Method of inhibiting restenosis using bisphosphonates
a technology of bisphosphonates and restenosis, which is applied in the direction of phosphorous compound active ingredients, inorganic non-active ingredients, drug compositions, etc., can solve the problems of precisely the problem, the patient is placed at risk of a variety of complications, and the proportion of restenosis is substantial, so as to achieve the effect of treating or preventing restenosis and preventing restenosis
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Liposomes of Clodronate
[0063] Stock solutions of clodronate were prepared by dissolving the drug in deionized water at a concentration of 0.11 M, pH=7.
Liposome Preparation
[0064] 38.9 mg of distearoylphosphatidylglycerol (DSPG), 118.5 mg of distearoyl-phosphatidylcholine (DSPC) and 38.7 mg of cholesterol were accurately weighed and dissolved in 20 ml of chloroform: methanol (9:1) in a round bottom vial. The vial was gently warmed, and the solvent was then evaporated in rotavapor. 20 mls of hydrated diisopropylether were then added and the vial was put into a water bath until the contents were dissolved. 8 mls of the clodronate solution prepared as described above were then added, and the solution was sonicated at 55° C. for a period of 45 minutes. The organic phase was then evaporated in rotavapor (55° C., 100 rpm). Similarly, other drug-containing liposomes can be prepared.
Purification of Prepared Liposomes
[0065] A Sephadex gel was prepared by dissolving 2.6 grams of Sephade...
example 2
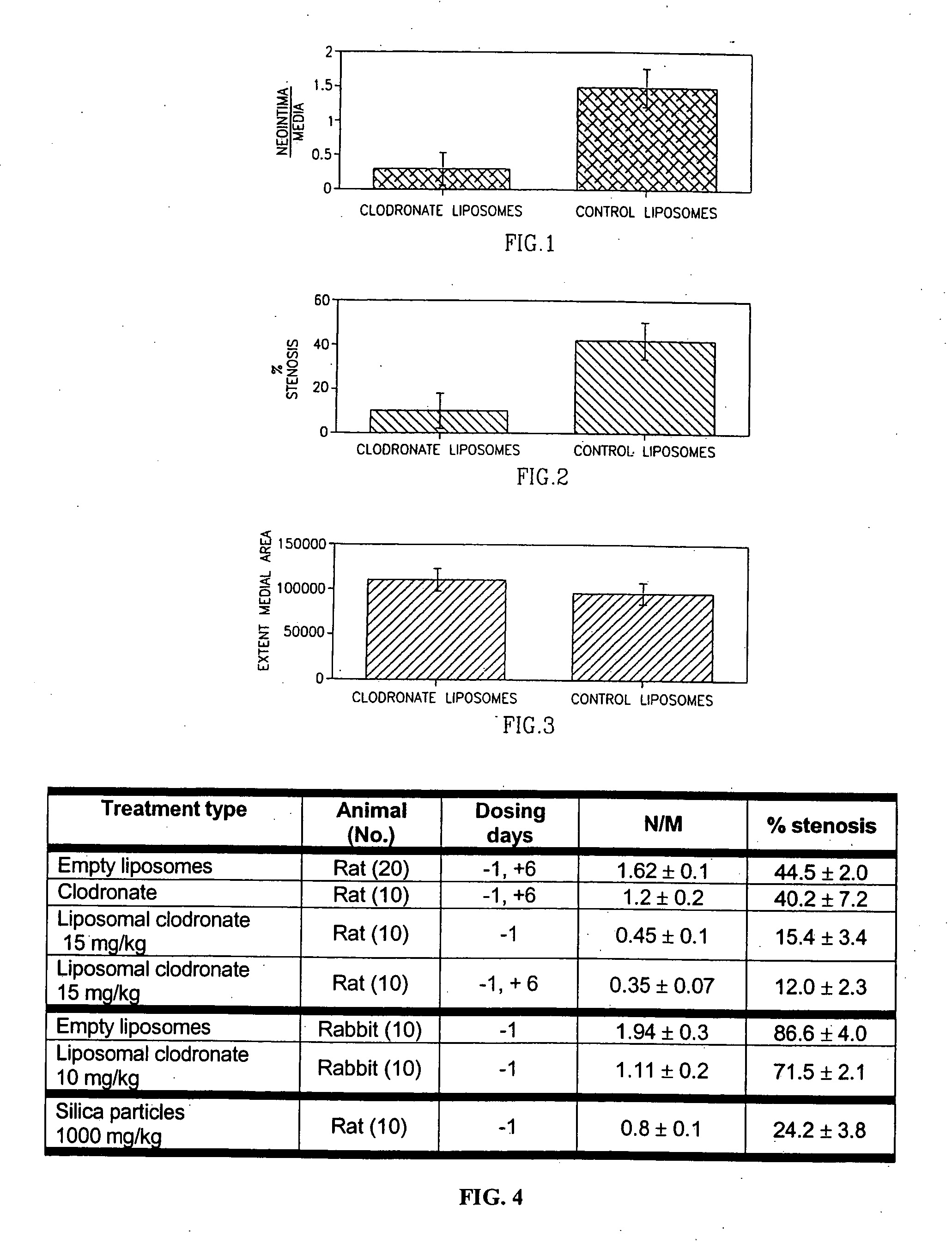
[0074] The antirestenotic effects of liposomal clodronate injections were studied in the balloon-injured rat and hypercholesterolemic rabbit carotid arterial models. The rats were treated by clodronate-containing liposomes, empty liposomes (control), and clodronate in solution (additional control). The dose of clodronate injected was 1.5 and 15 mg / kg administered one day before procedure (−1) and / or on day 6 (+6) post injury. The rabbits (following 30 days of atherosclerotic diet) were treated one day prior to balloon angioplasty by liposomal clodronate (10 mg / kg). The lumen, neointimal, medial and vessel areas and volumes were measured in the treated and control animal groups by digital planimetry of histological sections, at 14 and 30 days post injury in the rat and rabbit models, respectively.
