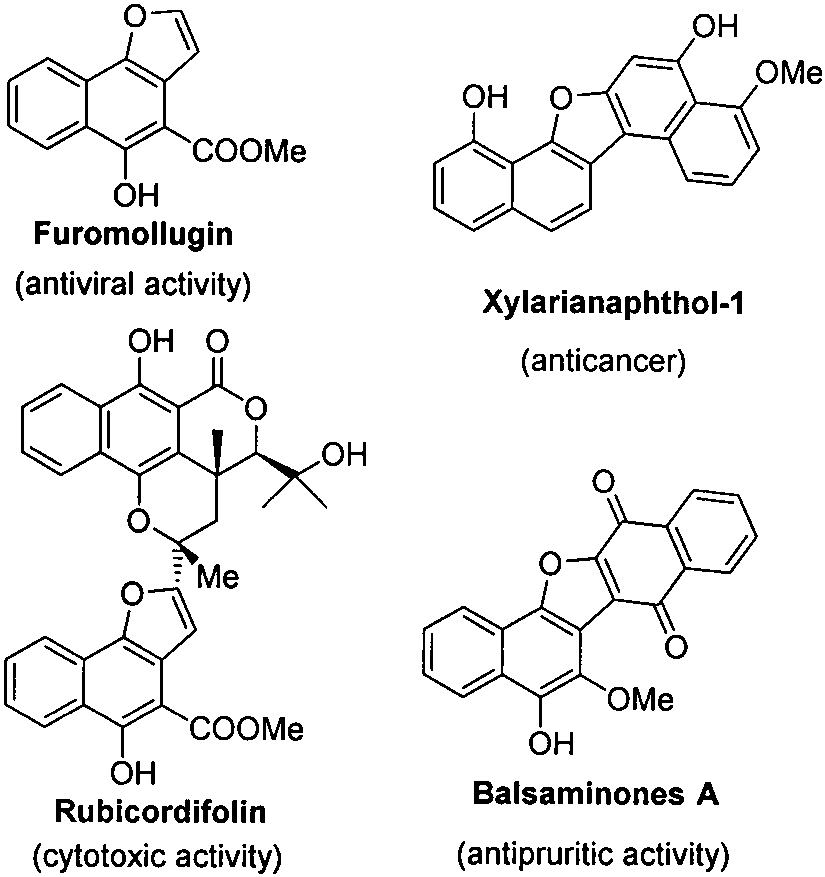
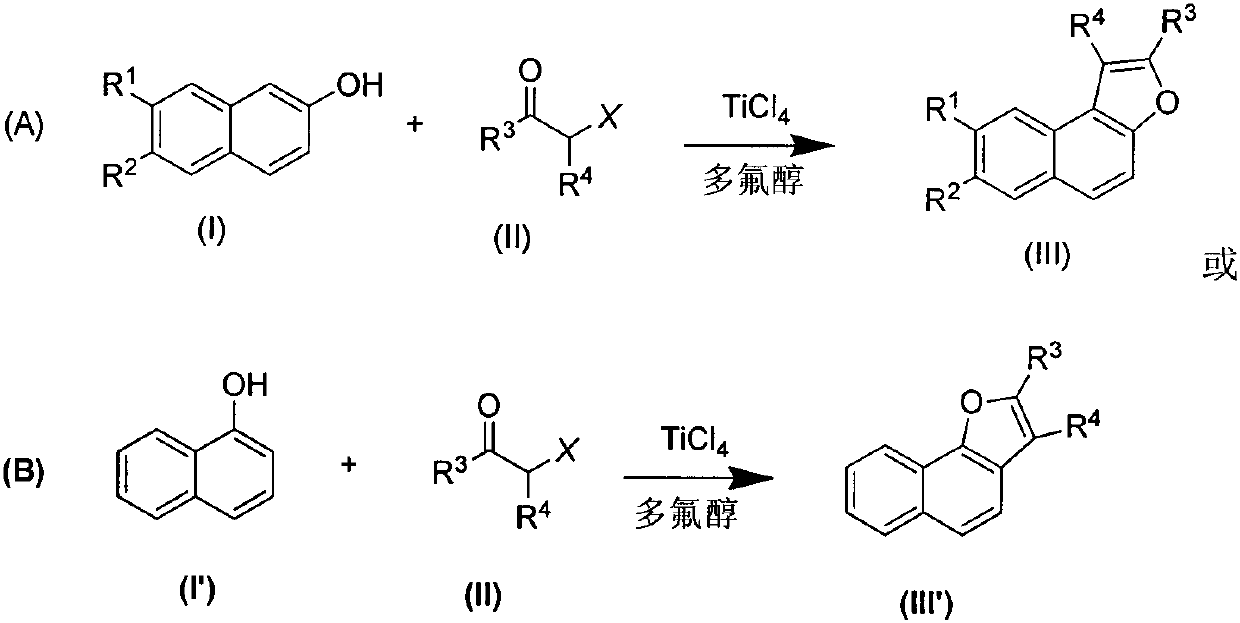
Preparation method of naphthofuran derivative
A technology for furan derivatives and naphthol is applied in the field of preparation of naphthofuran derivatives and achieves the effects of good regioselectivity, low cost and few side reactions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0024] Embodiment 1: the synthesis of naphthofuran derivative IIIa
[0025]
[0026] In a 50 mL two-necked flask equipped with a reflux condenser and under nitrogen protection, add β-naphthol Ia (2 mmol) and hexafluoroisopropanol (10 mL), and heat to reflux while stirring. After the β-naphthol Ia was completely dissolved, titanium tetrachloride (2mmol) was added, and finally a mixed solution of α-haloketone IIa (2mmol) and hexafluoroisopropanol (2mL) was slowly added dropwise, and the reflux reaction was continued. The reaction process was monitored by TLC. After the reaction was completed, it was quenched by adding saturated aqueous ammonium chloride solution (10 mL), and then extracted with dichloromethane (3×10 mL). The dichloromethane solution obtained by the mixed extraction was concentrated under reduced pressure, and then separated by silica gel column chromatography to obtain the target product IIIa as a yellow oil with a yield of 76%.
[0027] 1 H NMR (600MHz, C...
Embodiment 2
[0028] Embodiment 2: the synthesis of naphthofuran derivative III'a
[0029]
