Polypeptides substances, preparing method and use thereof
A technology of uses and aspects, applied in the preparation methods of peptides, peptide/protein components, chemical instruments and methods, etc., can solve the problems of peptide sequences and uses without relevant patents and literature reports, and achieve low cost, clear structure, and activity. high effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
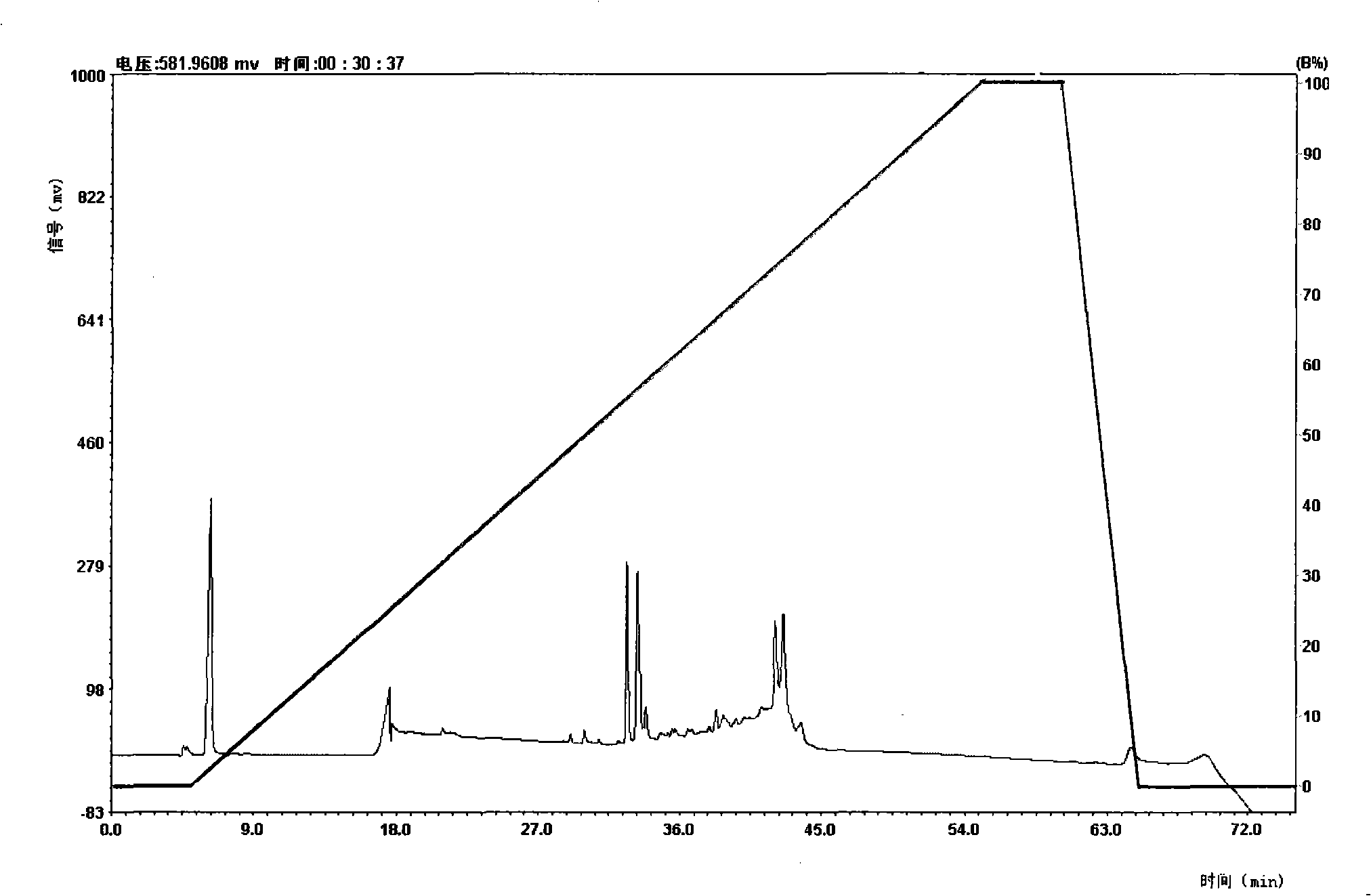
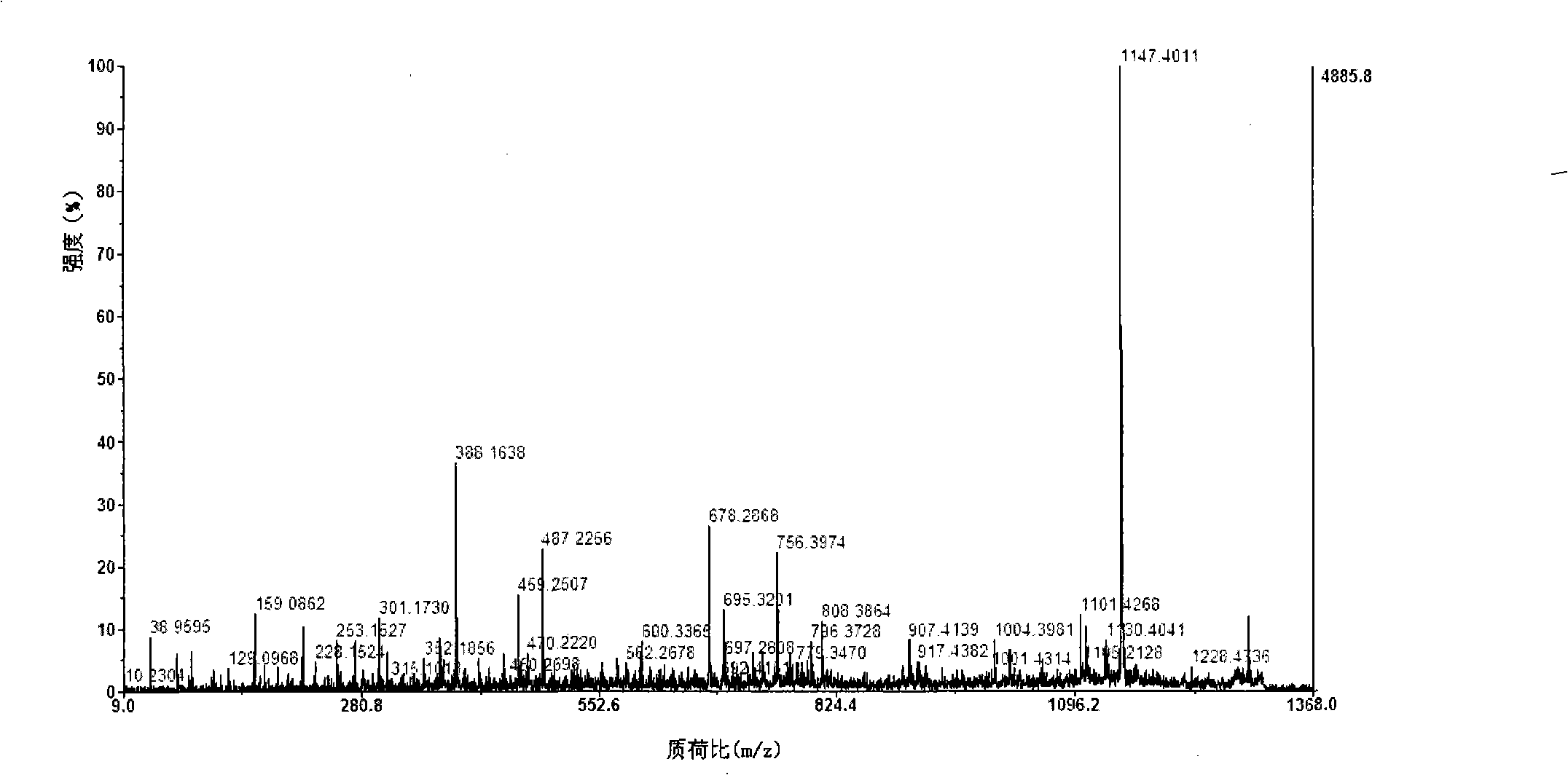
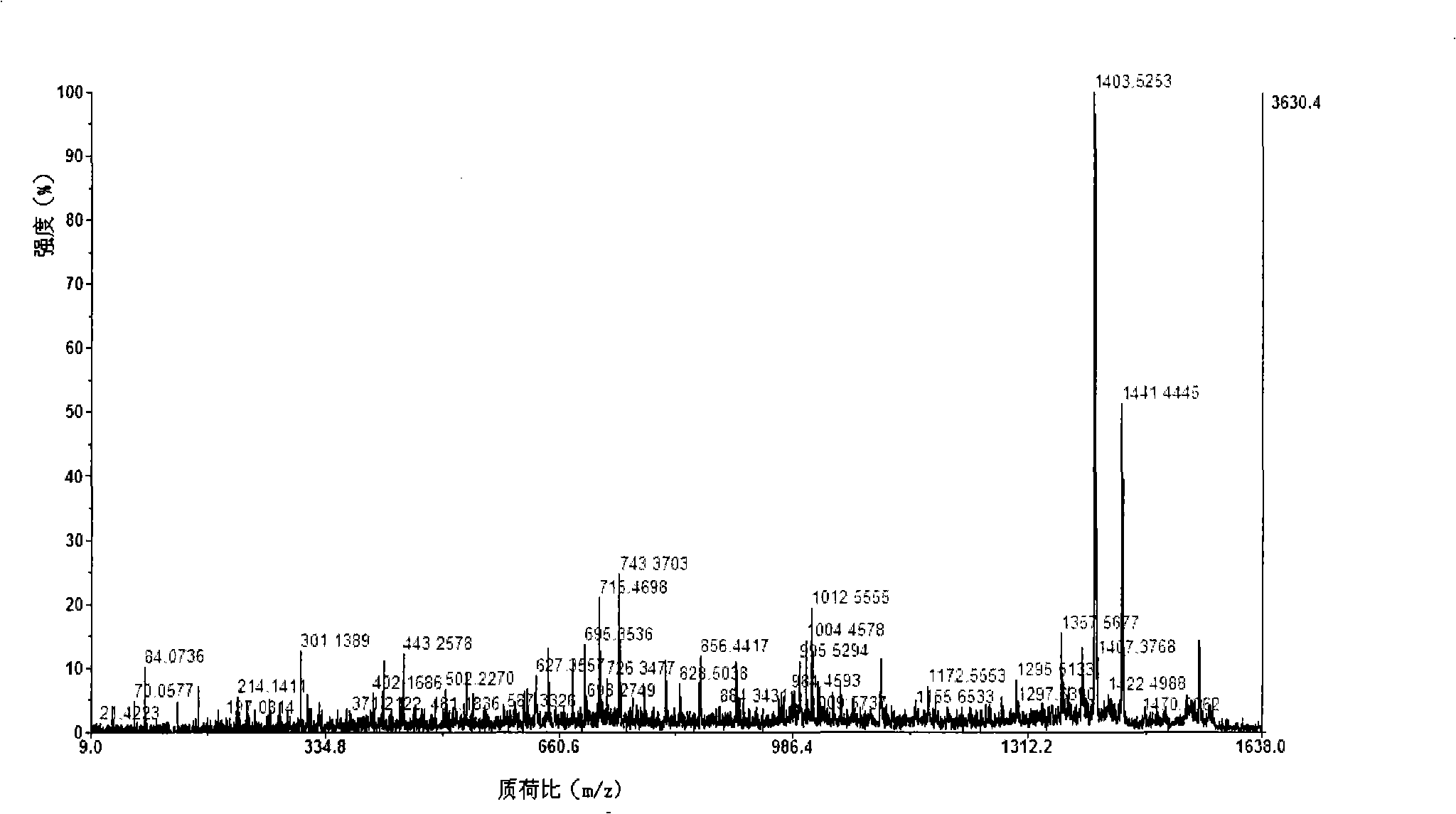
Image
Examples
Embodiment 1
[0058] This example relates to the synthesis of a polypeptide (cardiopeptide C).
[0059] The sequence of the polypeptide is Lys-Gly-Ala-Trp-Ser-Asn-Val-Leu-Arg-Gly-Met-Gly-Gly-Ala-Phe
[0060] 1. Synthesis: First, the amino group of the amino acid contained in the polypeptide is protected with an amino protecting group (9-fluorenylidene methyl ester group) (amount of 20-100 g / g), and the wang resin