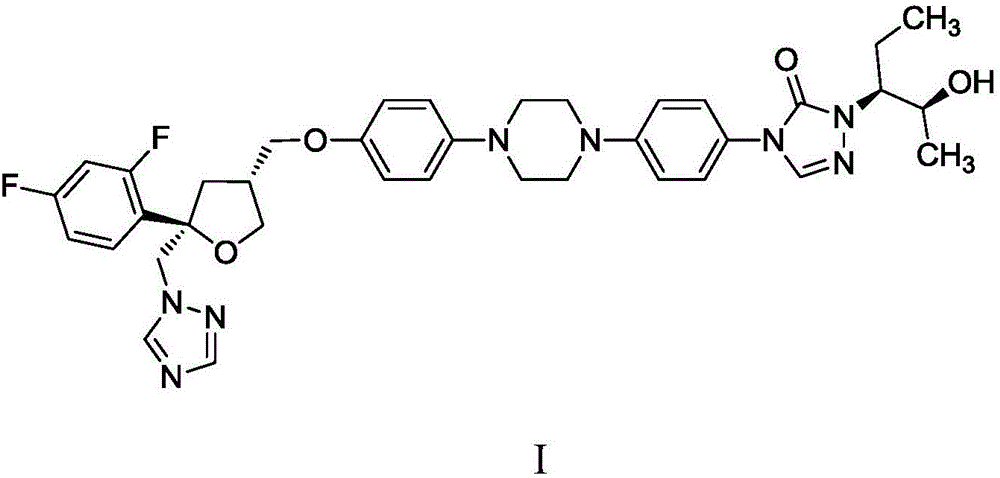
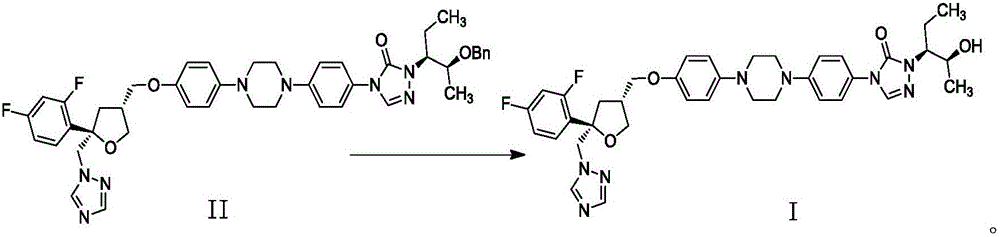
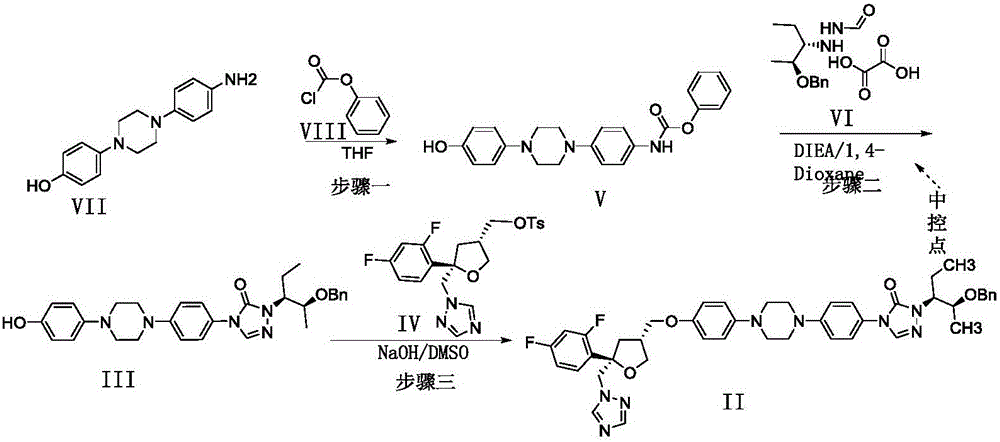
Preparation method of posaconazole
A technology for posaconazole and a purification method, which is applied in the field of preparation of posaconazole, can solve the problems of many by-products, complicated operation, low total yield and the like, and achieves few reaction by-products, simple and safe operation, and no heavy metals. residual effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0028] Example 1: Preparation of phenyl(4-(4-(4-hydroxy)-1-piperazinyl)phenyl)carbamate (V)
[0029]
[0030] Add 4.00 kg of 1-(4-aminophenyl)-4-(4-hydroxyphenyl)piperazine (VII) and 35.57 kg of tetrahydrofuran into a 100L glass reactor, turn on the stirring, and lower the temperature to 0-10°C. Keep the temperature below 25°C and add 2.56kg of phenyl chloroformate (VIII) dropwise. After 0.5 to 1 hour, the dropping is completed, and the temperature is controlled at 20 to 25°C to continue the reaction for 0.5 to 1.0 hour. The reaction was monitored by TLC. After the reaction was completed, the temperature was lowered to 0-10°C, a saturated aqueous sodium bicarbonate solution (2.00 kg of sodium bicarbonate dissolved in 38 kg of purified water) was added, and 20 kg of purified water was added, followed by stirring for 10 to 20 minutes. Centrifugal filter until almost no solvent flows out, then rinse with 8kg purified water, and centrifuge to filter until almost no solvent flows out....
Embodiment 2
[0032] Example 2: 2-((2S,3S)-2-(benzyl)-3-pentyl)-4-(4-(4-(4-hydroxy)-1-piperazine)phenyl)-2 , 4-Dihydro-3-hydro-1,2,4-triazol-3-one (III) preparation
[0033]
