Problems of existing methods and equipment are mainly due to the increase in the use of isotopes whose radiation energy is relatively high, and by the fact that a higher level of
automation is required due to the short half-life of said isotopes.
This handling exposes the hospital staff on a repeated basis, giving rise to an appreciable cumulative
exposure.
This
dose is generally limited by performing the operation behind lead protection such as a radiation-shielded glove box.
It is slow and labor-intensive.
It should be noted that
automation of the filling of syringes is possible and that such devices exist, but they do not solve all the production problems: the fixing of the needle or a stopper is not automated, nor is the placing of the
syringe in its transportation container.
The integrity of the product is not ensured during the transportation.
A multi-
dose radiopharmaceutical
vial dispensing device is of little interest to the American market since it does not allow the automatic filling of individual patient-doses which may be used directly in hospitals according to the common practice.
syringes are a form of packaging which does not lend itself readily automation.
The design of a
system for the automated filling of syringes, which would directly satisfy the need and habits of the American market, would be complex;
the filling of a
syringe as is currently performed in American radiopharmacies, in particular for PET applications, is essentially manual, slow and relatively inefficient;
However the configuration of this device implies that, before transportation, the
syringe has to be filled and the needle capped manually, thus exposing the operator.
Thus the
medical staff carrying out on-site injection is further exposed during uncapping and during injection. the fixing of the needle or a stopper is not automated, nor is the placing of the syringe in its transportation container.
The integrity of the product is not ensured during the transportation.
A multi-dose radiopharmaceutical
vial dispensing device is of little interest to the American market since it does not allow the automatic filling of individual patient-doses which may be used directly in hospitals according to the common practice.
syringes are a form of packaging which does not lend itself readily to automation.
The design of a
system for the automated filling of syringes, which would directly satisfy the need and habits of the American market, would be complex;
the filling of a syringe as is currently performed in American radiopharmacies, in particular for PET applications, is essentially manual, slow and relatively inefficient;
A first drawback of this device is the need to manually remove a sealing rubber stopper at the front face of the syringe in order to place the needle, which leads to personal exposure and possible
sterility loss or biological
contamination.
Moreover the cantilevered
gasket-
plunger connection is weakened owing to the weight of shielded parts. the fixing of the needle in a stopper is not automated, nor is the placing of the syringe in its transportation container.
This manipulation represents an exposure to radiation and presents a risk of
radioactive contamination of the staff, and also a risk of biological contamination of the substance.
A multi-dose radiopharmaceutical
