Synthetic polypeptide and application thereof, and anti-influenza-virus vaccine
An anti-influenza virus and synthetic peptide technology, applied in the field of immunology, can solve the problems of heavy workload, no exposure, and inability to guarantee 100% consistency, etc., and achieve the effect of simple design, low cost, and high conservatism
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0034] Example 1: Epitope and conservation detection
[0035] Sequence conservation was evaluated by sequence alignment analysis, and the sequence alignment database consisted of HA2 sequences of 10,763 strains of H3N2 subtype viruses from 1968 to 2014. The application software is ClustalWv1.4 and MEGA5.0, and the HA2 sequences of 1, 2, 3, 5, 7, and B subtype viruses are combined for conservative evaluation. The comparison results show that the synthetic polypeptide of the present invention has a certain degree of conservation with HA2 of subtypes 1, 2, 3, 5 and 7, and the conservation is as high as 97%.
[0036] At the same time, through the comprehensive analysis of vaccine accessibility, flexibility, and hydrophilicity, B / T cell epitopes were detected on the IEDB website, and the results showed that the synthetic polypeptide of the present invention also has B / T cell epitopes.
Embodiment 2
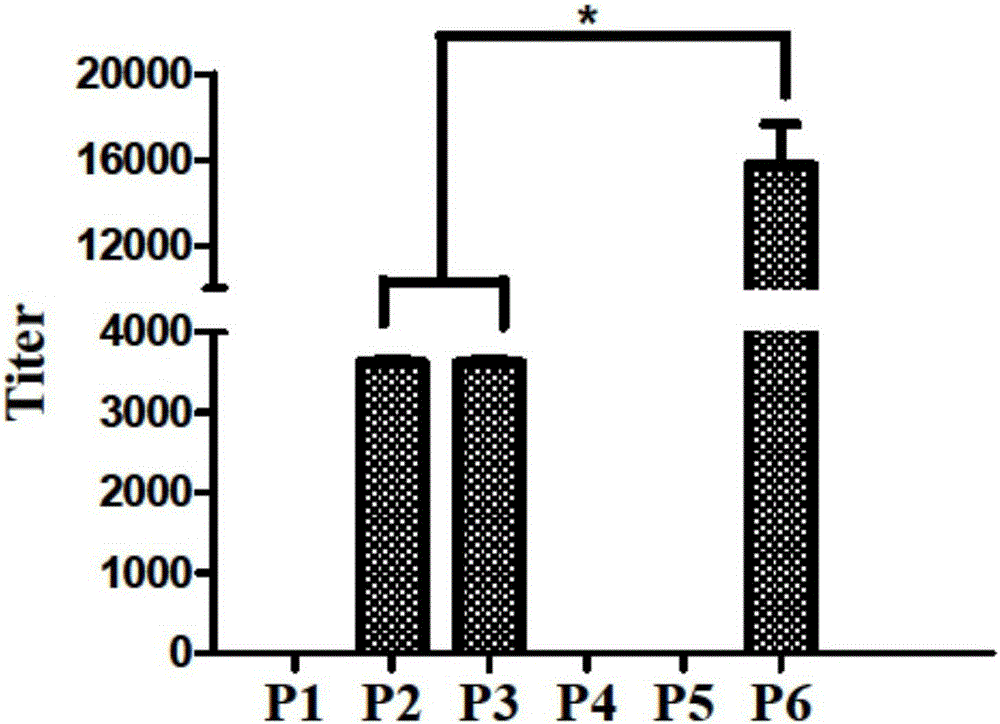
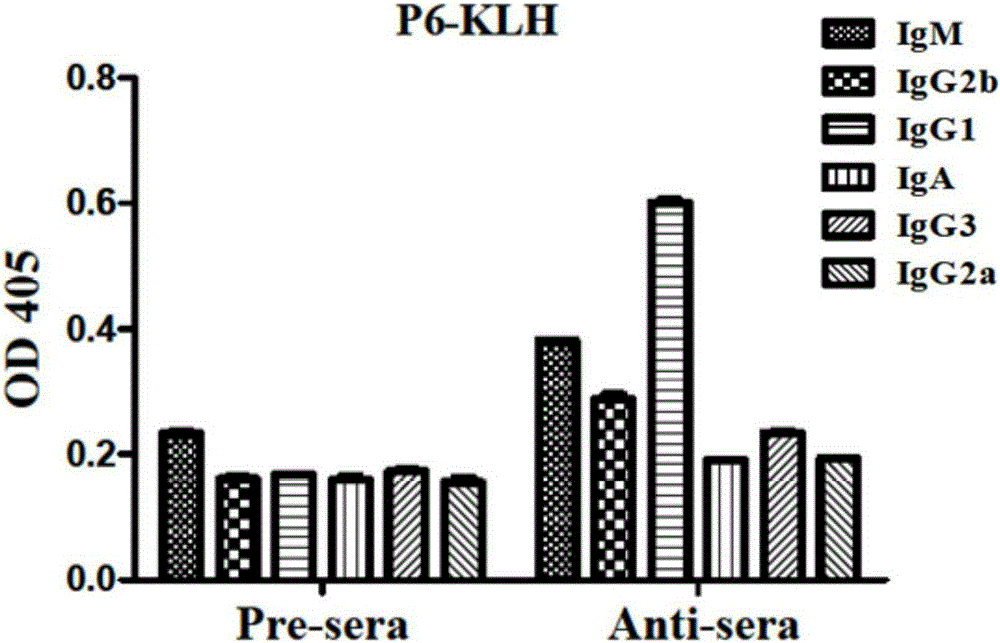
[0037] Example 2: Binding activity and antibody typing of synthetic polypeptide post-immunization serum to influenza virus strain HA protein
[0038] Animal immunization: BALB / c female mice aged 4-6 weeks were immunized, the immunization period was two weeks, and the immunogen was P1-P6 coupled KLH carrier. The immunization dose was 20ug / rat, and the immune adjuvant was Freund's adjuvant (Beijing Dingguo) mixed in a volume of 1:1, and blood was collected before each immunization. Four immunizations were performed.
