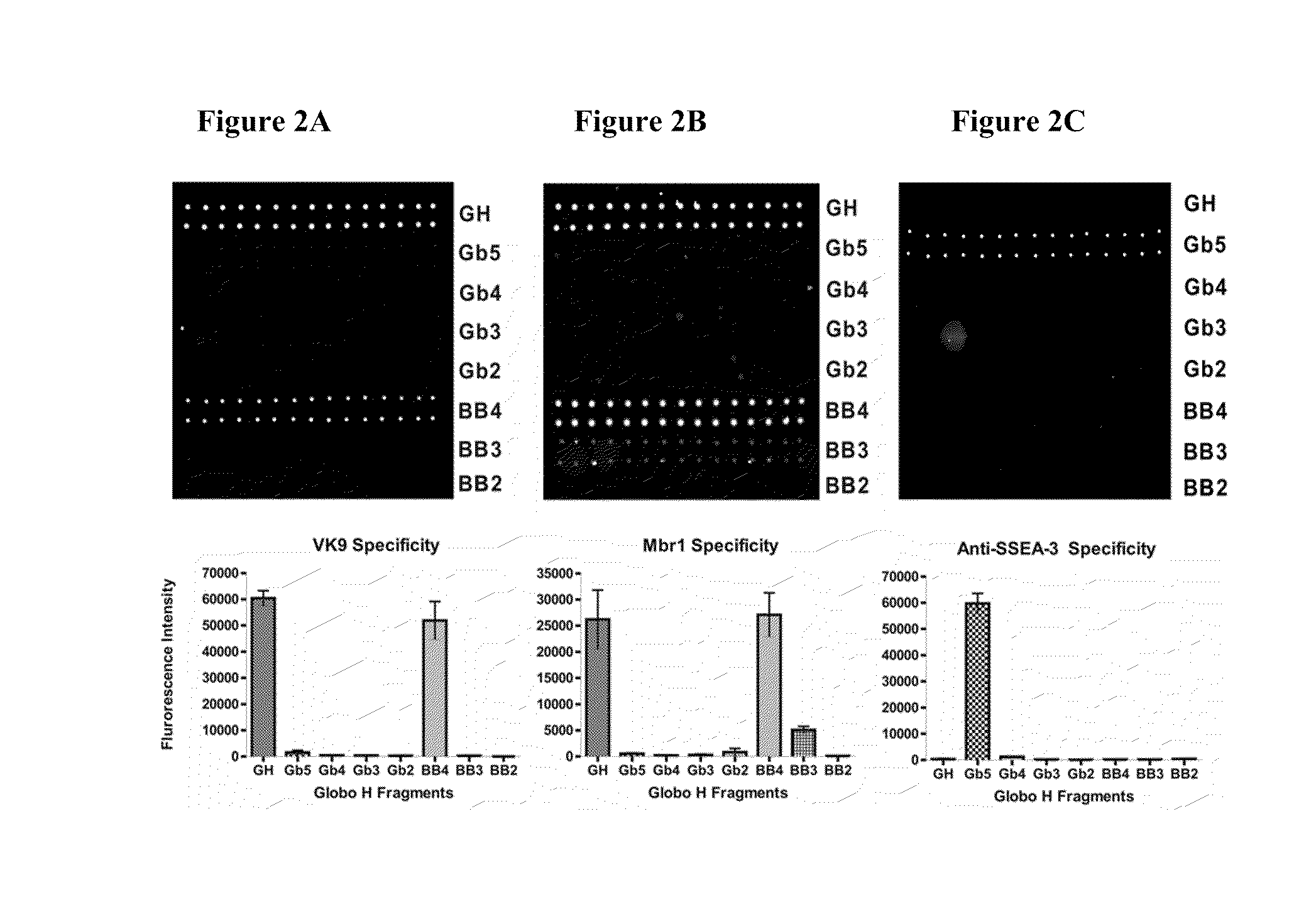
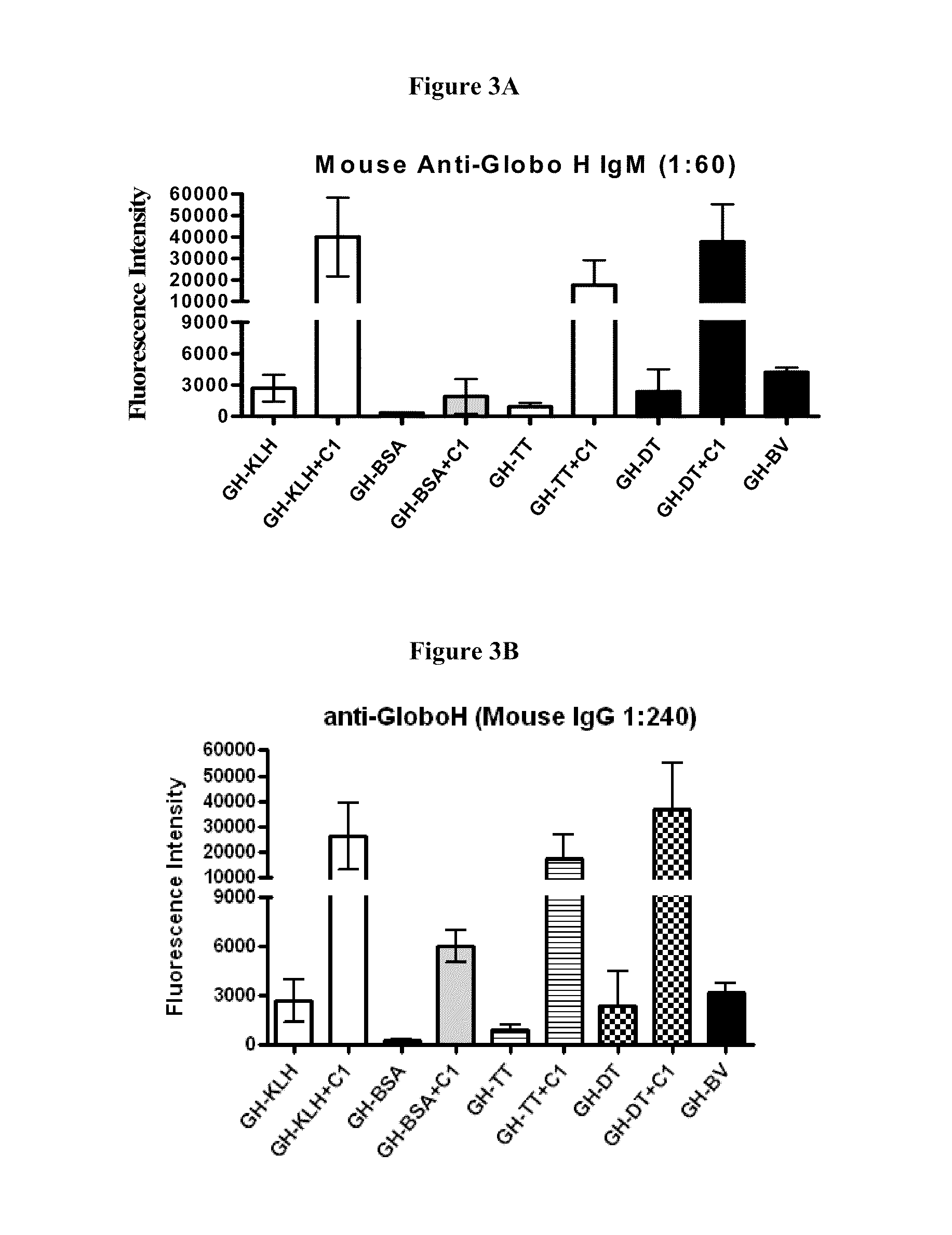
Globo h and related Anti-cancer vaccines with novel glycolipid adjuvants
ActiveUS20100136042A1Shrink tumorInhibit tumor growthOrganic active ingredientsSugar derivativesDendritic cellAdjuvant
Owner:ACAD SINIC
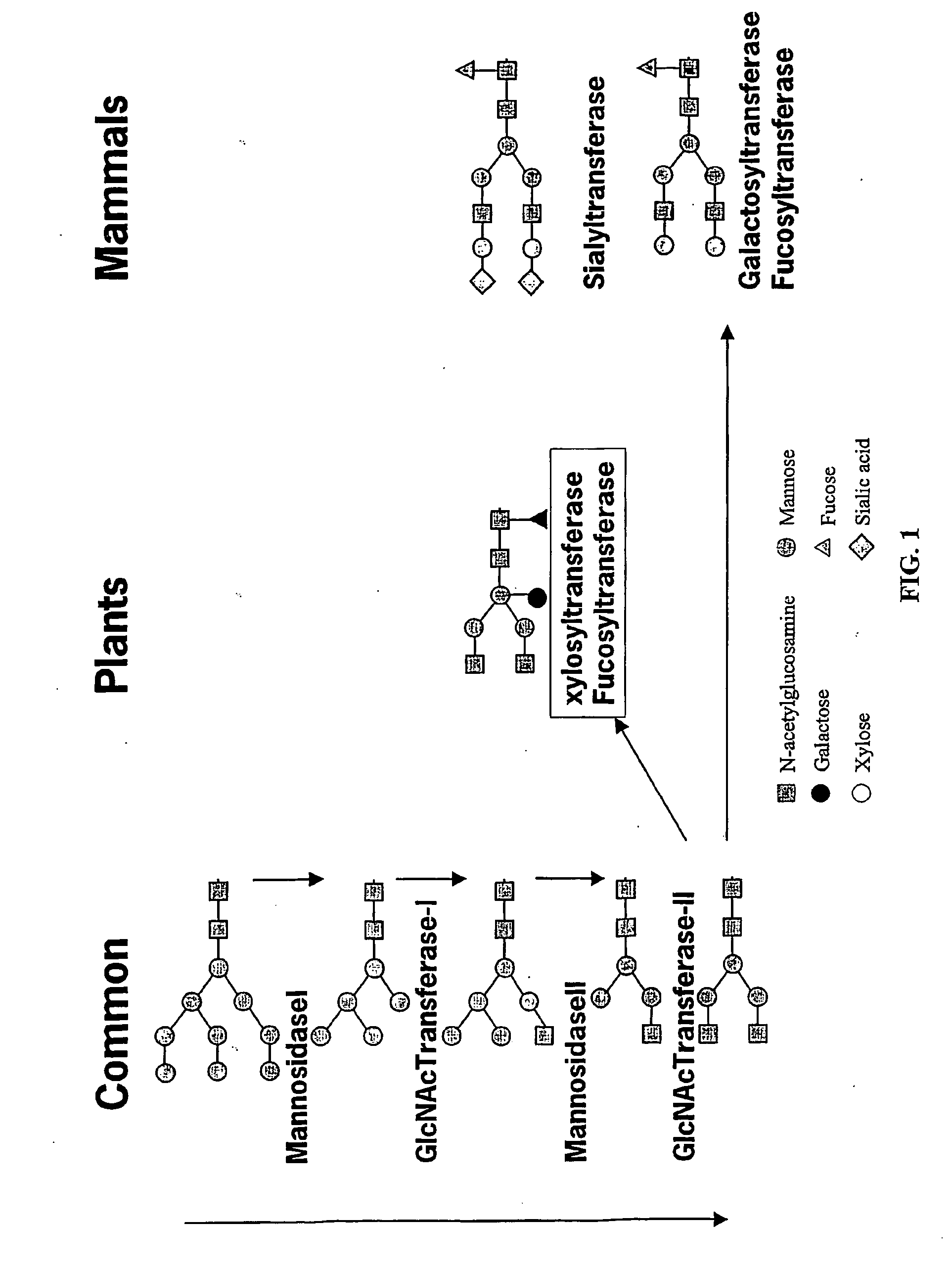
Compositions and methods for humanization and optimization of n-glycans in plants
InactiveUS20080066200A1Function increaseHigh antibody activityAntipyreticAnalgesicsGrowth plantGlycan
Methods for altering the N-glycosylation pattern of proteins in higher plants are provided. The methods comprise introducing into the plant a recombinant construct that provides for the inhibition of expression of α1,3-fucosyltransferase (FucT) and β1,2-xylosyltransferase (XylT) in a plant. Use of these constructs to inhibit or suppress expression of both of these enzymes, and isoforms thereof, advantageously provides for the production of endogenous and heterologous proteins having a “humanized” N-glycosylation pattern without impacting plant growth and development. Stably transformed higher plants having this protein N-glycosylation pattern are provided. Glycoprotein compositions, including monoclonal antibody compositions, having substantially homogeneous glycosylation profiles, and which are substantially homogeneous for the G0 glycoform, are also provided.
