Patents
Literature
2491 results about "Titanium tetrachloride" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Titanium tetrachloride is the inorganic compound with the formula TiCl₄. It is an important intermediate in the production of titanium metal and the pigment titanium dioxide. TiCl₄ is a volatile liquid. Upon contact with humid air, it forms spectacular opaque clouds of titanium dioxide (TiO₂) and hydrated hydrogen chloride. It is sometimes referred to as "tickle" or "tickle 4" due to the phonetic resemblance of its molecular formula (TiCl₄) to the word.
Process for making durable rutile titanium dioxide pigment by vapor phase deposition of surface treatment
The present invention relates to a process for making durable titanium dioxide pigment by vapor phase deposition of surface treatments on the titanium dioxide particle surface by reacting titanium tetrachloride vapor, an oxygen containing gas and aluminum chloride in a plug flow reactor to form a product stream containing titanium dioxide particles; and introducing silicon tetrachloride into the reactor at a point down stream of the point where the titanium tetrachloride and oxygen were contacted and where at least 97% of the titanium tetrachloride has been converted to titanium dioxide or where the reaction temperature is no greater than about 1200° C., and preferably not more than about 1100° C.
Owner:THE CHEMOURS CO FC LLC
Titanium dioxide nanopowder manufacturing process
Owner:EI DU PONT DE NEMOURS & CO
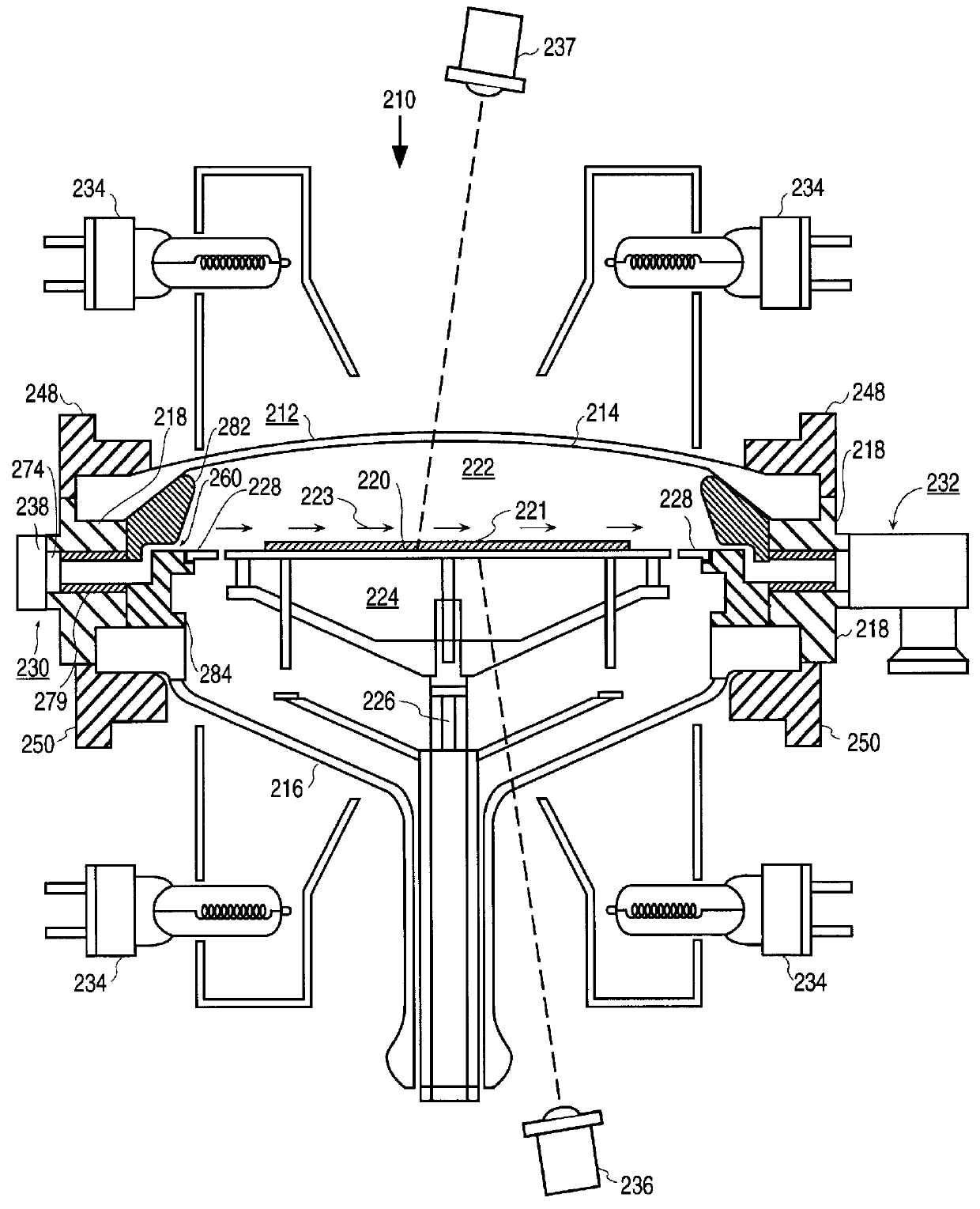
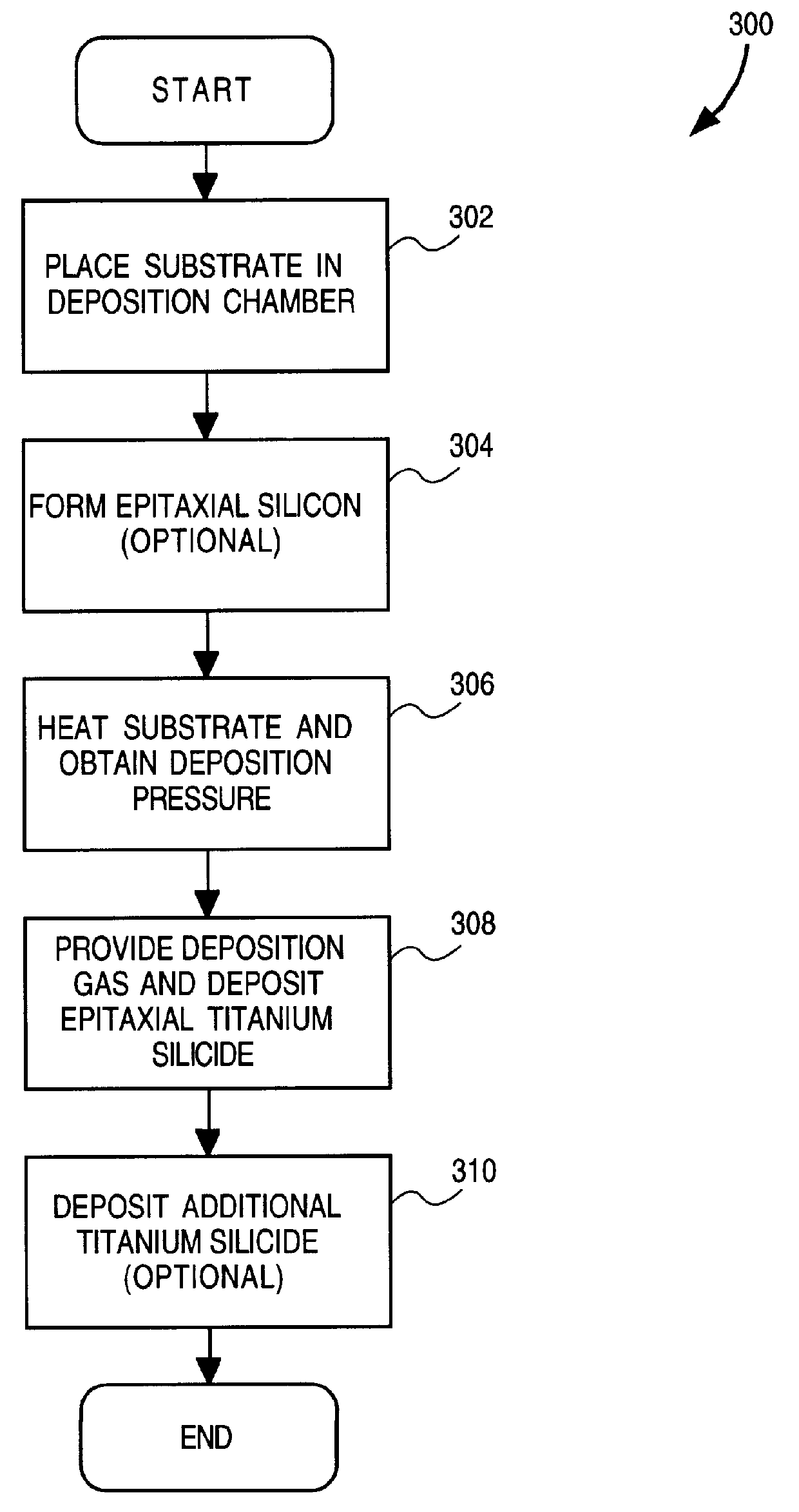
Method and apparatus for forming an epitaxial titanium silicide film by low pressure chemical vapor deposition
InactiveUS6019839APolycrystalline material growthSemiconductor/solid-state device manufacturingChemical vapor depositionTitanium tetrachloride
A method and apparatus for forming an epitaxial titanium silicide film is described. According to the present invention, a monocrystalline silicon substrate is placed in a deposition chamber and heated to a temperature between 710-770 DEG C. A silicon source gas and titanium tetrachloride are then provided into the deposition chamber. The deposition pressure is maintained between 5-10 torr. An epitaxial titanium silicide film is then formed on the substrate from the silicon source gas and the titanium tetrachloride.