is swollen with chloroethyl ether. The amount of chloroethyl ether used is 50-100 ml / g, and then the first amino acid is added to the resin, thereby immobilizing it on the resin. After washing with dichloromethane, the protecting group of the amino group can be deprotected with hexahydropyridine (10-80%, preferably 20-40%). At the same time, the carboxyl group of the second amino acid was activated by HBTU[2-(1H-benzotriazolyl)-1,1,3,3-tetramethylfurfural-hexafluorophosphate] and di After isopropylethylamine is neutralized, it is added to the resin combined with the first amino a...
Embodiment 2
[0062] This example relates to the chemical synthesis method of cardiopeptide X.
[0063] The amino acid sequence of the cardiac peptide C is: Trp-Ser-Asn-Val-Leu-Arg-Gly-Met-Gly-Gly-Ala-Phe.
[0064] The method used is as in Example 1.
Embodiment 3
[0066] This example relates to the chemical synthesis method of cardiopeptide C.
[0067] The sequence of the polypeptide is Lys-Gly-Ala-Trp-Ser-Asn-Val-Leu-Arg-Gly-Met-Gly-Gly-Ala-Phe.
[0068] Described method comprises the following steps:
[0069] (1) Removal of protecting group Fmoc: take Fmoc-Lys(Boc)-CLTR resin, soak and wash with dichloromethane 3 times, each time for 2min, then drain, add 50% piperidine in dichloromethane solution at room temperature 10mL, reacted for 5min and drained, and then reacted with the same amount of the above solution for another 30min, then drained, and then washed with 8mL-10mL of dichloromethane, ethanol and dichloromethane, drained, and took a small amount of resin (2mg~ 10mg) the total amino group was determined by the salicylaldehyde quantitative free radical method.
[0070] (2) Formation of peptide bonds (DCC-HOBt condensation method): Add the dichloromethane solution of Fmoc-Gly (t-Bu) and the dichloromethane of 1-hydroxybenzotria...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
| molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More