Owner:SYNTHON BIOPHARMACEUTICALS BV +1
Optimizing glycan processing in plants
InactiveUS20060253928A1Reduce diversityGalactosylation is improvedFungiBacteriaBiological bodyComplex type
The invention is directed to methods for optimizing glycan processing in organisms (and in particular, plants) so that a glycoprotein having complex type bi-antennary glycans and thus 5 containing galactose residues on both arms and which are devoid of (or reduce in) xylose and fucose can be obtained. The invention is further directed to said glycoprotein obtained and host system comprising said protein.
Owner:STICHTING DIENST LANBOUWKUNDIG ONDERZOEK
Glycan-interacting compounds
The present invention provides glycan-interacting antibodies useful in the treatment and prevention of human disease, including cancer. Such glycan-interacting antibodies include monoclonal antibodies, derivatives and fragments thereof as well as compositions and kits comprising them.
Owner:SEAGEN INC
Antibody-based therapeutics with enhanced ADCC activity
Owner:GENZYME CORP
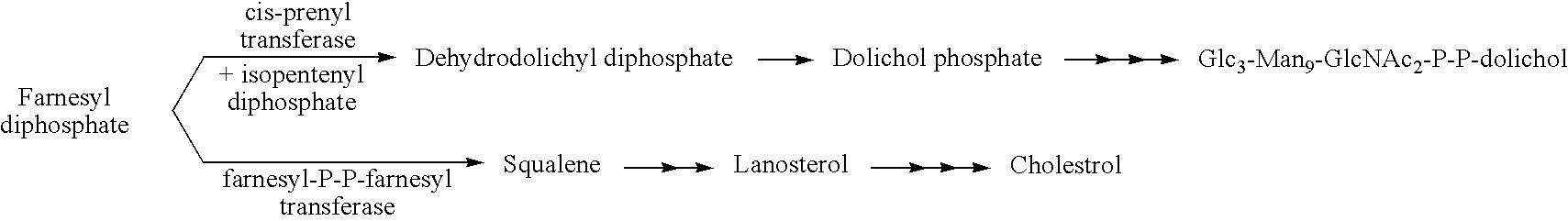
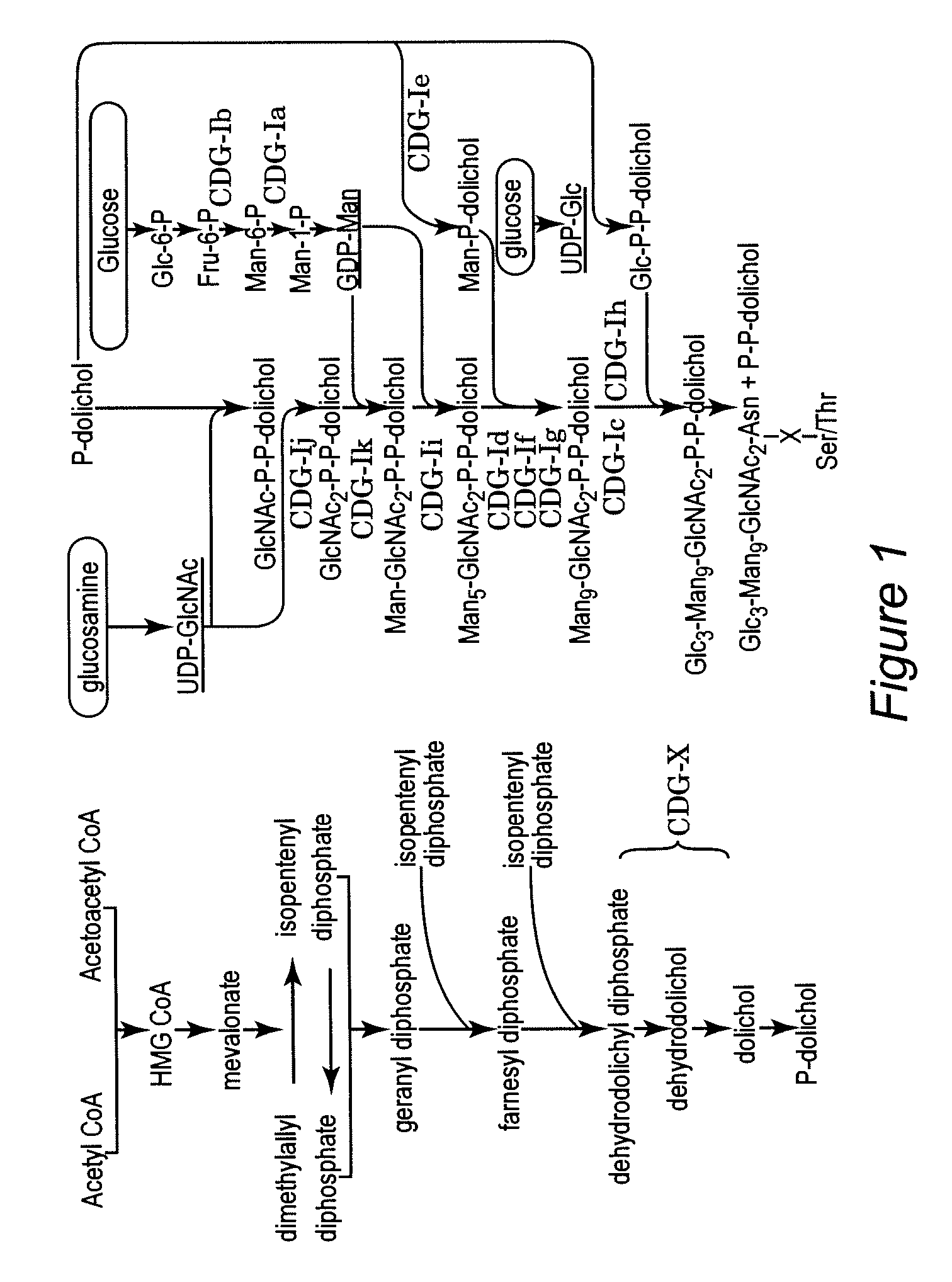
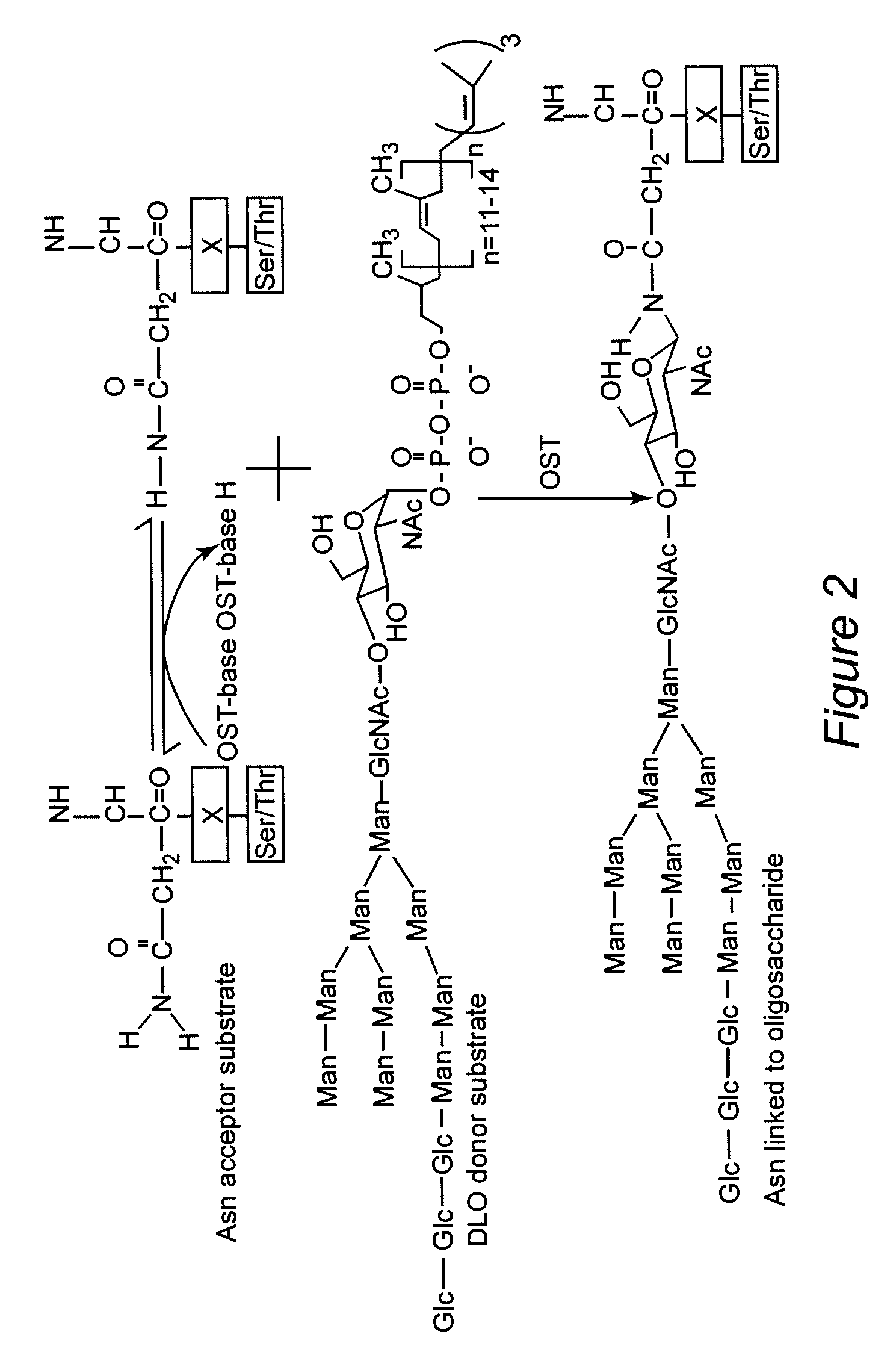
Protein N-glycosylation of eukaryotic cells using dolichol-linked oligosaccharide synthesis pathway, other N-gylosylation-increasing methods, and engineered hosts expressing products with increased N-glycosylation
InactiveUS20060252672A1Increase the number ofImprove accessibilityNervous disorderMetabolism disorderLipid formationHEK 293 Cell Line
The level of glycosylation on products produced by a host (such as CHO cells, HEK cells and other mammalian cells, and non-mammalian cells) or patient can be increased by engineering, such as by supplying the host or patient with a gene sequence. For example, the host or patient can be made to produce desirably glycosylated products by increasing one or both of expression of N-glycan substrate containing lipid-liked oligosaccharide and expression of oligosaccharide (OST) transferase.