Owner:APPLIED MATERIALS INC
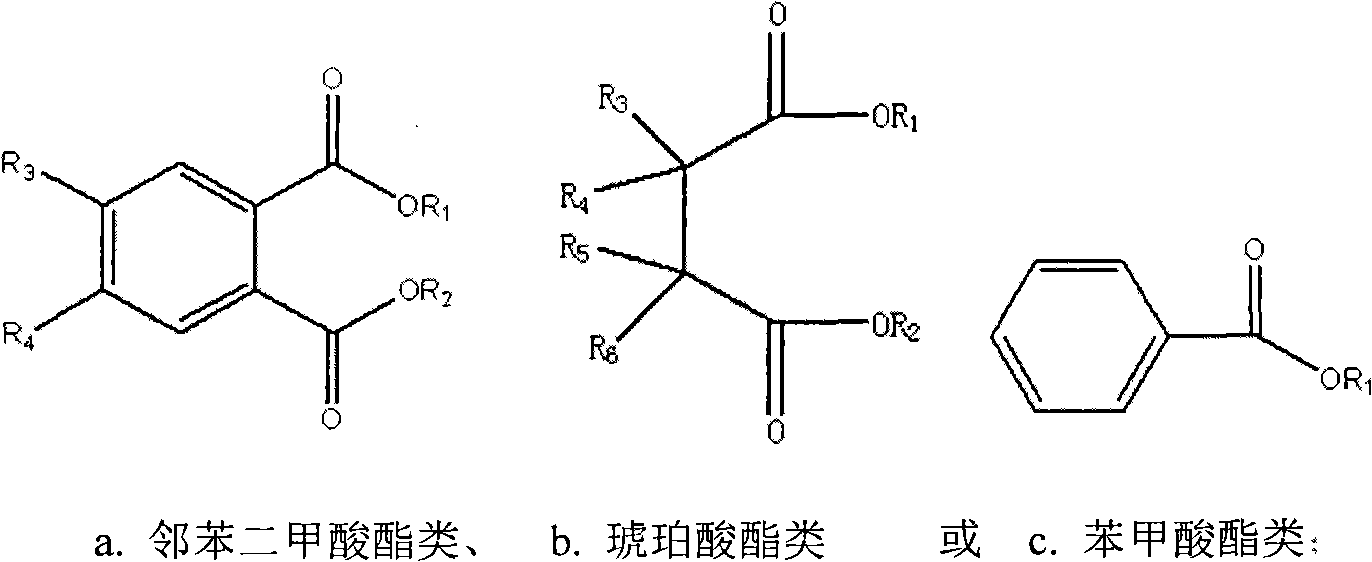
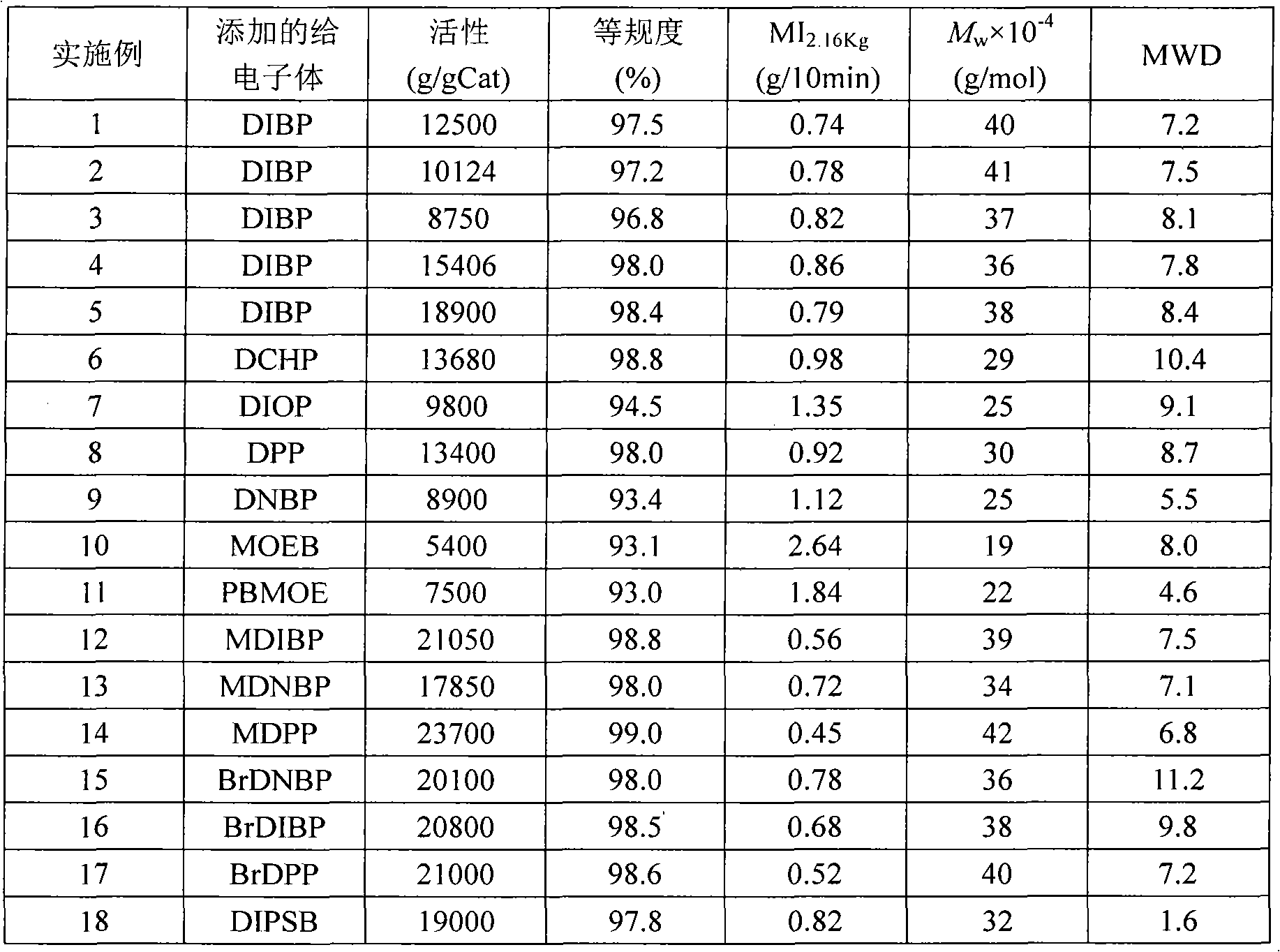
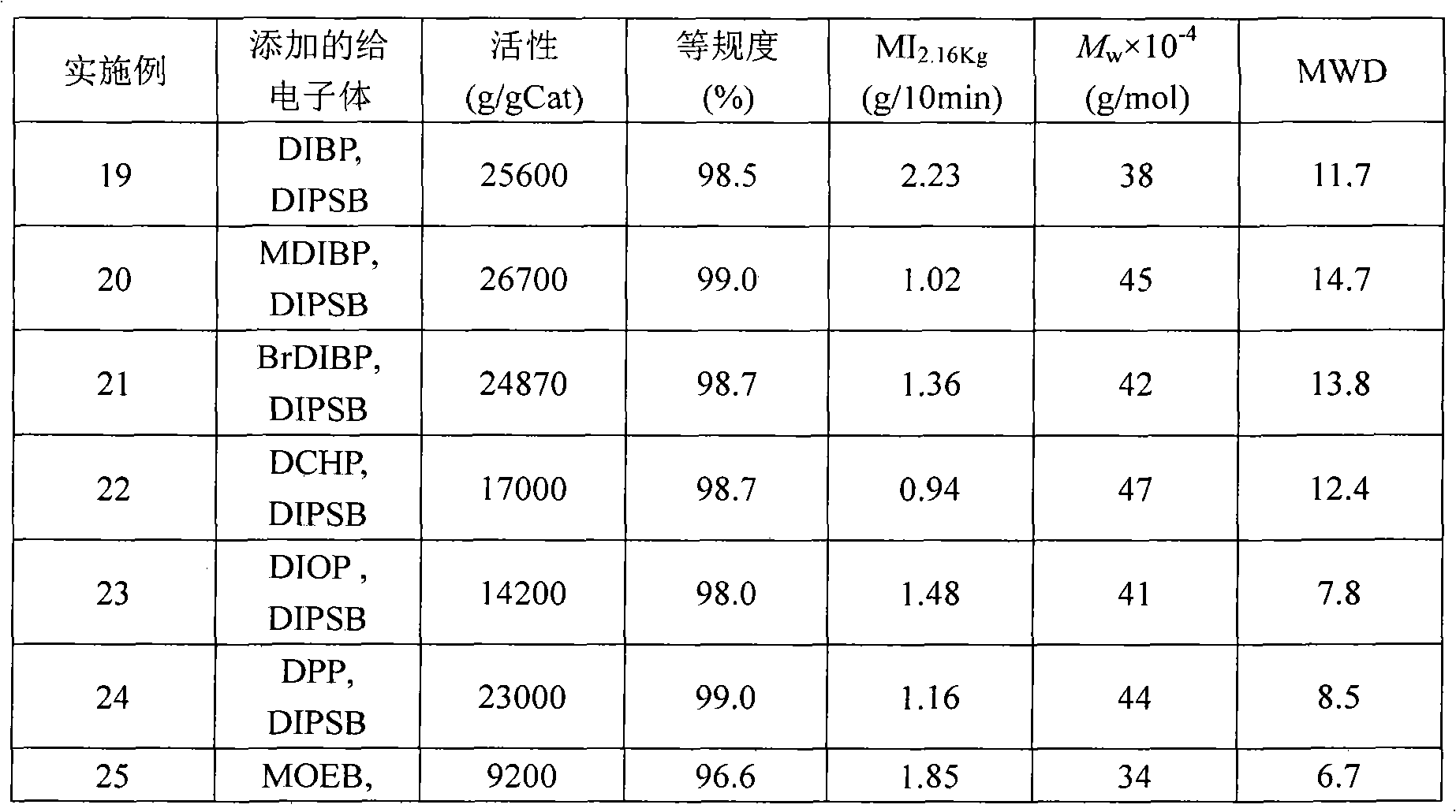
Solid catalyst component for alpha-olefin polymerization, catalyst for alpha-olefin polymerization, and process for producing alpha-olefin polymer
InactiveUS6187883B1High catalytic activityNarrow particle size distributionOrganic-compounds/hydrides/coordination-complexes catalystsCatalyst activation/preparationOrganic acidEther
The present invention relates to a solid catalyst component for alpha-olefin polymerization having a narrow particle size distribution of not less than 6.0 in terms of the value of N in a Rosin-Rammler function of particle size distribution and giving a catalytic activity of not less than 10,000 ((g-polymer produced / g-solid catalyst component) / hour) in polymerization. The catalyst component is obtained by a process which comprises reducing a titanium compound with an organomagnesium compound in the presence of an organosilicon compound and an ester to obtain a solid product, then treating the solid product with: (a) a mixture of titanium tetrachloride and an ether, (b) an organic acid halide, and (c) a mixture of titanium tetrachloride and an ether optionally containing an ester.
Owner:SUMITOMO CHEM CO LTD
Electrothermal film and manufacturing method thereof
ActiveCN101668359ASimple processEfficient processHeating element materialsResistors adapted for applying terminalsTitanium chlorideMetallurgy
The invention relates to an electrothermal film and a manufacturing method thereof, belonging to the technical field of semiconductor heating. The electrothermal film is mainly prepared by adopting stannic chloride, titanium tetrachloride, stannic chloride, titanium trichloride, ferric chloride, antimony trichloride, calcium chloride, potassium chloride, cadmium chloride, stannic dioxide, stannictetroxide, hydrofluoric acid, boric acid, ethanol, isopropyl alcohol and inorganic water. By adopting the above formula, the mixture is mixed, stirred and heated to prepare into electrothermal film treating fluid, a semi-finished product of the electrothermal film is obtained by spraying the electrothermal film treating fluid at negative pressure on the electrothermal film carrier, and then silveroxide slurry is coated on the semi-finished product of the electrothermal film for baking to form a finished product of the electrothermal film. The electrothermal film has reasonable proportion andsimple manufacturing process, can be manufactured into various electrothermal film heating devices, has a working temperature capable of being up to 500 DEG C, and has wider application range. The electrothermal film of the invention also has the function of far infrared radiation, can play a role of physical therapy and health care to human body, and can help improve the quality and output of agricultural products.
Owner:GUANGDONG HALLSMART INTELLIGENCE TECH CORP LTD
Method for quickly synthesizing small-crystallite titanium-silicon molecular sieve in cheap system
ActiveCN101913620ALower synthesis costFast crystallization rateCrystalline aluminosilicate zeolitesCyclohexanoneHigh activity
The invention provides a method for preparing a small-crystallite titanium-silicon molecular sieve in a cheap system by changing the adding mode of seed crystals and by a hydrothermal process. The titanium-silicon molecular sieve is prepared by using silicon sol as a silicon source, titanium tetrachloride or tetrabutyl titanate as a titanium source, tetrapropylammonium bromide ( TPABr) as a template agent, organic amine as an alkali source and unseparated nanoscale TS-1 mother liquor as seed crystals directly and by the hydrothermal process. The grain size of the titanium-silicon molecular sieve is less than 1 micrometer, and the titanium-silicon molecular sieve has high activity for selective oxidization reactions using hydrogen peroxide as an oxidant, such as epoxidation of olefins, hydroxylation of phenol, ammoxidation of cyclohexanone and the like. The method simplifies the synthesis process of small crystallite TS-1 in the cheap system and reduces synthesis time and cost.
