Polypeptide compositions toxic to anthonomus insects, and methods of use
a technology of polypeptide compositions and anthonomus insects, applied in the field of molecular biology, to achieve the effect of increasing or enhancing the resistance of such a plant to attack
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
5.1 Example 1
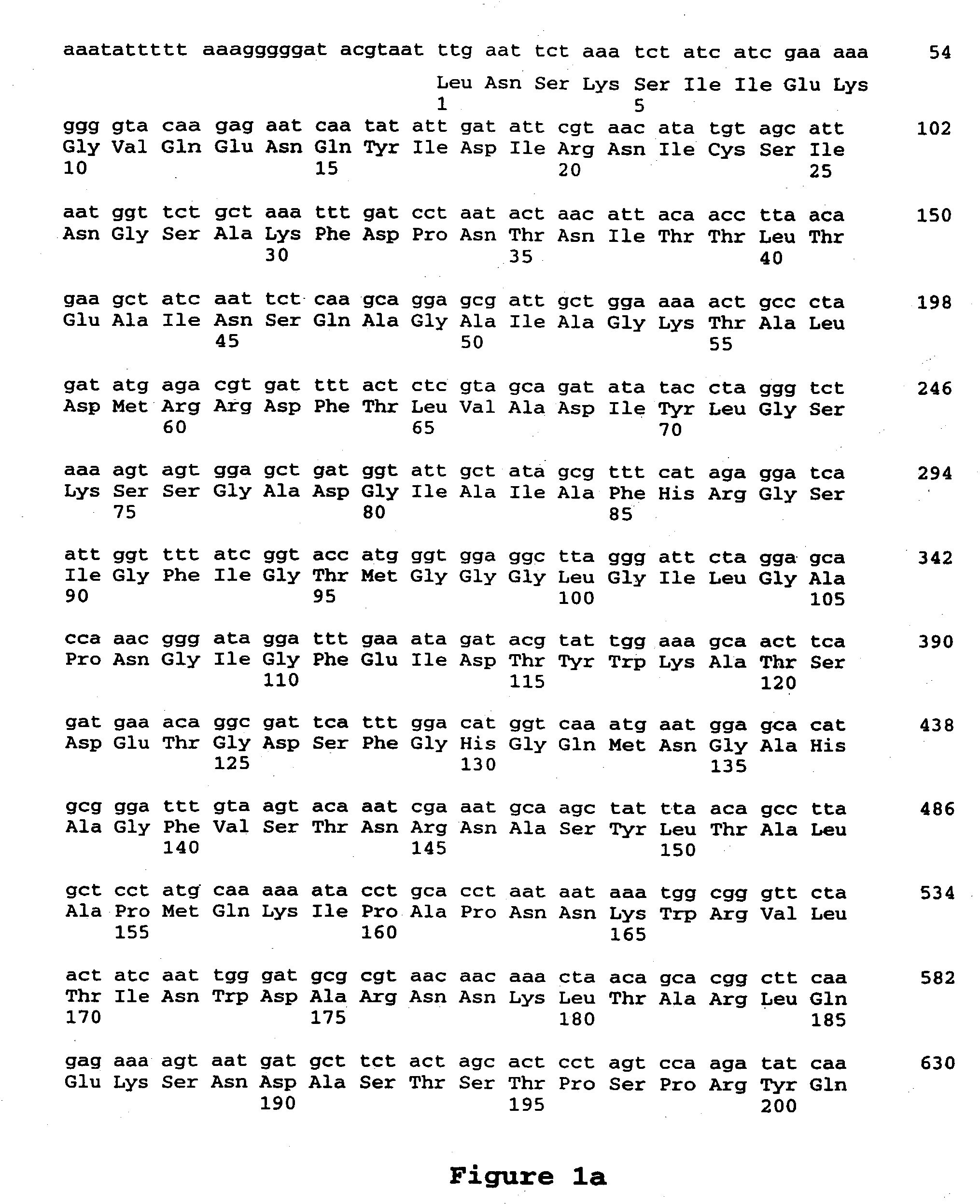
[0196] Bacillus thuringiensis Stains with Sequences Related to CryET70
[0197] We previously identified a B. thuringiensis strain expressing a protein which we designated CryET70. The CryET70 protein had effective coleopteran specific bioactivity when provided in bioassay feeding studies to western corn rootworm larvae, but not against southern corn rootworm larvae. We were interested in identifying additional B. thuringiensis strains which contained DNA encoding CryET70 and closely related genes. Colony blot hybridization experiments were completed as indicated below, using a probe prepared from cryET70 DNA. Wild-type B. thuringiensis strains were patched onto LB plates and incubated at 30.degree. C. for four hours. A Nytran.RTM. Maximum-Strength Plus (Schleicher and Schuell, Keene, N.H.) circular (82 mm) membrane filter was then placed on the plates and the plates and filters were incubated at 25.degree. C. overnight. The filters, which contained an exact replica of the...
example 2
5.2 Example 2
[0199] Production of Antibody to CryET70
[0200] CryET70 specific polyclonal antibody was prepared so that proteins containing CryET70-related epitopes could be identified using immunological methods. Recombinant B. thuringiensis strain EG11839 containing plasmid pEG1648 expressing CryET70 was grown in C2 medium for four days at 25.degree. C. The resulting spores and crystals were washed in 2.5.times. volume H.sub.2O and resuspended at 1 / 20 the original volume in 0.005% Triton X-100.RTM.. The spore-crystal suspension was then loaded on a sucrose step gradient consisting of 79%, 72% and 55% sucrose. The gradient was spun overnight in a Beckman SW28 at 18,000 RPM. CryET70 crystals banded between the 79% and the 72% sucrose layers. CryET70 crystals were washed several times in H.sub.2O and resuspended in 0.005% Triton X-100.RTM.. The purified crystals were then solubilized in 50 mM sodium carbonate (pH 10), 5 mM DTT, and any contaminating vegetative cells or spores were remo...
example 3
5.3 Example 3
[0201] Southern and Western Blot Analyses
[0202] Strains identified in Example 1 as containing sequences related to cryET70 were examined further by Southern and Western blot analyses.
[0203] Total DNA was prepared from the strains by the following procedure. Vegetative cells were resuspended in a lysis buffer containing 50 mM glucose, 25 mM Tris-HCl (pH8.0), 10 mM EDTA, and 4 mg / ml lysozyme. The suspension was incubated at 37.degree. C. for one hr. Following incubation, SDS was added to 1%. The suspension was then extracted with an equal volume of phenol:chloroform:isoamyl alcohol (50:48:2). DNA was precipitated from the aqueous phase by the addition of one-tenth volume 3 M sodium acetate, and two volumes of 100% ethanol. The precipitated DNA was collected with a glass rod, washed with 70% ethanol, and resuspended in dH.sub.2O.
[0204] Total DNA was digested with EcoRI and separated on a 0.8% agarose gel in TAE buffer (40 mM Tris-acetate, 2 mM Na.sub.2EDTA, pH 8). The DNA ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More