Owner:THE JOHN HOPKINS UNIV SCHOOL OF MEDICINE
Antibody-based therapeutics with enhanced ADCC activity
Methods for producing antibody-based therapeutics with enhanced ADCC activity are disclosed. The enhanced ADCC activity is attributed to oligomannose-type N-glycans on the antibodies and Fc fusion proteins of the invention. Also disclosed are methods of using such antibody-based therapeutics for targeted killing of cells in a mammal, including therapeutic methods of treating cancers, autoimmune diseases and other diseases.
Owner:GENZYME CORP
Process for producing molecules containing specialized glycan structures
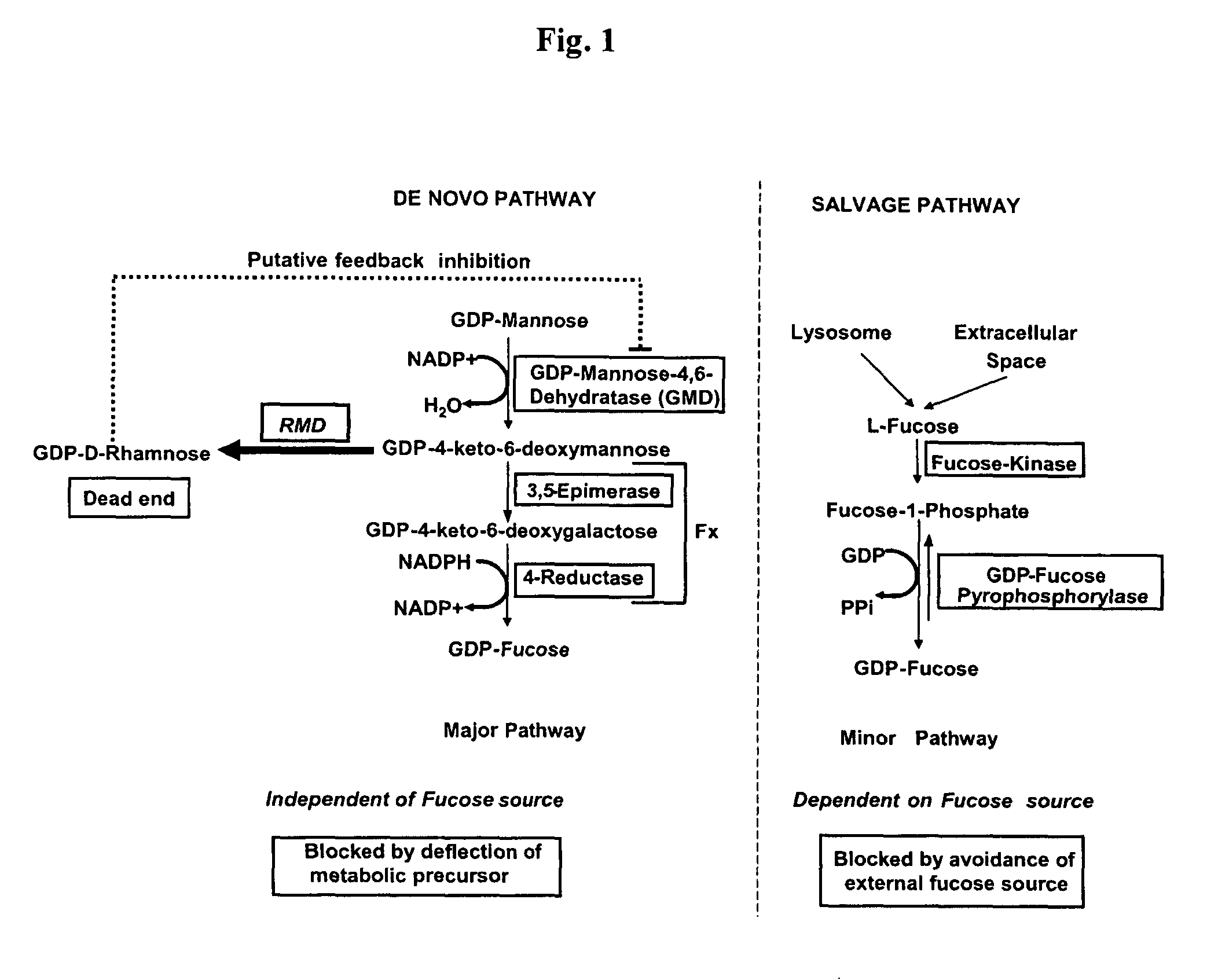
ActiveUS8642292B2Effectively block the fucose de novo pathwayReduce and eliminate allergic reactionBacteriaImmunoglobulinsGlycanSugar
The present invention relates to cells for producing a molecule lacking fucose, having a reduced amount of fucose, or having other atypical sugars on its glycomoieties. It also relates to methods for producing a