Owner:DALIAN UNIV OF TECH
Solid catalyst component and catalyst for olefins polymerization
InactiveUS6770586B2Particles in good shapeImprove handlingOrganic chemistryOrganic-compounds/hydrides/coordination-complexes catalystsPropylene PolymersOlefin polymerization
Owner:TOHO TITANIUM CO LTD
Photocatalytic composite material and method for preparation thereof
InactiveUS20050214533A1Highly active andHighly durableGas treatmentLiquid surface applicatorsFiberWater vapor
A photocatalytic composite material having a high activity and good durability is produced by coating the surface of a substrate with a continuous film of titanium oxide by vapor deposition from titanium tetrachloride. In the case of a substrate which is a mass of inorganic fibers such as glass cloth, the individual fibers or filaments in the mass are coated with titanium oxide. The vapor deposition is performed by contacting the substrate, such as a mass of inorganic fibers, which has been heated to 100-300° C., with a mixture of distilled pure titanium tetrachloride vapor and water vapor to form a film of a titanium oxide precursor on the surface of the substrate. Then, the substrate is heated at 300-600° C. in an oxidizing atmosphere, resulting in the formation on the substrate surface of a continuous film of a photocatalyst having a high activity and good adhesion to the substrate and comprising crystalline titanium oxide with an average crystallite diameter of 50 nm or smaller.
Owner:OSAKA TITANIUM TECHNOLOGIES
Method for producing fine particles of metal oxide
A method for producing fine particles of metal oxide characterized in that metal halide is hydrolyzed in the presence of organic solvent. According to this invention, under hydrolysis of titanium tetrachrolide, anatase type titanium oxide can be obtained by selecting hydrophilic organic solvent, and rutile type titanium oxide can be obtained by selecting hydrophobic organic solvent.
Owner:SHOWA DENKO KK
Method for reclaiming valuable element through gradient chlorination of vanadium slag
ActiveCN101845552ANo pollution in the processAvoid pollutionProcess efficiency improvementChemical industryTitanium tetrachloride
The invention relates to a method for reclaiming valuable elements through gradient chlorination of vanadium slag, which belongs to the technical field of chemical industry and is used for solving the problem of efficiently reclaiming multiple metal elements in the vanadium slag. The method is characterized by comprising the steps of proportioning raw materials, fractionating vanadium tetrachloride and vanadium oxytrichloride, fractionating ferric trichloride, fractionating titanium tetrachloride, chromium trichloride and silicon tetrachloride, collecting fractionation products, processing chlorination tailings and the like. By directly adopting the vanadium slag as the raw material in the method of the invention, the highly efficient and comprehensive utilization of vanadium-titanium magnetite resources is realized in one process; the valuable elements such as iron, vanadium, chromium, silicon, titanium and the like are enriched through gradient chlorination and selective separation, and the purifying process is relatively simple; closed circulation can be realized by using chlorine in the process and has no environmental pollution; and the whole flow is disengaged from an aqueous system, thereby water resources are saved, and meanwhile, the pollution of the traditional flow to the environment is avoided. The method of the invention has the advantages of short flow, high element yield, no pollution, high benefit and the like.