[0030] In a 50mL two-necked flask equipped with a reflux condenser and under nitrogen protection, α-naphthol I'a (2mmol) and trifluoroethanol (15mL) were added, and heated to reflux while stirring. After α-naphthol was completely dissolved, titanium tetrachloride (3mmol) was added, and finally a mixed solution of α-haloketone IIa (3mmol) and trifluoroethanol (2mL) was slowly added dropwise, and the reflux reaction was continued. TLC monitoring. After the reaction was completed, it was quenched by adding saturated aqueous ammonium chloride solution (10 mL), and then extracted with dichloromethane (3×10 mL). The dichloromethane solution obtained by the mixed extraction was concentrated under reduced pressure, and then separated by silica gel column chromatography to obtain the target product III'a as a yellow oil with a yield of 78%.
[0031] 1 H NMR (600MHz, CDCl 3 )δ8.27(d, J=8.2Hz, 1H), 7.91...
Embodiment 3
[0032] Embodiment 3: the synthesis of naphthofuran derivative IIIb
[0033]
[0034] β-Naphthol Ib (2 mmol) and trifluoroethanol (10 mL) were added to a 50 mL two-neck flask under nitrogen protection with a reflux condenser installed, and heated to reflux while stirring. After the β-naphthol Ib was completely dissolved, titanium tetrachloride (4mmol) was added, and finally a mixed solution of α-haloketone IIa (3mmol) and trifluoroethanol (2mL) was slowly added dropwise, and the reflux reaction was continued. Monitored by TLC. After the reaction was completed, it was quenched by adding saturated aqueous ammonium chloride solution (10 mL), and then extracted with dichloromethane (3×10 mL). The dichloromethane solution obtained by the mixed extraction was concentrated under reduced pressure, and then separated by silica gel column chromatography to obtain the target product IIIb as a yellow oil with a yield of 89%.
[0035] 1 H NMR (600MHz, CDCl 3 )δ8.06(d, J=1.8Hz, 1H), 7...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More