molecule lacking fucose, having a reduced amount of fucose, or having other atypical sugars on its glycomoieties using said cells and to molecules obtainable by said methods. The present invention further relates to molecules having an artificial glycosylation pattern.
Owner:PROBIOGEN AG
Arrays with cleavable linkers
InactiveUS20070213278A1Treating and preventing HIV infectionAntibacterial agentsBiocideGlycanTest sample
Owner:THE SCRIPPS RES INST
Novel Imino Sugar Derivatives Demonstrate Potent Antiviral Activity and Reducted Toxicity
InactiveUS20110189771A1Good effectImprove performanceOrganic chemistryTissue cultureBovine Viral Diarrhea VirusesSide chain
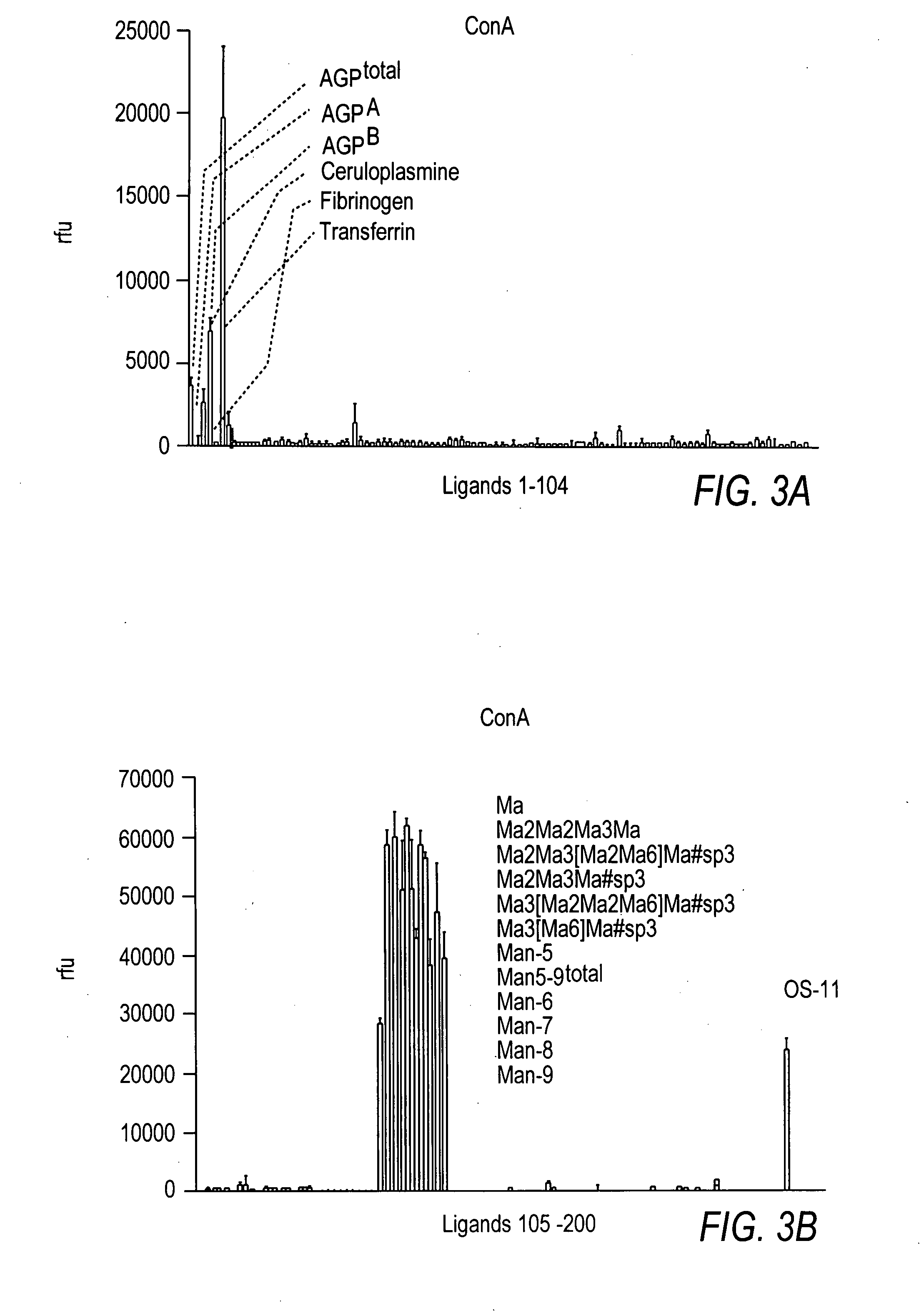
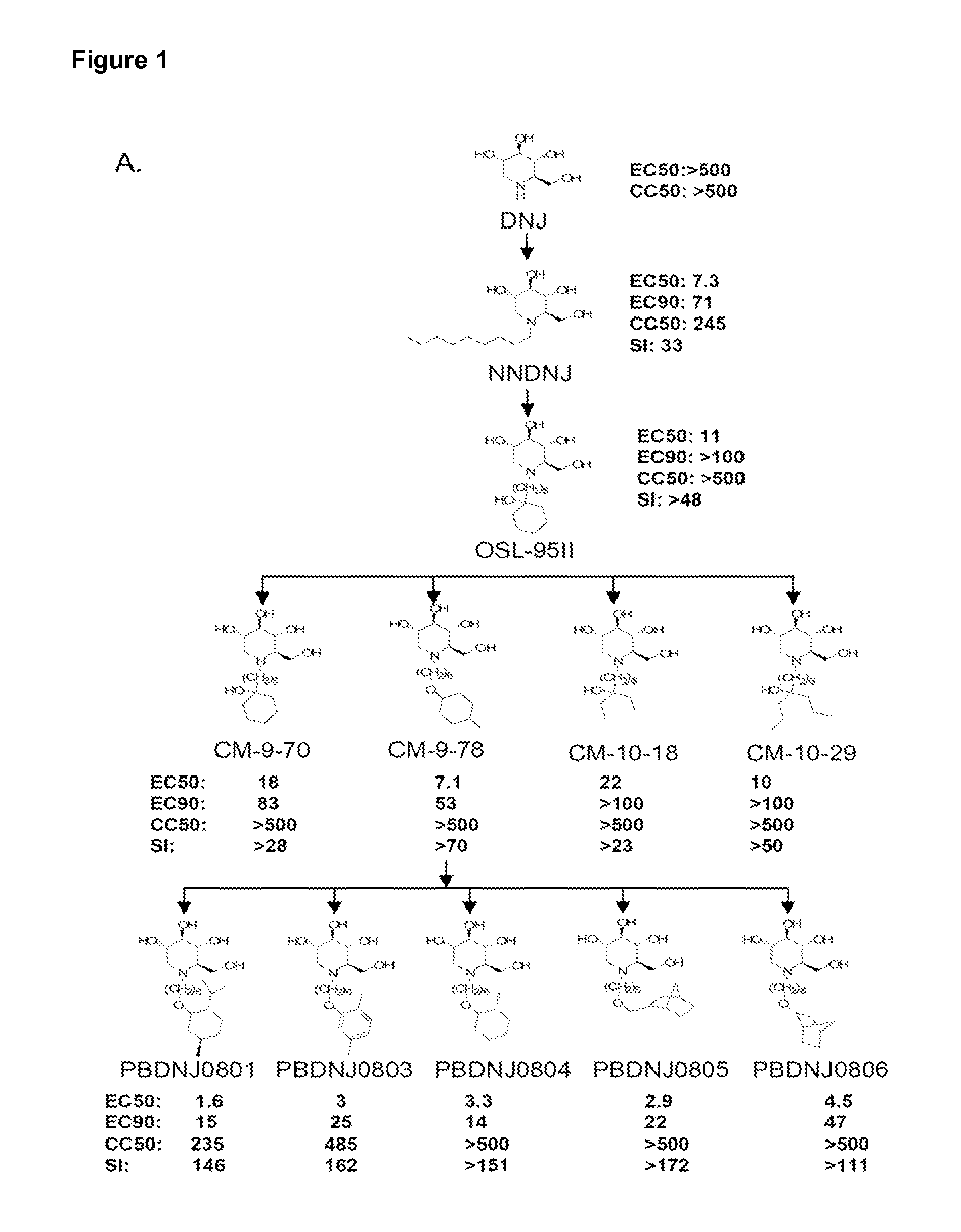
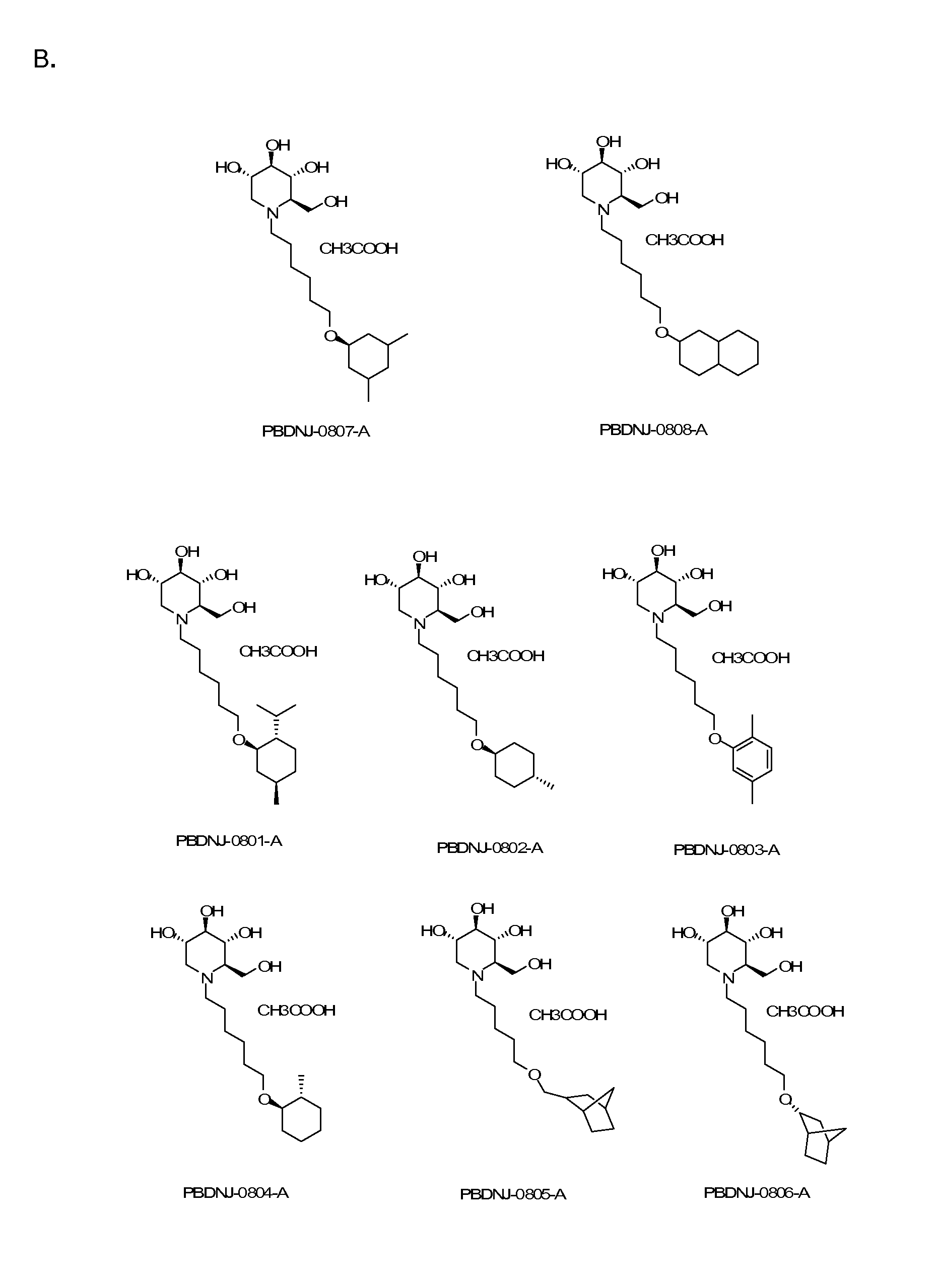
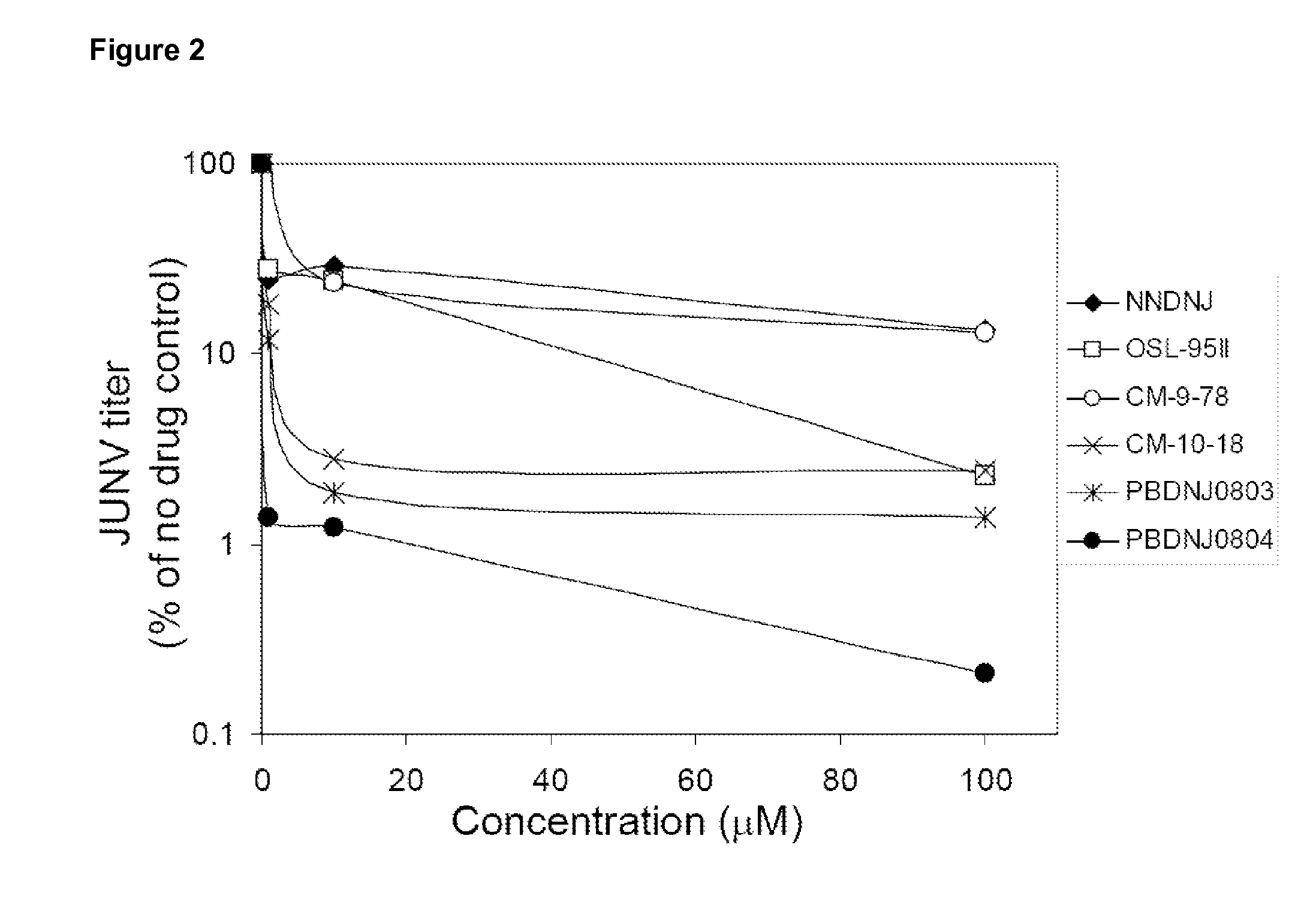
Imino sugars, such as deoxynojirimycin (DNJ), are glucose analogues that selectively inhibit cellular α-glucosidase I and II (enzymes that process N-linked glycans in glycoprotein) and exhibit broad spectrum antiviral activities against many enveloped viruses. Previously we have reported a novel DNJ derivative, OSL-95II, with antiviral activity and reduced cytotoxicity. In order to develop imino sugars with more potent antiviral activity as well as improved toxicity profile, OSL-95II was modified by diversifying the nitrogen linked alkylated side chain. The antiviral activities were initially tested in bovine viral diarrhea virus (BVDV) infected MDBK cells, yielding several imino sugar derivatives with novel structure and superior antiviral activity and toxicity profile. Furthermore, these new compounds were shown to be active against Dengue virus (DV) and West Nile virus (WNV) infection in BHK cells where potent anti-DV activity having submicromolar EC50 values and SI of greater than 900. These compounds represent a new generation of iminio sugars and their analogues, having application in the clinical treatment of infection of DV and other members of flaviviridae.