Owner:HEBEI IRON AND STEEL
Process for making durable rutile titanium dioxide pigment by vapor phase deposition of surface treatments
The present invention relates to a process for making durable titanium dioxide pigment by vapor phase deposition of surface treatments on the titanium dioxide particle surface by reacting titanium tetrachloride vapor, an oxygen containing gas and aluminum chloride in a plug flow reactor to form a product stream containing titanium dioxide particles; and introducing silicon tetrachloride into the reactor at a point down stream of the point where the titanium tetrachloride and oxygen were contacted and where at least 97% of the titanium tetrachloride has been converted to titanium dioxide or where the reaction temperature is no greater than about 1200° C., and preferably not more than about 1100° C.
Owner:THE CHEMOURS CO FC LLC
Process for preparing metallic titanium and titanium master alloy
The invention relates to the non-ferrous metal metallurgy fused salt electrolysis field, a method mainly comprises the steps of: preparing titanium dioxide, titanium tetrachloride, titanium dichloride and fluotitanate as raw materials, electrolyzing one or a plurality of combinations of TiO2, TiC14 and the fluotitanate in an electrolysis bath, preparing metallic titanium or titanium-based master alloy through an electrolysis method or a thermal reduction-electrolysis combined method, performing the TiO2 and connecting direct current to deoxidize or adopting metal (or metallic compound) for heat reducing the TiO2 beforehand, preparing the metallic titanium which contains oxygen (O) with certain concentration, and then electrolyzing aluminium, alkali metal, alkaline earth, rare earth metal, metallic copper, metallic zinc or metallic lead to deoxidize finally in the electrolysis bath. The purpose of the method is to reduce the production cost of the metallic titanium, simplify the production procedures and lower the environmental pollution in the production process, especially the titanium dioxide taken as the raw materials, the production flow is shortened, the storage and the transportation are convenient, none chlorine gas takes part in the reaction, and green metallurgy of the metallic titanium can be realized.
Owner:曹大力 +1
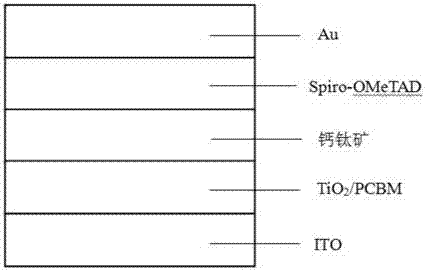
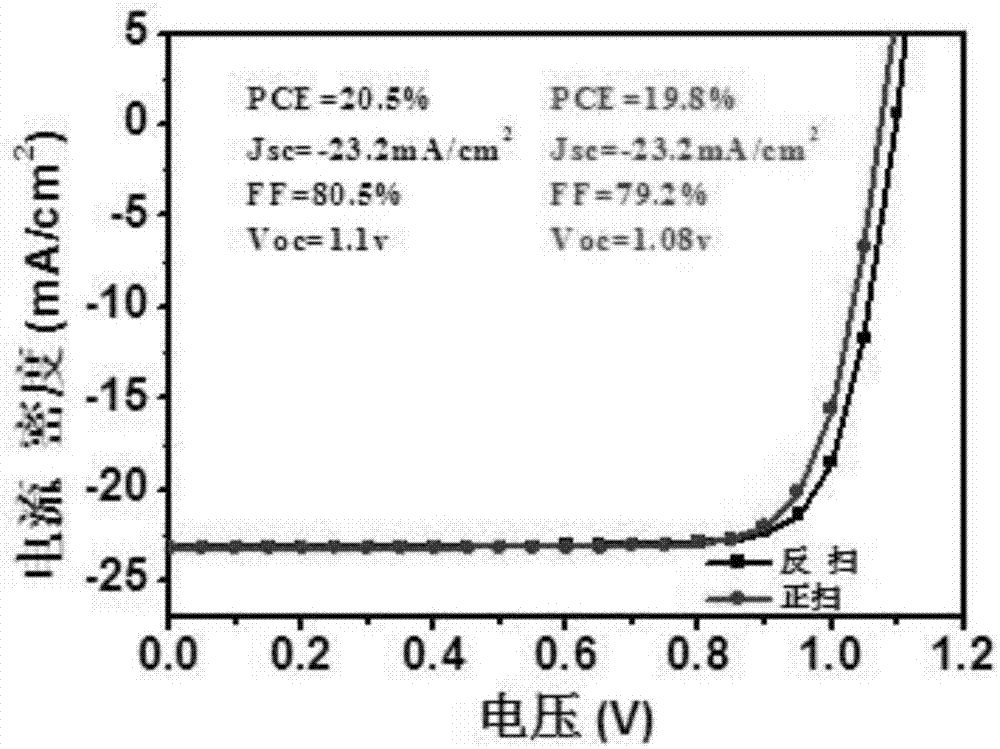
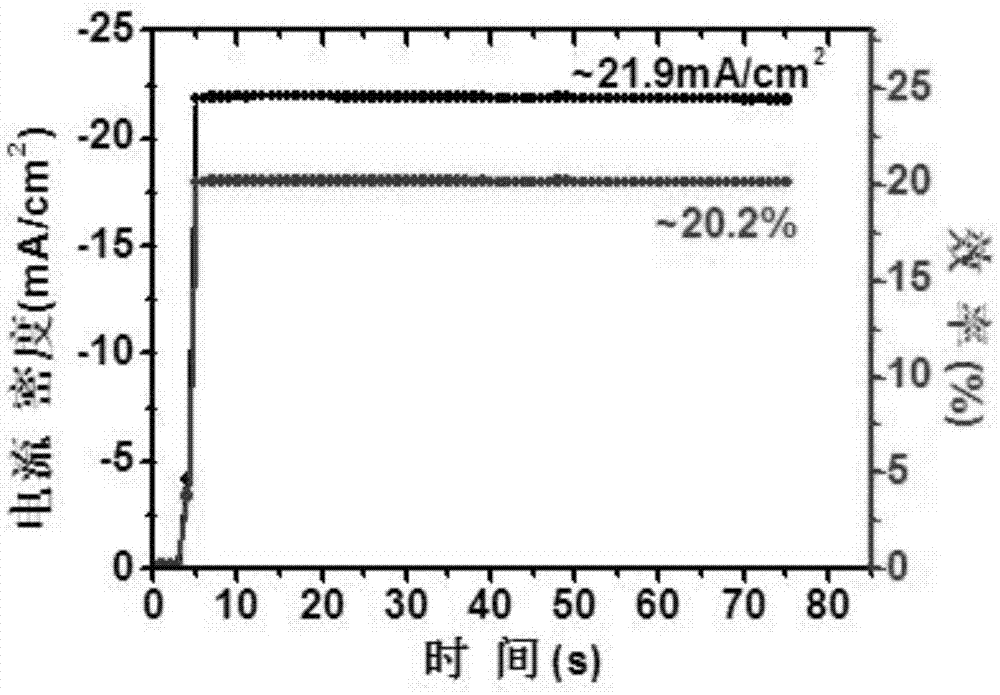
Perovskite solar cell and preparation method thereof
InactiveCN107316942AGood dispersionSpeed up extractionSolid-state devicesSemiconductor/solid-state device manufacturingHysteresisHole transport layer
The invention belongs to the field of a solar cell, and discloses a perovskite solar cell. The perovskite solar cell sequentially comprises a transparent conductive substrate, an electron transmission layer, an interface modification layer, a modified perovskite active layer, a hole transmission layer and a positive electrode, wherein the electron transmission layer is a nanometer TiO2 particle layer, and the interface modification layer is a fullerene derivative layer. A sol-gel method is employed, high-crystallization TiO2 nanoparticles are synthesized by taking titanium tetrachloride is used as a precursor, the TiO2 nanoparticles are applied to the perovskite solar cell by employing a low-temperature annealing process, a fullerene derivative is directly spin-coated on a surface of the obtained nanometer TiO2 particle layer for modification, the defects in TiO2 and perovskite are passivated, a novel perovskite synthesis path is employed, the obtained perovskite solar cell has high efficiency and does not have hysteresis effect under a low-temperature preparation process, and the device can be used for stable transmission; and moreover, the related preparation method is simple, is low in energy consumption and is suitable for promotion and application.
