Method for testing cyclic dipeptide in white spirits
A cyclic dipeptide and liquor technology, applied in the field of chemical analysis, can solve the problems of false positives, difficult separation and low content of qualitative unknowns, and achieve the effects of good stability, high accuracy and good repeatability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0101] a. Take 50mL of Luzhou-flavor liquor sample (52° Luzhou-flavor liquor), pass through the SPE C18 column, then elute with 50mL of methanol, and concentrate the eluate to 2mL at 40°C to obtain the sample to be tested;
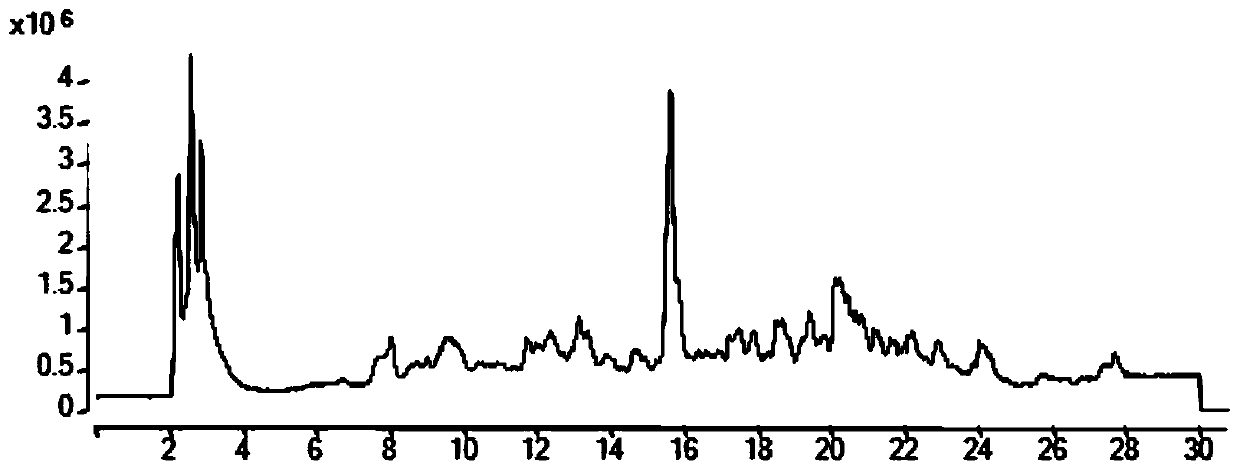
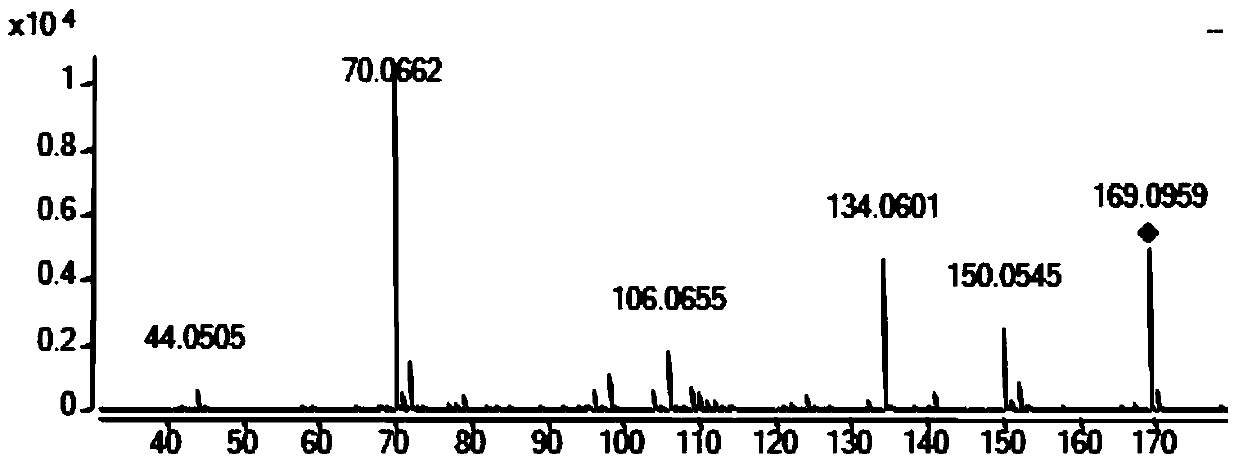
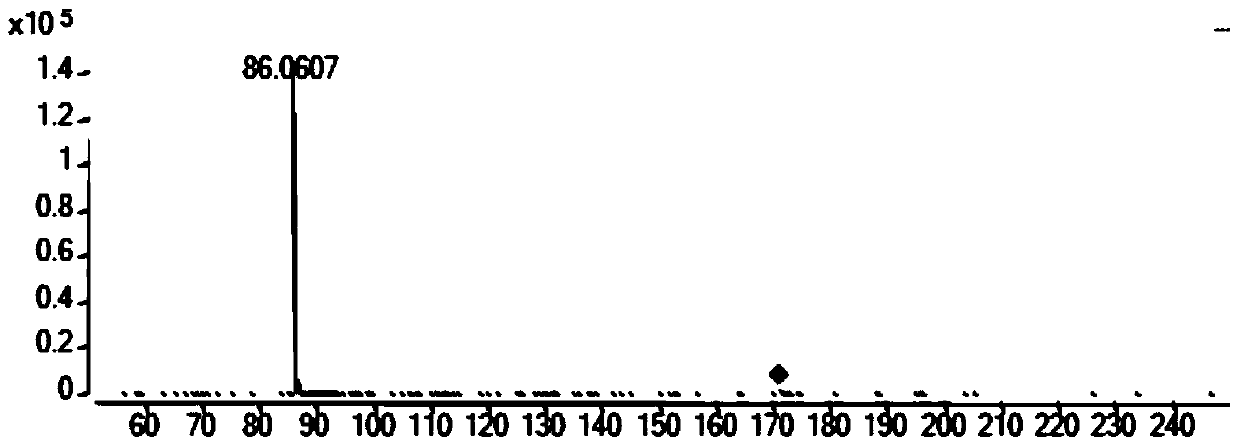
[0102] b. Load the sample to be tested in step a to a liquid phase-mass spectrometer (LC-QTOF), and after mass spectrometry full-ion scanning, obtain a full-ion scanning chart (see figure 1 ), through all ion scanning peaks, it is preliminarily judged that there are ring (alanine-proline), ring (diaminobutyric acid), ring (leucine-alanine), ring (isoleucine-valine acid), ring (threonine-proline), ring (proline-phenylalanine), ring (phenylalanine-alanine), ring (leucine-isoleucine), Cyclic (valine-valine), cyclic (tyrosine-proline), cyclic (phenylalanine-valine), cyclic (proline-leucine), cyclic (diphenylalanine amino acid) exists;
[0103] Among them, liquid chromatography conditions: chromatographic column: Agilent Eclipse Plus C18 2.1*100mm 1.8-micron;...
Embodiment 2
[0113] a. Take 100mL of Fen-flavor liquor sample (53° Fen-flavor liquor), pass through the SPE C18 column, then elute with 50mL of methanol, and concentrate the eluate to 2mL at 30°C to obtain the sample to be tested;