Owner:INST FOR HEPATITS & VIRUS RES +1
Conjugate vaccines for non-proteinaceous antigens
InactiveUS20070231344A1Strong and rapid effectStrong and rapid immune responseBacterial antigen ingredientsLipid/lipoprotein ingredientsAntigenConjugate vaccine
The present invention is directed to pharmaceutical compositions that can be used to immunize subjects using, for example, lipid, glycan, or nucleic acid antigens. These antigens are conjugated to a glycosphingolipid.
Owner:THE BRIGHAM & WOMEN S HOSPITAL INC +1
Compositions and methods for humanization and optimization of n-glycans in plants
InactiveUS20080060092A1Enhanced effector functionHigh antibody activityAntipyreticAnalgesicsGrowth plantGlycan
Methods for altering the N-glycosylation pattern of proteins in higher plants are provided. The methods comprise introducing into the plant a recombinant construct that provides for the inhibition of expression of α1,3-fucosyltransferase (FucT) and β1,2-xylosyltransferase (XylT) in a plant. Use of these constructs to inhibit or suppress expression of both of these enzymes, and isoforms thereof, advantageously provides for the production of endogenous and heterologous proteins having a “humanized” N-glycosylation pattern without impacting plant growth and development. Stably transformed higher plants having this protein N-glycosylation pattern are provided. Glycoprotein compositions, including monoclonal antibody compositions, having substantially homogeneous glycosylation profiles, and which are substantially homogeneous for the G0 glycoform, are also provided.
Owner:BIOLEX INC +1
N-acetylglucosaminyltransferase III expression in lower eukaryotes
The present invention relates to eukaryotic host cells having modified oligosaccharides which may be modified further by heterologous expression of a set of glycosyltransferases, sugar transporters and mannosidases to become host-strains for the production of mammalian, e.g., human therapeutic glycoproteins. The process provides an engineered host cell which can be used to express and target any desirable gene(s) involved in glycosylation. Host cells with modified lipid-linked oligosaccharides are created or selected. N-glycans made in the engineered host cells exhibit GnTIII activity, which produce bisected N-glycan structures and may be modified further by heterologous expression of one or more enzymes, e.g., glycosyltransferases, sugar transporters and mannosidases, to yield human-like glycoproteins. For the production of therapeutic proteins, this method may be adapted to engineer cell lines in which any desired glycosylation structure may be obtained.
Owner:GLYCOFI
Hemagglutinin polypeptides, and reagents and methods relating thereto
InactiveUS20080241918A1Immunoglobulins against blood coagulation factorsPeptide/protein ingredientsHemagglutininGlycan
The present invention provides a system for analyzing interactions between glycans and interaction partners that bind to them. The present invention also provides HA polypeptides that bind to umbrella-topology glycans, and reagents and methods relating thereto.
Owner:MASSACHUSETTS INST OF TECH
Immunoprotective influenza antigen and its use in vaccination
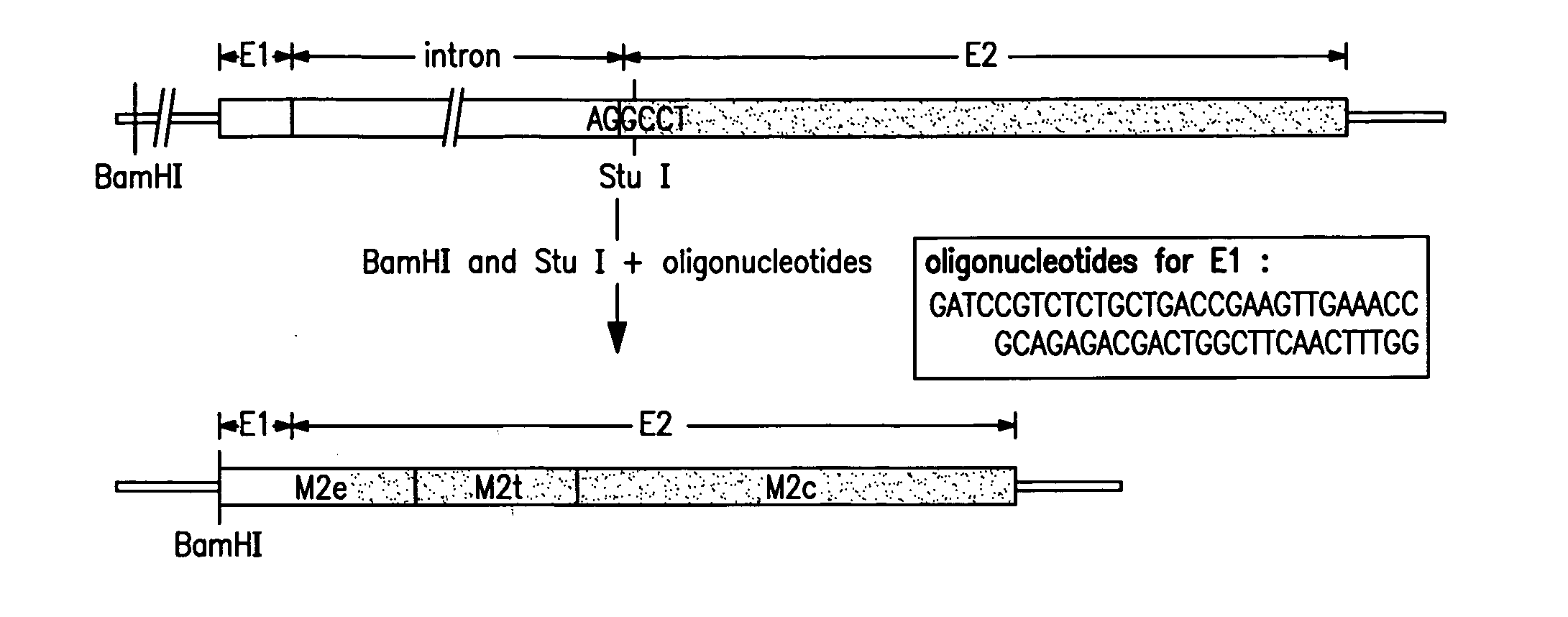
The present invention relates to an influenza antigen, comprising a fusion product of at least the extracellular part of a conserved influenza membrane protein or a functional fragment thereof and a presenting carrier, which may be a presenting (poly)peptide or a non-peptidic structure, such as glycans, peptide mimetics, synthetic polymers. The invention further relates to a vaccine against influenza, comprising at least an antigen of the invention, optionally in the presence of one or more excipients. The invention also relates to use of the antigen, a method for preparing the antigen and acceptor cells expressing the antigen.
Owner:VLAAMS INTERUNIVIR INST VOORS BIOTECH