Owner:WUHAN UNIV OF TECH
Low temperature crystallized nano titanium dioxide powder and sol synthesizing method
InactiveCN1935669AImprove photocatalytic activityStable storageTitanium dioxideNanocrystalTitanic acid
The invention belongs to an organic metal oxide-TiO2 photocatalyst preparing process, especially relating a synthesizing method of low temperature-crystallizing nanometer TiO powder and sol with high catalytic activity. And it obtains n-titanic acid deposit by making alkali neutralization and dilution hydrolyzation or heating hydrolyzation on titanic solution, and implements synthesizing related system nano crystal materials with normal-pressure and low-temperature liquid phases by such a processing process of re-dispersing the deposit by oxydol. And it largely reduces the cost of raw materials and avoids using titanium tetrachloride with strong corrosivity, reducing equipment requirements.
Owner:TECHNICAL INST OF PHYSICS & CHEMISTRY - CHINESE ACAD OF SCI
Preparation method for titanium dioxide group catalyst carrier
InactiveCN101513620AGood formabilityImprove catalytic performanceCatalyst carriersDispersed particle separationTitanium tetrachlorideMetatitanic acid
The invention provides a preparation method for a titanium dioxide group catalyst carrier with excellent moldability, high strength and high specific surface. The method comprises the following steps: using titanium dioxide or metatitanic acid prepared by hydrolyzation and neutralization of precipitate with titanium tetrachloride as the raw material; by weight portions, mixing and grinding 60-85% of titanium dioxide or dried metatitanic acid powder (counted as titanium dioxide), 1-10% of temporary adhesive and 1-5% of pore-forming material into uniform powder; adding 5-35% of constitutive adhesive, 0.5-2% of molding adhesion promoter, 1-10% of extrusion aid and water followed by evenly mixing and making into mud; extruding for molding after the mud decays; seasoning the extrudate at room temperature; baking and dehydrating at a temperature below 80 DEG C; and then iglossing at 400-600 DEG C to obtain the titanium dioxide group carrier. After molding, the specific surface of the carrier is 80-150m / g, and the pressive strength thereof is 15-100N / mm. The carrier is mainly used for selective catalytic reduction (SCR) to remove nitrogen oxide.
Owner:NANJING UNIV
Process for preparing nm crystal TiO2 photocatalyst carried by metallic wire screen skeleton
InactiveCN1342518AHigh bonding strengthSmall air resistanceCatalyst activation/preparationActive layerPowder suspension
A metallic wire screen skeleton material-carried nm-clas cystol TiO2 photocatalyst is prepared through preparing transistion layer sol from n-butyl titanate as precursor, preparing active layer sol from n-butyl titanium or TiCl4 and pore-forming agent, coating the transition layer sol on the cleaned metallic wire screen skeleton-shaped carrier, directly coating the active layer sol, drying and calcining; or preparing nm TiO2 powder suspension, and attaching it the said carrier to form the photocatalyst. Its advantages include high adhesion, low gas resistance and high photocatalytic efficiency and activity.
Owner:TSINGHUA UNIV
Production of TiO2 naometer light catalyzing net from foam metal carrier
InactiveCN1712131AStable chemical propertiesImprove breathabilityPhysical/chemical process catalystsPhoto catalyticWater quality
A foam metal carried nano-TiO2 photocatalyst mesh with high chemical stability, air permeability, specific surface area and photo catalytic activity is prepared through dipping the foam metal substrate in the sol of SiO2, Al2O3 or aluminium silicate, drying, calcining to generate a transition layer, immersing in the sol of tetrabutyl titanate, TiCl4, or TiCl3, drying, and calcining.
Owner:SHANGHAI JIAO TONG UNIV
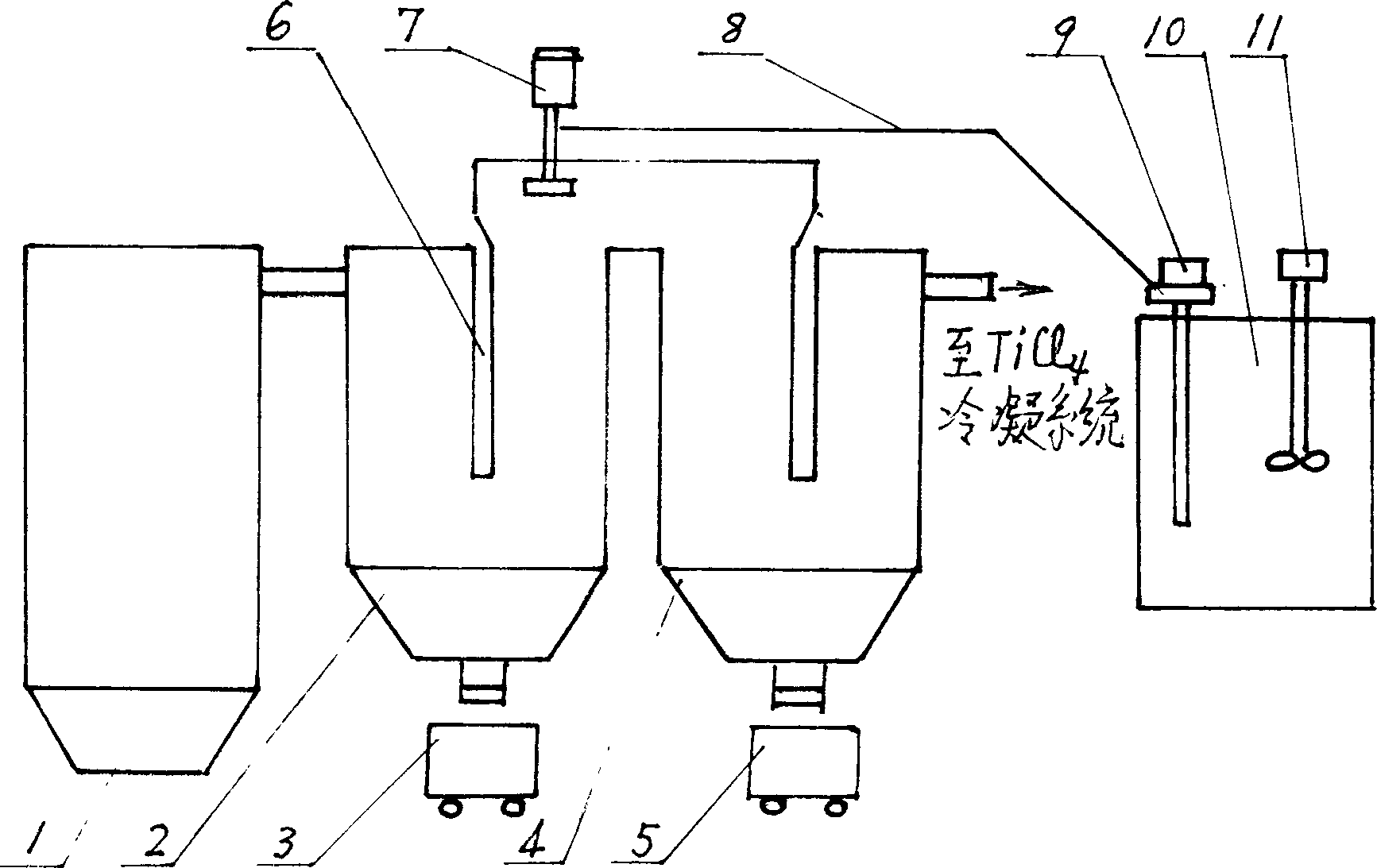
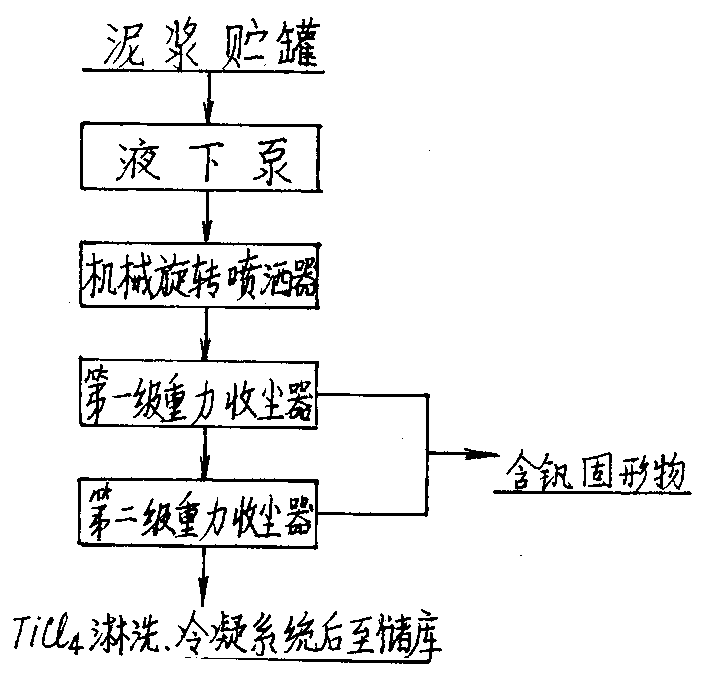
Technique and method for treating titanium tetrachloride slurry containing vanadium
InactiveCN1415547ASave moneySave energySludge treatmentTitanium halidesThermal energyCollection system
A process for treating the sludge containing CCl4 and V to recover TiCl4 includes such steps as pumping said sludge from sludge tank to the stage-1 gravitational dust collector in chlorinating system, countercurrent heat exchange between the sludge sprayed from top and the high-temp gas mixture from chlorinating furnace to gasifying the atomized TiCl4 drops, introducing the gas mixture to stage-2 dust collector, depositing the most of V-contained solid into ash collector while further separating, and condensing the purified TiCl4 gas for collecting it in original system. Its advantages are high recovery rate of TiCl4 and no pollution.
