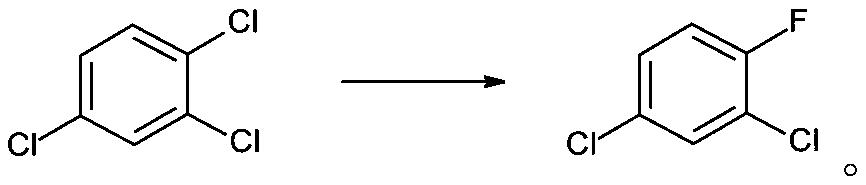
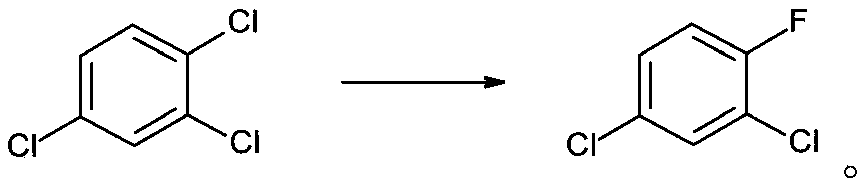
Synthesis method of 2, 4-dichlorofluorobenzene
A technology of dichlorofluorobenzene and a synthesis method, which is applied in the field of medicinal chemical synthesis, can solve the problems of high cost, high toxicity and high safety risk, and achieves the effects of short reaction route and safe synthesis process
Active Publication Date: 2019-06-07
ZHEJIANG BENLI TECH CO LTD
View PDF6 Cites 2 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
[0010] The purpose of the present invention is to provide a synthesis method of 2,4-dichlorofluorobenzene, which overcomes the disadvantages of the current synthesis of 2,4-dichlorofluorobenzene, such as long process route, high safety risk, high toxicity, difficulty in treating three wastes and high cost
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
specific Embodiment 1
[0030] 5.74g of trimethylolpropane borate, 1.08g of p-tert-butylcalix[6]arene and 35.15g of KF were added to a solution of 100g of 1,2,4-trichlorobenzene dissolved in 150mL of sulfolane. The mixture was transferred into a 500mL pressure reactor, purged with nitrogen and replaced, sealed, heated to 185°C, and maintained at this temperature for 5h. The reactor was cooled to normal temperature, opened, and filtered to obtain 43.56 g of potassium salt; the filtrate was rectified under reduced pressure to obtain 83.66 g of 2,4-dichlorofluorobenzene with a yield of 92%.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention discloses a synthetic method of 2, 4-dichlorofluorobenzene. The method comprises the following steps: 1,2,4-trichlorobenzene is used as a raw material, a calix[6]arene and trimethylolpropane borate compound is used as a phase transfer catalyst, spray-dried potassium fluoride is used as a fluorinating agent, and the 2,4-dichlorofluorobenzene is produced through one step of reaction; herein, the molar ratio of trimethylolpropane borate to potassium fluoride is 1:10-1:20, preferably 1:15; the molar ratio of the potassium fluoride to the 1,2,4-trichlorobenzene is 1:1.1; the calix[6]arene is p-tert-butyl calix[6]arene; and the molar ratio of the p-tert-butyl calix[6]arene to the 1,2,4-trichlorobenzene is 0.001:1-0.05:1, preferably 0.02:1. By using the calixarene and trihydroxymethyl borate composite catalyst, the catalytic efficiency is greatly improved, and the product yield is as high as 92%. The boiling point of the 2,4-dichlorofluorobenzene is 168-169 DEG C, the boiling point of the raw material 1,2,4-trichlorobenzene is 214 DEG C, and the boiling point of the solvent sulfolane is 285 DEG C, so that the product is simple to separate and purify. The method has the advantages of one-step synthesis, short reaction route, avoidance of nitrification and high-temperature chlorination, safer synthesis process and suitability for industrial production.
Description
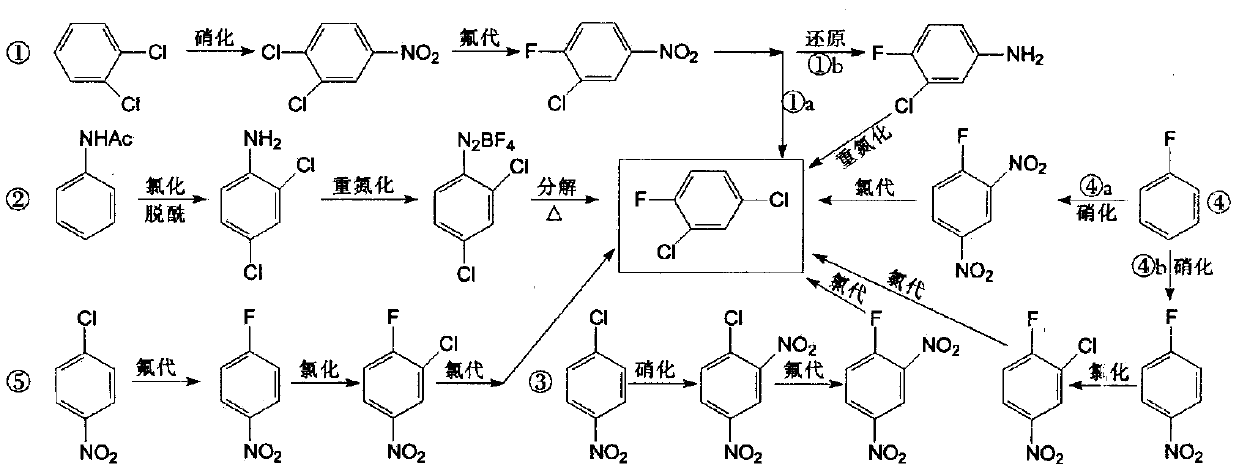
technical field [0001] The invention belongs to the technical field of pharmaceutical chemical synthesis, and in particular relates to a synthesis method of 2,4-dichlorofluorobenzene. Background technique [0002] 2,4-Dichlorofluorobenzene is a key pharmaceutical intermediate used in the production of fluoroquinolone antibacterial agents such as fleroxacin, pefloxacin, ciprofloxacin and norfloxacin, and can also be used in liquid crystal materials Synthesis. [0003] There are many synthetic routes of 2,4-dichlorofluorobenzene, which are summarized in the following formula: [0004] [0005] Among them, route ① is the current mainstream production process. [0006] The process route is long, and mixed acid nitration and high-temperature chlorine gas chlorination processes are adopted, which has high safety risks, high toxicity, and high difficulty and cost in the treatment of three wastes. [0007] The synthesis of the key intermediate 3-chloro-4-fluoronitrobenzene in ...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): C07C17/20C07C25/13
Inventor 余永志顾海宁吴政杰
Owner ZHEJIANG BENLI TECH CO LTD