Hydroquinone synthesis method
A technology of hydroquinone and synthesis method, which is applied in chemical instruments and methods, preparation of organic compounds, organic chemistry, etc. It can solve problems such as high temperature, large amount of tar, harsh reaction conditions, etc., and achieve the effect of process cleaning and environmental protection
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0034] Example 1 (reaction part)
[0035] Hydroquinone preparation method is carried out as follows:
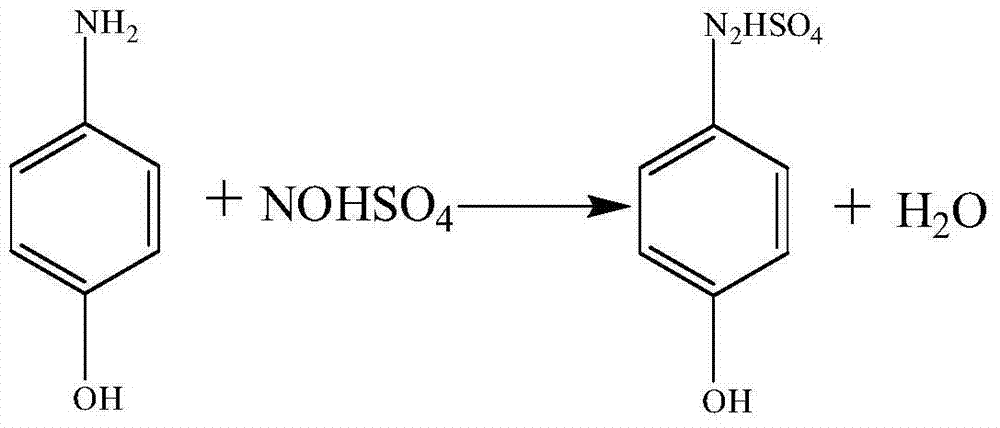
[0036] Diazotization reaction: Add 245g of 15% sulfuric acid and 27.8g of 98% p-aminophenol for industrial products into a 1000mL reactor, stir and cool down to -5~0℃, add 87.3g of 40% nitrosylsulfuric acid solution dropwise, and add the time 1-1.5hr, keep warm for 0.5hr after dripping, add 1.5g of urea, stir for 15min, when the release of nitrogen is not obvious, add 1.5g of thiourea and stir for another 15min. The dropwise addition, heat preservation and feeding temperature were all kept at -5-5°C to obtain 350-360g of diazonium solution, which was stored at 0°C.
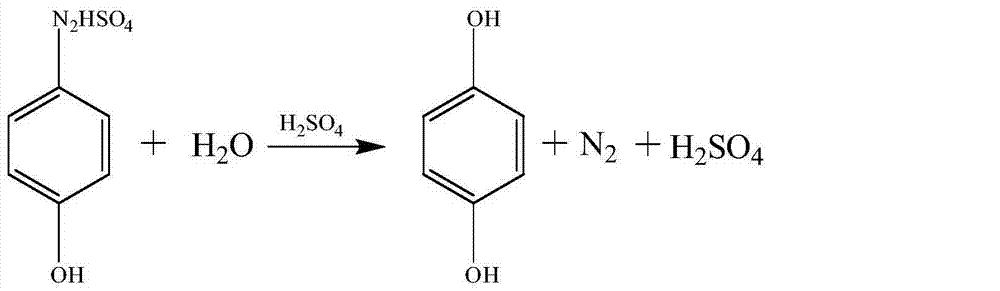
[0037] Hydrolysis reaction: first add 1000g of 40% sulfuric acid to a 2000mL reactor, heat to 106-110°C, add the above diazo solution dropwise at around 0°C, add dropwise for 1-1.5hr, and keep warm at 106-110°C for 3-4hr, Obtain about 1350g of hydrolyzate.
[0038] Extraction: The hydrolyzed liquid is lowered ...
Embodiment 2-6
[0040] The preparation method is the same as in Example 1, except that the concentration of sulfuric acid in the diazonium reaction is changed.
[0041]
Embodiment 7-12
[0043] The preparation method is the same as in Example 1, except that the temperature of the diazonium reaction is changed.
[0044]
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More