[0034] Add 4.36kg of N'-((2S,3S)-2-benzyloxy)pentyl-3-carboxhydrazine oxalate (VI) and 45.36kg of dioxane into a 100L glass reactor, turn on the stirring, Add 3.45 kg of N,N-diisopropylethylamine. After stirring for 1 to 1.5 hours, 5.47 kg of phenyl(4-(4-(4-hydroxy)-1-piperazinyl)phenyl)carbamate (V) is added. After the addition is completed, the temperature is raised to 80±5°C and reacted for 24-30 hours. TLC monitoring. After the reaction is completed, the system is cooled to 15-25°C, 57.79kg of dichloromethane and 21.81kg of pure water are added, stirred for 10-20 minutes, allowed to stand for separation, collect the lower organic phase, and discard the aqueous phase. The organic phase was transferred to a 100L glass reactor, 21.81 kg of purified water was added to the reactor, stirred for 10-20 minutes, allowed t...
Embodiment 3
[0035] Example 3: 4-(4-(4-(4-(((3R,5R)-5-((1-hydrogen-1,2,4-l-1-triazolyl)methyl)-5 -(2,4-Difluorophenyl)-3-tetrahydrofuranyl)methoxy)phenyl)-1-piperazinyl)phenyl)-2-((2S,3S)-2-(benzyl) -3-pentyl)-2,4-dihydro-3-hydro-1,2,4-triazol-3-one (II) preparation
[0036]
[0037] 35.01kg of dimethyl sulfoxide, with a mass concentration of 50% sodium hydroxide aqueous solution (1.24kg of sodium hydroxide dissolved in 1.24kg of pure water, the mass concentration means that the mass of sodium hydroxide accounts for the total mass of sodium hydroxide aqueous solution %), add to a 50L glass reactor, stir to clear, then add 5.31kg compound (III), stir for 20-30 minutes, add 4.60Kg (3S, 5R)-toluene-4-sulfonic acid 5-(2, 4-Difluorophenyl)-5-(1H-1,2,4-triazol-1-yl)methyltetrahydrofuran-3-ylmethyl ester (IV). After the addition is completed, the temperature is controlled at 25±5°C to react for 8-12 hours. TLC monitors the reaction, after the reaction is complete. Control the temperature to 20-3...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More