Owner:中信钛业股份有限公司
Production of titanium dioxide optical catalyst sol
InactiveCN1778685ANo secondary pollutionImprove photocatalytic effectPhysical/chemical process catalystsTitanium dioxideWater bathsCatalytic effect
Production of titania light catalyst colloidal sol includes adding at least one among titanic chloride, titanous sulfate and butyrin titanate into alcohol diluent solvent while agitating, dripping the mixed solution into hydrolysable solvent under water bath, agitating, and laying aside colloidal sol. It has super oxidative ability to decompose organics and harmful gas and can be used for glass, ceramic, household apparatus and public places. It has non toxic and no harm, no second pollution, better light catalytic effect, simple process and lasting performance.
Owner:ZHEJIANG UNIV
Preparation method for bisphenol F
ActiveCN102491879AEasy to separate and recycleThe synthesis process is simpleOrganic chemistryOrganic compound preparationAluminium chlorideSynthesis methods
The invention discloses a synthesis method for bisphenol F, in which aluminium chloride, titanium tetrachloride and modified cation exchange resin are adopted as catalysts, and phenyl hydroxide and formaldehyde are adopted as raw materials. The invention has the technical effects as follows: the aluminium chloride, the titanium tetrachloride and the modified cation exchange resin are adopted for catalysis to synthesize the bisphenol F, and the resin catalyst is easy to separate and recover and can be used repeatedly; 2, a recrystallization and reduced pressure distillation combined separation technology is adopted for coproduction of high-purity bisphenol F and common bisphenol F products, the maximum yield of the bisphenol F reaches 90 percent, the content of 4, 4'-bisphenol F in a dimethyl benzene recrystallization product reaches 93.5 percent, reduced pressure distillation after-products are mainly other two isomers, and the separation of the 4, 4'-bisphenol F is realized effectively; 3, the requirement for the concentration of the formaldehyde raw material is not strict while the traditional one stage process require that the concentration of formaldehyde is above 40 percent; and 4, the synthesis technology is simple, the operation is convenient, and the realization of industrial production is easy.
Owner:湖南嘉盛德材料科技股份有限公司
Preparing method for titanium dioxide fibre
The producing method of two-oxidation titanium fibre belongs to function fibre material technology scopes. Compose poly-acetamide acetone titanium to be the van-substance. Let chemic dosage of four-chloridize titanium, distilled water, acetamide acetone and three-ethylamine dilute in methanol respectively. Mix them and react when mixing round at 0-25 deg.C. Compose poly-acetyl acet, titanium. Remove hydrochloric three-ethylamine by using four-hydrogen furan. Dissolve poly-acetyl acet, titanium in methanol to make filature liquid. Obtain van-substance short fibre by using acentric swing method. Obtain van-substance long fibre by using dry filature method. And we can obtain two-oxidation titanium fibre by using high pressure or normal pressure vapor heat treatment technique agglomeration. The intension of extend of this invention is 100 MPa-1.2GPa. The diameter is 3um-20um. The one-thread series length is 1 cm-50 cm or higher than 1 m. The crystal diameter is 5 nm-100 nm. The appearance is two-oxide ation short fibre or consecutive fibre with titanium droff-titanium-mine posture or rutile posture or coexistence.
Owner:SHANDONG UNIV
Method of preparing titanium dioxide, stannum dioxide and doping composite fiber material thereof
InactiveCN101033082AGood lookingSimple structureNanostructure manufactureTitanium dioxideTin dioxideNitrogen gas
This invention relates to a method for preparing fiber TiO2, SnO2 and its doped compound material characterizing in utilizing a fiber material as a template, taking TBT, titanium tetrahalide, tin tetrachloride or a mixture as a precursor, which is carried by nitrogen or argon and enters into a reactor with a gas containing ammonia for chemical gaseous phase deposition, in which, the gas flow, deposition temperature and the sinter atmosphere and temperature are controlled, the inert gas is heated to get load-type fiber material to be sintered under the atmosphere of oxidation and reaction to remove the fiber template and form an oxide fiber material, and utilize the active carbon fiber template and ignition process to form porous fiber or tubular fiber material.
Owner:DALIAN UNIV OF TECH
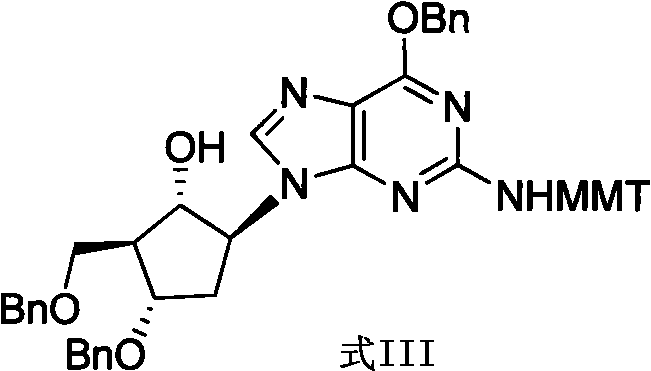
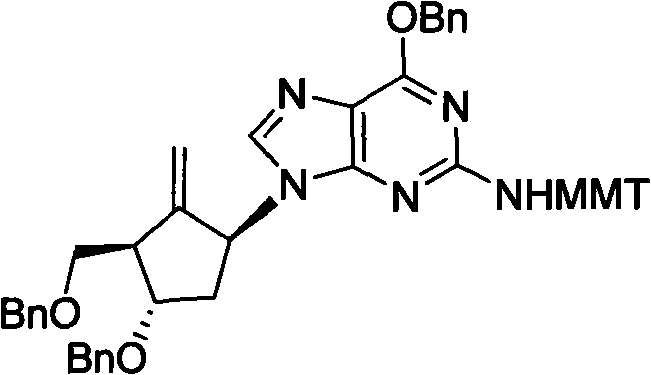
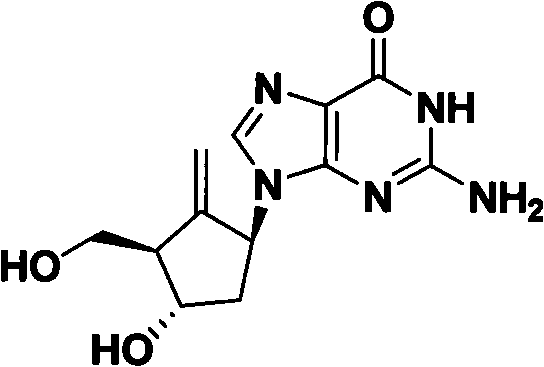
Method for preparing antiviral medicinal entecavir intermediate
The invention relates to a method for preparing an antiviral medicinal entecavir intermediate compound with a formula I. An oxidation system formed by TEMPO and iodine under an alkali condition is adopted to oxidize a compound with a formula III to obtain a compound with a formula II, and methylenation is carried out on the formula II by a methylenation system formed by titanium tetrachloride, magnesium, methylene chloride and tetrahydrofuran to obtain the compound with the formula I. The invention has easy acquirement of raw materials and simple and convenient method and is suitable for industrialized production.
