Immunological detection methods and kits for Mycoplasma pneumoniae
A technology for Mycoplasma pneumoniae and immunoassay, which is applied in immunoassay, immunoglobulin, chemical instruments and methods, etc., can solve the problems of unclear specificity and reactivity, and has not yet been determined, and achieves the effect of high sensitivity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0080] (Example 1: Expression and purification of recombinant P30 protein)
[0081] The amino acid sequence of P30 protein of Mycoplasma pneumoniae M129 strain was obtained from DDBJ (Database of National Institute of Genetics). From the amino acid sequence of the aforementioned P30 protein, the amino acid sequence (AA74-274) shown in SEQ ID NO: 2, which is the extracellular region except the membrane-penetrating domain, was specified, and the corresponding gene sequence was synthesized. The His-tag expression carrier, pET302 / NT-His, was cleaved with the restriction endonuclease EcoRI, treated with alkaline phosphatase as a dephosphorylation treatment, and mixed with the aforementioned gene sequence, using DAN Ligation Kit Ver .2 (Takara Bio) for the ligation reaction. The recombinant P30 plasmid incorporating the gene of interest was introduced into E. coli BL(DE3)pLysS (manufactured by Novagen), a host for recombinant protein expression. The introduced bacteria were cultur...
Embodiment 2
[0082] (Example 2: Preparation of monoclonal antibody against recombinant P30 protein)
[0083] The recombinant P30 protein obtained in Example 1 was used as an antigen for immunization to prepare a monoclonal antibody against the recombinant P30 protein (hereinafter referred to as anti-P30 antibody). Production of monoclonal antibodies is carried out according to conventional methods. Mice (BALB / c, 5-week-old, Japanese SLC) were immunized three times by mixing 100 μg of recombinant P30 protein with an equal amount of Aduvant Complete Freund (manufactured by Difco), and the spleen cells were used for cell fusion. Sp2 / 0-Ag14 cells (Shulman et al., 1978), which are mouse myeloma cells, were used for cell fusion. The following culture solution was used for the culture of the cells: 0.3 mg / ml of L-glutamine, 100 units / ml of potassium penicillin G, 100 μg / ml of streptomycin sulfate, gander Gentacin (Gentacin) 40 µg / ml (hereinafter referred to as DMEM) was added with fetal bovine ...
Embodiment 3
[0084] (Example 3: Preparation of monoclonal antibody)
[0085] The clonally propagated cells were inoculated intraperitoneally into mice (BALB / c, retired, Japan SLC) previously inoculated with pristane (2,6,10,14-Tetramethylpentadecane (manufactured by Sigma)) , to collect ascites. This ascitic fluid was supplied to a protein G column, and the monoclonal antibody was purified. The isotype of the prepared monoclonal antibody was identified using Mouse Monoloconal Antibody Isotyping Reagents (manufactured by Sigma).
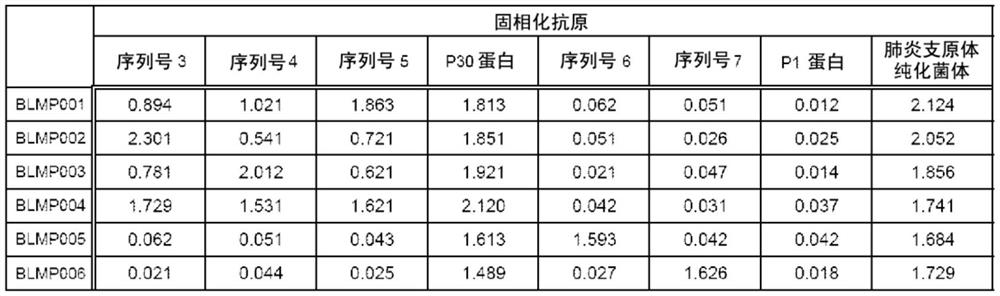
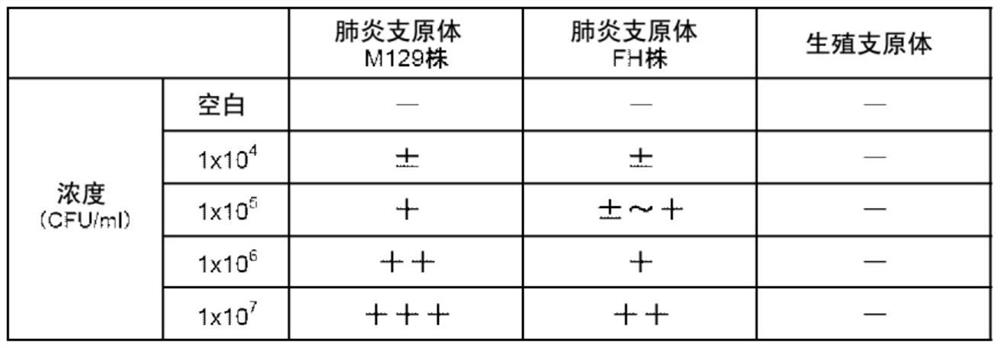
[0086] Finally, 6 clones of monoclonal antibody-producing cells against the P30 protein were obtained.
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More