[0039] A / Brisbane / 10 / 2007 (H3) HA protein was mixed with coating solution (final concentration 5-10 μg / mL) and coated in a 96-well plate, overnight at 4°C, blocked with 1% BSA / PBS at 37°C for 1 hour. Washed with PBST for 3 times, the serum after the first immunization was used as the primary antibody, diluted in PBST, the first dilution was 1:200, 5-fold serial dilution, and incubated at 37°C for 1.5h. Wash 3 times with PBST, add goat anti-mouse typing antibody t...
Embodiment 3
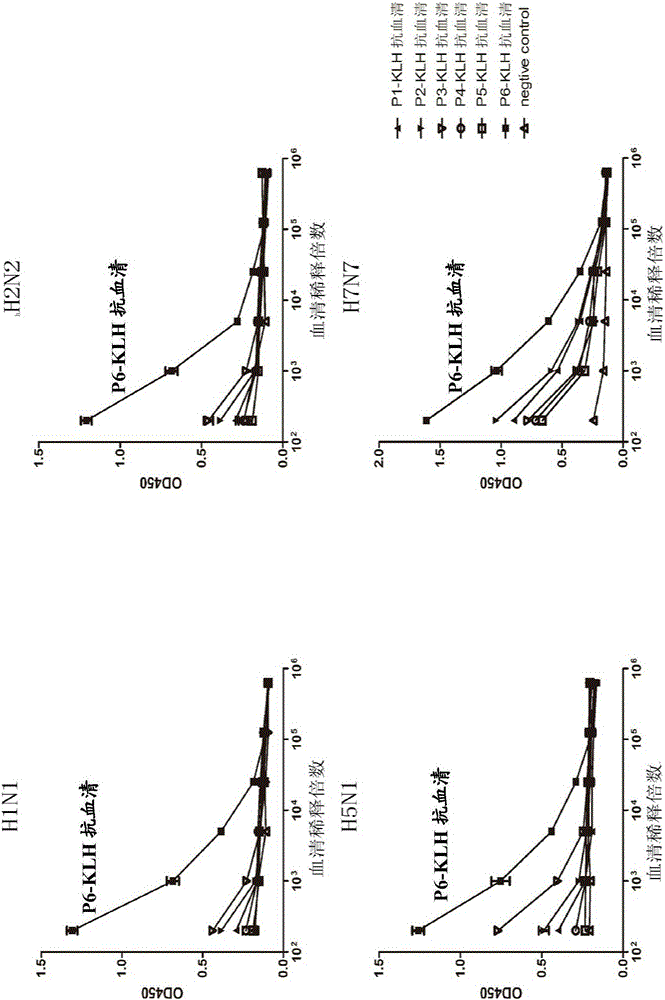
[0041] Example 3: Binding activity of synthetic polypeptide post-immunization serum to HA protein of different subtype influenza virus strains
[0042] Animal immunization: BALB / c female mice aged 4-6 weeks were immunized, the immunization period was two weeks, and the immunogen was P1-P6 coupled KLH carrier. The immunization dose was 20ug / rat, and the immune adjuvant was Freund's adjuvant (Beijing Dingguo) mixed in a volume of 1:1, and blood was collected before each immunization. Four immunizations were performed.
[0043] A / Brisbane / 10 / 2007(H3) HA protein, A / California / 04 / 2009(H1N1) HA protein, A / Canada / 720 / 2005(H2N2) HA protein, A / Anhui / 1 / 2005(H5N1) ) HA protein, A / Netherlands / 219 / 03 (H7N7) HA protein mixed with coating solution (final concentration 5-10μg / mL) coated in 96-well plate, overnight at 4°C, 1% BSA / PBS at 37°C Closed for 1h. Washed with PBST for 3 times, the serum after the first immunization was used as the primary antibody, diluted in PBST, the first diluti...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com