Owner:SHANGHAI QINGSONG PHARMA
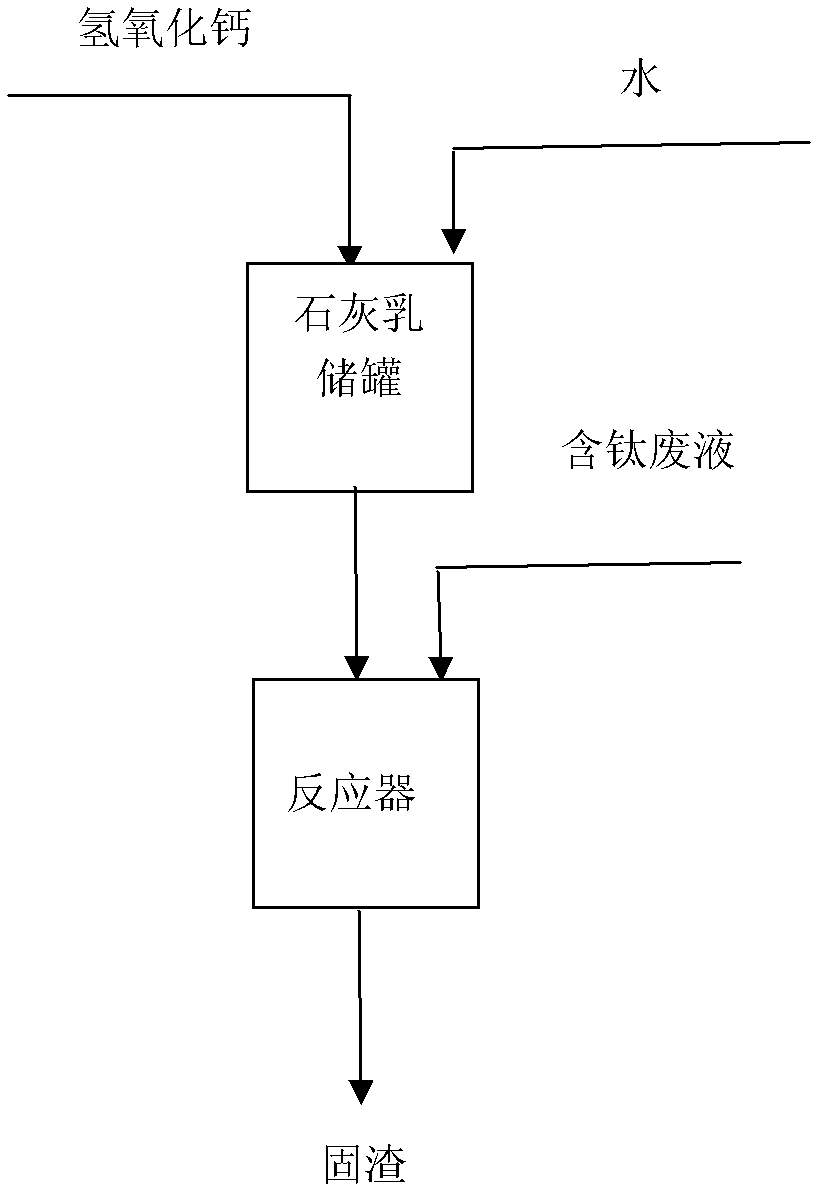
Method for treating titanium tetrachloride-containing waste liquid
InactiveCN103028224AEasy to follow upReduce the amount of waterChemical protectionLiquid wasteReaction temperature
The invention discloses a method for treating a titanium-containing waste liquid, which belongs to the field of treatment of industrial waste liquids. The method comprises the following process steps of: preparing white lime (Ca(OH)2) and water into lime milk according to a certain proportion in a lime milk storage tank; putting a certain amount of the prepared lime milk into a reactor; and directly adding a certain amount of titanium-containing waste liquid into the reactor, controlling the adding speed of the titanium-containing waste liquid, keeping the reaction temperature below or equal to 90 DEG C, and finally reacting to form solid waste residues TiO2 / CaCl2.nH2O. White lime is taken as an alkali neutralizing agent, and the titanium-containing waste liquid directly undergoes a hydrolysis neutralizing reaction with lime milk without a hydrolysis reaction, so that the amount of water needed in the conventional method is saved, and solid waste residues are taken as a final treatment product and are convenient to transport; and moreover, conventional measures such as burning and landfilling can be adopted in a subsequent treatment way, so that the treatment cost is saved.
Owner:CHINA PETROLEUM & CHEM CORP +1
Method for preparation of unidimensional monocrystal titanium dioxide nano material
InactiveCN1699636ALow priceEasy to getPolycrystalline material growthFrom normal temperature solutionsPotassium hydroxideSingle crystal
The present invention belongs to the field of nanometer material technology. The procedure is: 1) adding 1-30 parts of TiCl4 or TiO2 powder to 70-99 parts of organic solvent, obtaining evenly distributed mixture, 2) dissolving 10-60 parts of potassium hydroxide in 40-90 parts of deionization water, obtaining NaOH solution, 3) adding 20-80 parts NaOH solution of step 2)to 10-50 parts solution of step 1), stirring and moving to an autoclave, thermostatically heating at 50-300 degree C, 4) cooling down and pouring out the supernatant liquid, washing the bottom precipitation with deionization water and diluted acid solution, followed by centrifugal separating and drying at 10-200 degree C. The product obtained is one dimension single crystal nanometer material (nano-wire, nano-rod, and nano-tube) of TiO2 with diameter of 10-100 nm and length of micrometer order of magnitude. The product of the present invention is of high purity and high yield rate, and the form and pattern can be adjusted through selecting different solvents.
Owner:TECHNICAL INST OF PHYSICS & CHEMISTRY - CHINESE ACAD OF SCI
Particulate titanium oxide, method and apparatus for manufacturing the same, and treatment methods using such titanium oxide
InactiveUS20050271578A1No decline in photocatalytic activityReduce probabilityPolycrystalline material growthFrom normal temperature solutionsParticulatesSingle crystal
A particulate titanium oxide is obtained which has a large specific surface area and a high crystallinity with few internal defects, and thus a high photocatalytic activity as a photocatalyst is expected. Particulate titanium oxide in which the particles are box-shaped polyhedra is used as the photocatalyst. The particles are each a box-shaped polyhedron composed of one or more titanium oxide single crystalline polyhedron. When this single crystalline polyhedron has a flatness ratio of 0.33 to 3.0, the crystallinity is even higher. The particulate titanium oxide typically has a rutile transition ratio R(700-24) of not more than 7.5% and a rutile transition ratio R(500-24) of not more than 2.0%. Titanium oxide particles of these shapes are manufactured by feeding titanium tetrachloride vapor and oxygen into a reaction tube made of silica glass and applying heat from outside the tube to effect thermal oxidation.
Owner:THE FUJIKURA CABLE WORKS LTD
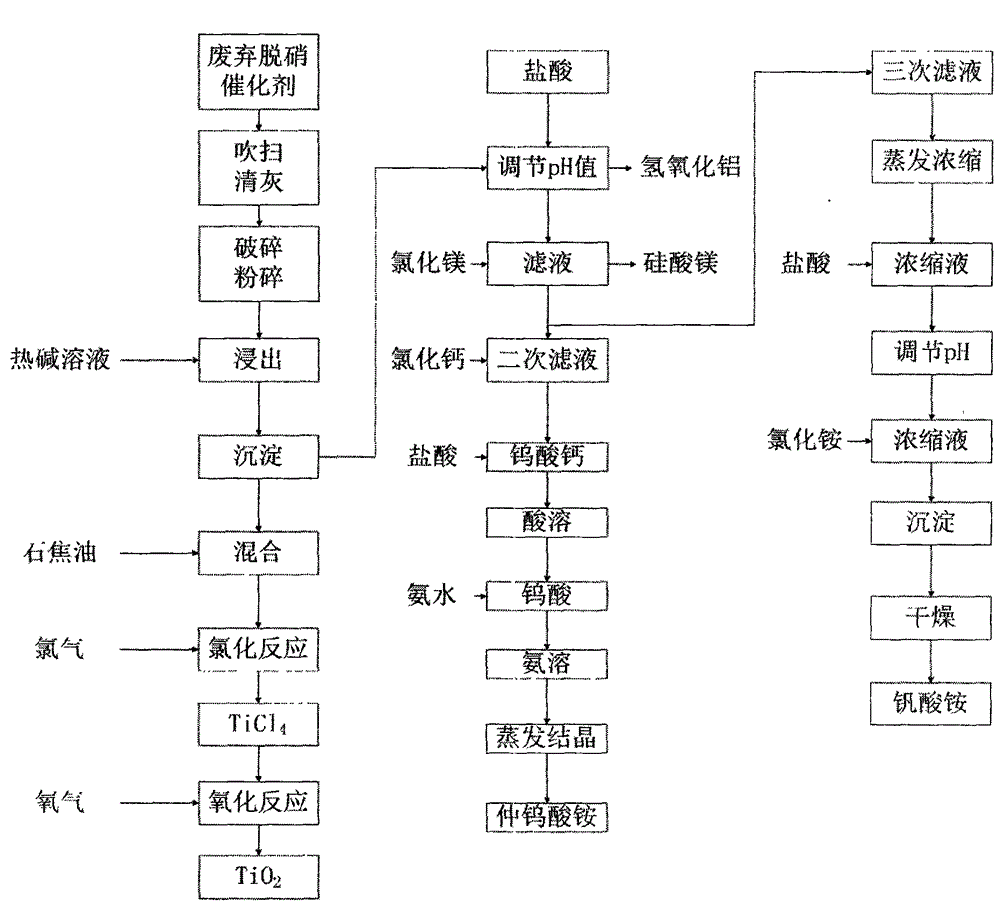
Recycling method for waste denitration catalyst
InactiveCN104099476AEmission reductionShort production processProcess efficiency improvementMagnesium saltAmmonium paratungstate
A recycling method for waste denitration catalyst is characterized by comprising the following steps: (1) the waste denitration catalyst is smashed into powder of 100 to 200 meshes; (2) the powder reacts with an alkali solution under a heating and stirring condition to obtain a titanium-rich material and a solution containing elements such as vanadium, tungsten, silicon and aluminum; (3) the titanium-rich material reacts with chlorine gas to generate titanium tetrachloride, then the titanium tetrachloride is condensed and reacts with oxygen to generate titanium dioxide, and the titanium dioxide is treated with surface finish and drying to obtain a titanium dioxide finished product; (4) the pH value of the solution obtained in the step (2) is adjusted; a magnesium salt is added into the solution to remove silicate ions and obtain a solution containing vanadium and tungsten; calcium chloride powder is added into the solution containing vanadium and tungsten to generate calcium tungstate; the calcium tungstate reacts with hydrochloric acid to generate tungstic acid; the tungstic acid is treated with ammonia dissolution and evaporative crystallization to obtain ammonium paratungstate crystals; (5) a precipitant ammonium chloride is added, and centrifugal drying is carried out to obtain solid ammonium vanadate. The method has the advantages that the operation is easy; three wastes are reduced; the economic efficiency is improved; the waste denitration catalyst is recycled.