[0075] The results of the antirestenotic effects of liposomal clodronate are shown in FIG. 4. As illustrated, no significant differences were found between treatments with empty liposomes, ...
example 3
Effect of Liposomal Clodronate on IL-1β Production and Transcription and MMP-2 Activity
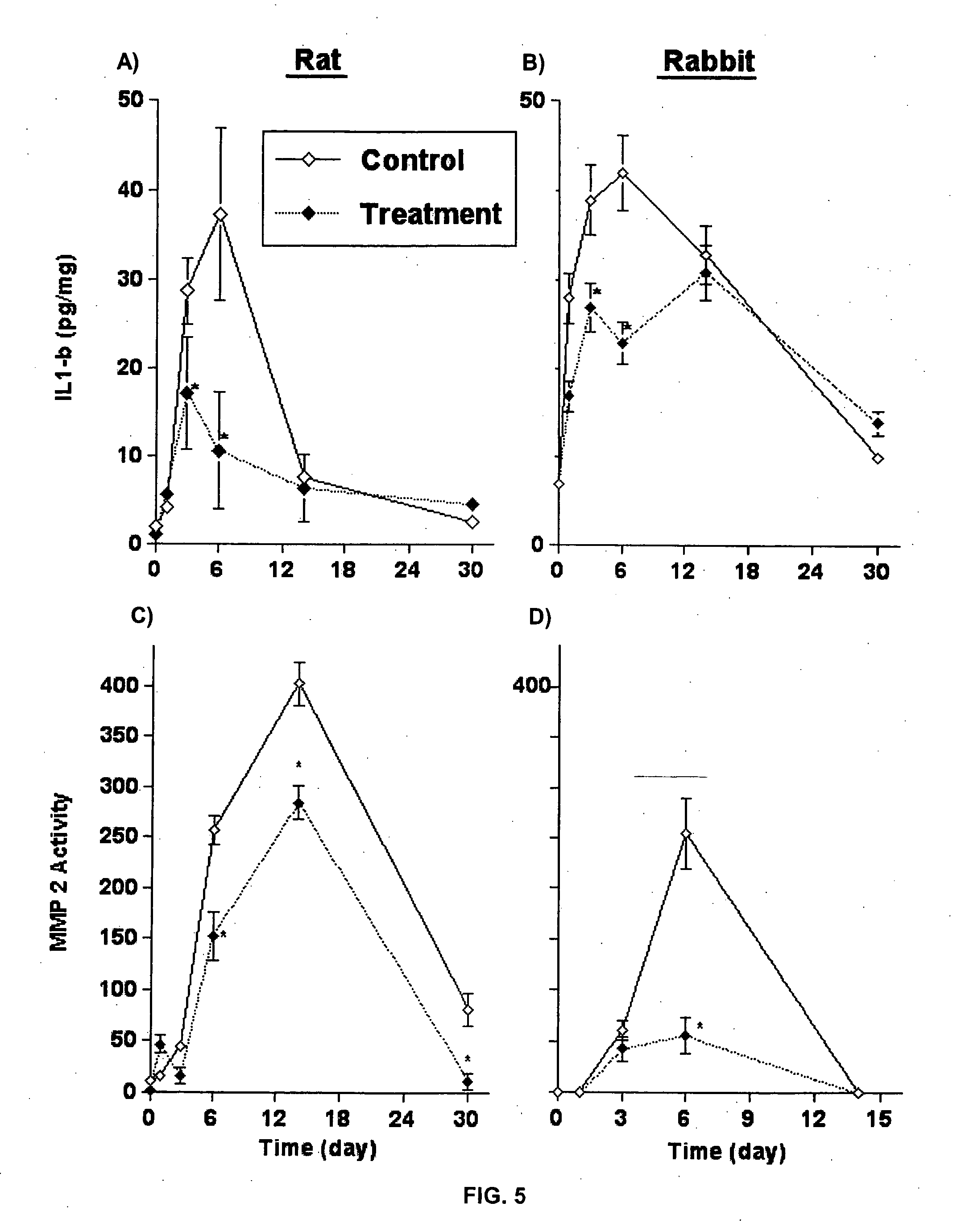
[0078] The effects of liposomal clodronate on interleukin 1-β (IL-1β) production and transcription and matrix metalloproteinase-2 (MMP-2) activity were studied in the balloon-injured rat and the hypercholesterolemic rabbit carotid arterial models. A group of male Sabra Rats was prepared according to the rat carotid catheter injury model described supra, in Example 1. The hypercholesterolemic rabbit model consisted of New Zealand White rabbits weighing 2.5-3.5 kg. The rabbits were fed an atherogenic diet of 2% cholesterol and 6% peanut oil starting 30 days before angioplasty and hypercholesterolemia was established (plasma cholesterol>1,200 mg / dL). The rabbits were then anesthesized by xylazine (7 mg / kg) and ketamine (40 mg / kg). Heparin (200 units / kg), atropine (0.05 mg) and norfloxacin nicotinate (70 mg) were also administered to the rabbits. Thereafter, balloon injury was performed on the left c...
PUM
| Property | Measurement | Unit |
|---|---|---|
| diameter | aaaaa | aaaaa |
| diameter | aaaaa | aaaaa |
| size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More