vial dispensing device is of little interest to the American market since it does not allow the automatic filling of individual patient-doses which may be used directly in hospitals according to the common practice.
syringes are a form of packaging which does not lend itself readily to automation.
The design of a
system for the automated filling of syringes, which would directly satisfy the need and habits of the American market, would be complex;
the filling of a syringe as is currently performed in American radiopharmacies, in particular for PET applications, is essentially manual, slow and relatively inefficient
Possible contamination or exposure of the radiopharmacies staff is thus a risk inherent to
manual handling of syringes.
However usual syringes constitute a form of packaging which does not lend itself readily to automation.
The design of a system for the automated filling of syringes, which would directly satisfy the need and habits of the American market, would be complex;
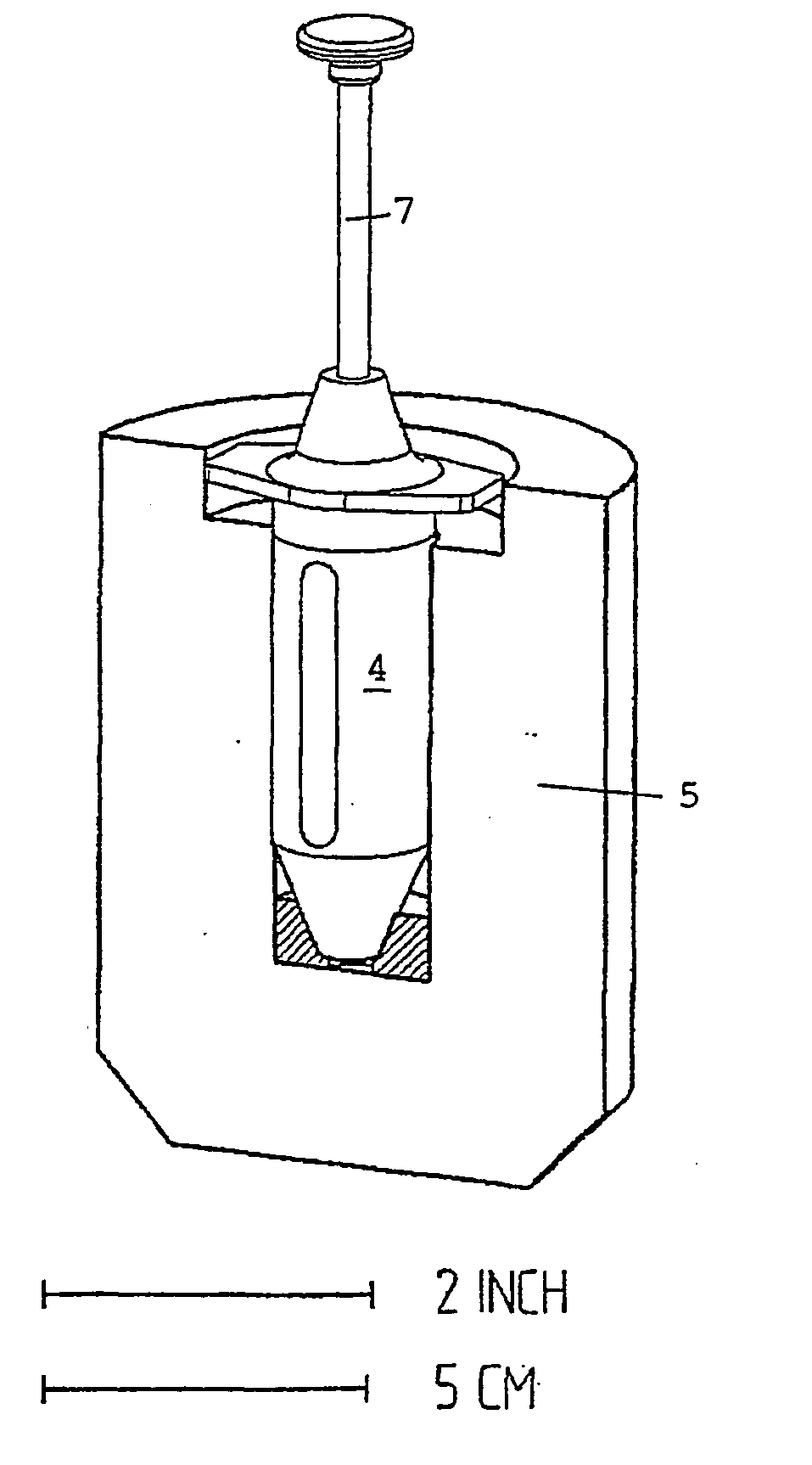
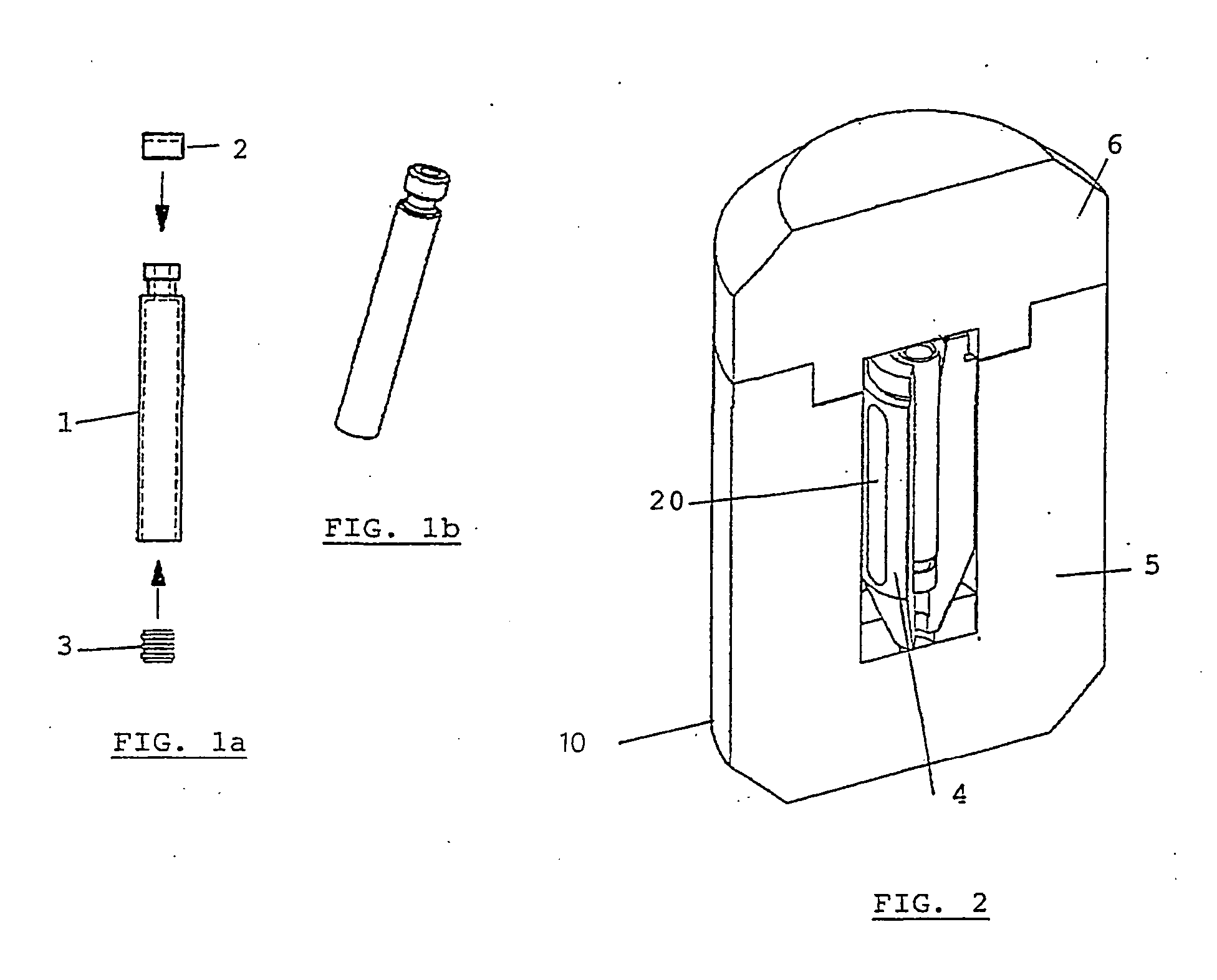
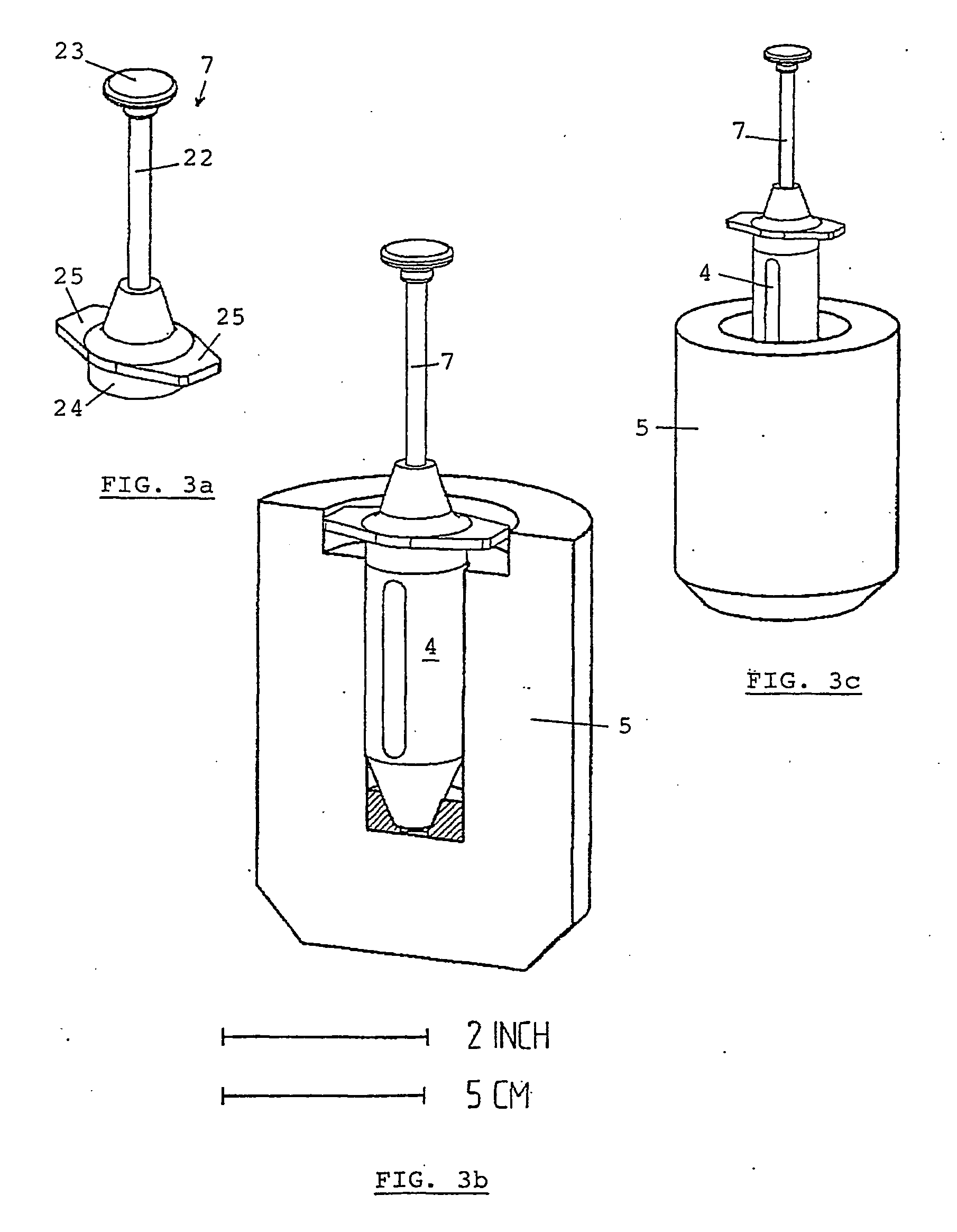
the use of individual syringes does not entirely avoid manipulations at the hospital since they must be transferred from their transportation radiation shielding container to the syringe radiation shielding;
pre-filled individual syringes, possibly comprising needle, syringe body and
plunger, are very long and require large, and thus heavy, transportation radiation shielding containers;
when the syringes are delivered with a stopper, an additional manipulation is necessary, which exposes both the product and the operator.
 Login to View More
Login to View More