[0114] b. Load the sample to be tested in step a to a liquid phase-mass spectrometer (LC-QTOF), and after mass spectrometry full-ion scanning, obtain a full-ion scanning diagram (see Figure 15 ), through all-ion scanning peaks, it is preliminarily determined that there are rings (alanine-proline), rings (phenylalanine-alanine), rings (leucine-isoleucine), rings (tyramine acid-proline), ring (phenylalanine-valine), ring (proline-leucine), ring (diphenylalanine) exist;
[0115] Among them, liquid chromatography conditions: chromatographic column: Agilent Eclipse Plus C18 2.1*100mm 1.8-micron; column temperature: 25°C; injection volume: 10uL; mobile phase: A: 0.1% formic acid aqueous solution, B: methanol; flow rate: 0.1 mL / min; Gradient elution method: 0-18min:...
Embodiment 3
[0125] a. Take 20 mL of Maotai-flavored liquor sample (53° Maotai-flavored liquor), pass it through the SPE C18 column, then elute with 50 mL of methanol, and concentrate the eluate to 2 mL at 60°C to obtain the sample to be tested;
[0126] b. Load the sample to be tested in step a to a liquid phase-mass spectrometer (LC-QTOF), and after mass spectrometry full-ion scanning, obtain a full-ion scanning chart (see Figure 23 ), through all ion scanning peaks, it is preliminarily judged that there are ring (alanine-proline), ring (diaminobutyric acid), ring (leucine-alanine), ring (isoleucine-valine acid), ring (threonine-proline), ring (proline-phenylalanine), ring (phenylalanine-alanine), ring (leucine-isoleucine), Cyclic (valine-valine), cyclic (tyrosine-proline), cyclic (phenylalanine-valine), cyclic (proline-leucine), cyclic (diphenylalanine acid);
[0127] Among them, liquid chromatography conditions: chromatographic column: Agilent Eclipse Plus C18 2.1*100mm 1.8-micron; ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More