Owner:CECEP L&T ENVIRONMENTAL TECH
Spherical catalyst for 1-butene polymerization as well as preparation method and application thereof
The invention relates to polymerization of 1-butene, in particular to a spherical catalyst of polybutylene-1. The catalyst consists of a carrier, an active component and a modifier, wherein the active component is titanium tetrachloride, the carrier is a magnesium chloride spherical carrier and the modifier is an electron donor compound; and the catalyst comprises the major components in percentage by mass of: 1.1-4.5% of Ti and 0.15-12.37% of electron donors. The spherical catalyst suitable for butane-1 polymerization solves the problems that the activity of the general catalyst is low, the normality of polymer is difficult to control, and the molecular weight of the polymer and the molecular weight distribution are difficult to control. The obtained polybutylene polymer has very high steric regularity, the normality of polybutylene is as high as 99%, the crystallinity of the polybutylene-1 polymer is greater than 60% and the fusion point is 130.5 DEG C. The invention can simplify the polymerizing process and save the production cost.
Owner:HEBEI UNIV OF TECH
Preparation method of ordered macroporous materials with titanium dioxide inverse opal structures
InactiveCN102336434AHigh degree of orderImprove uniformityTitanium dioxideEmulsion polymerizationRoom temperature
The invention discloses a preparation method of ordered macroporous materials with titanium dioxide inverse opal structures, which comprises the following steps of: step 1, adopting an emulsifier-free emulsion polymerization method, preparing PS micro-balloons by taking a PS monomer as raw materials and dispersing the micro-balloons into ethanol solution to form PS micro-balloon dispersion liquid; step 2, utilizing the PS micro-balloon dispersion liquid in the step 1 to deposit and prepare a PS template with an opal structure by adopting a vertical deposition method under the conditions of constant temperature and constant humidity; step 3, preparing titanium tetrachloride hydrochloric acid solution, vertically immersing the PS template prepared in the step 2 into the titanium tetrachloride hydrochloric acid solution and heating to 70-90 DEG C, slowly filling hydrated titanium oxide on the PS temperate in a constant-temperature state and then taking out after 3-5 hours; and step 4, placing the PS template treated by the step 3 at a room temperature for drying, treating for 30 minutes at a high temperature and obtaining the ordered macroporous materials with the TiO2 inverse opal structures. Compared with the prior art, the prepared materials are uniform, have high order degree, less impurity and simple preparation process and are beneficial to popularization and implementation.
Owner:SOUTHWEAT UNIV OF SCI & TECH
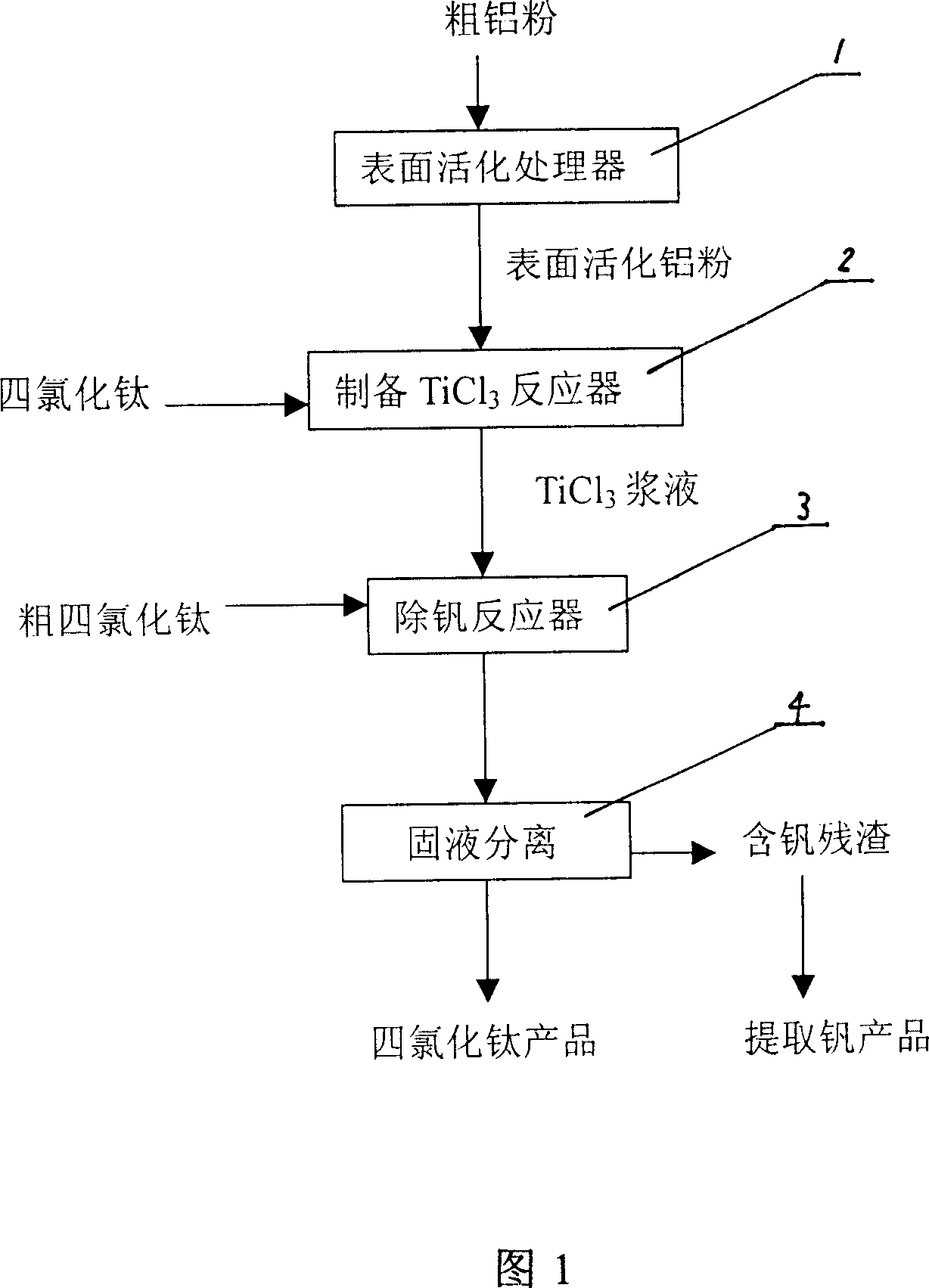
Method for removing vanadium in titanium tetrachloride
InactiveCN101054201ASimple processSimple waste treatmentTitanium halidesTetrachlorideBiological activation
The invention relates to a method for removing vanadium impurity from titanic tetrachloride. The invention prepares titanium trichloride serum by the reaction between the cost aluminum powders with the average grain size no bigger than 150mum after surface activation treatment. TiCl3 in the serum reduces VOCl3 in the titanic tetrachloride to be VOCl2 deposit so as to remove the vanadium. And the titanic tetrachloride after removing the vanadium contains no more than 0.0007% vanadium. The process of vanadium removal can be implemented together with rectification process in order to realize continuous operation. The invention has the advantages of simple in process, low in cost, safe in operation, high in product quality and recovery rate and simple in treating the ''three-waste'' pollution.
Owner:GENERAL RESEARCH INSTITUTE FOR NONFERROUS METALS BEIJNG +1