Nutritional composition for bariatric surgery patients
A composition, patient's technology, applied in the field of nutritional composition, able to solve the problems of clinical deficiency, reduced compliance, etc.
Inactive Publication Date: 2011-05-18
NV NUTRICIA
View PDF1 Cites 5 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
The need to replenish several times a day can lead to a decrease in compliance over time, thus leading to a lack of clinical
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
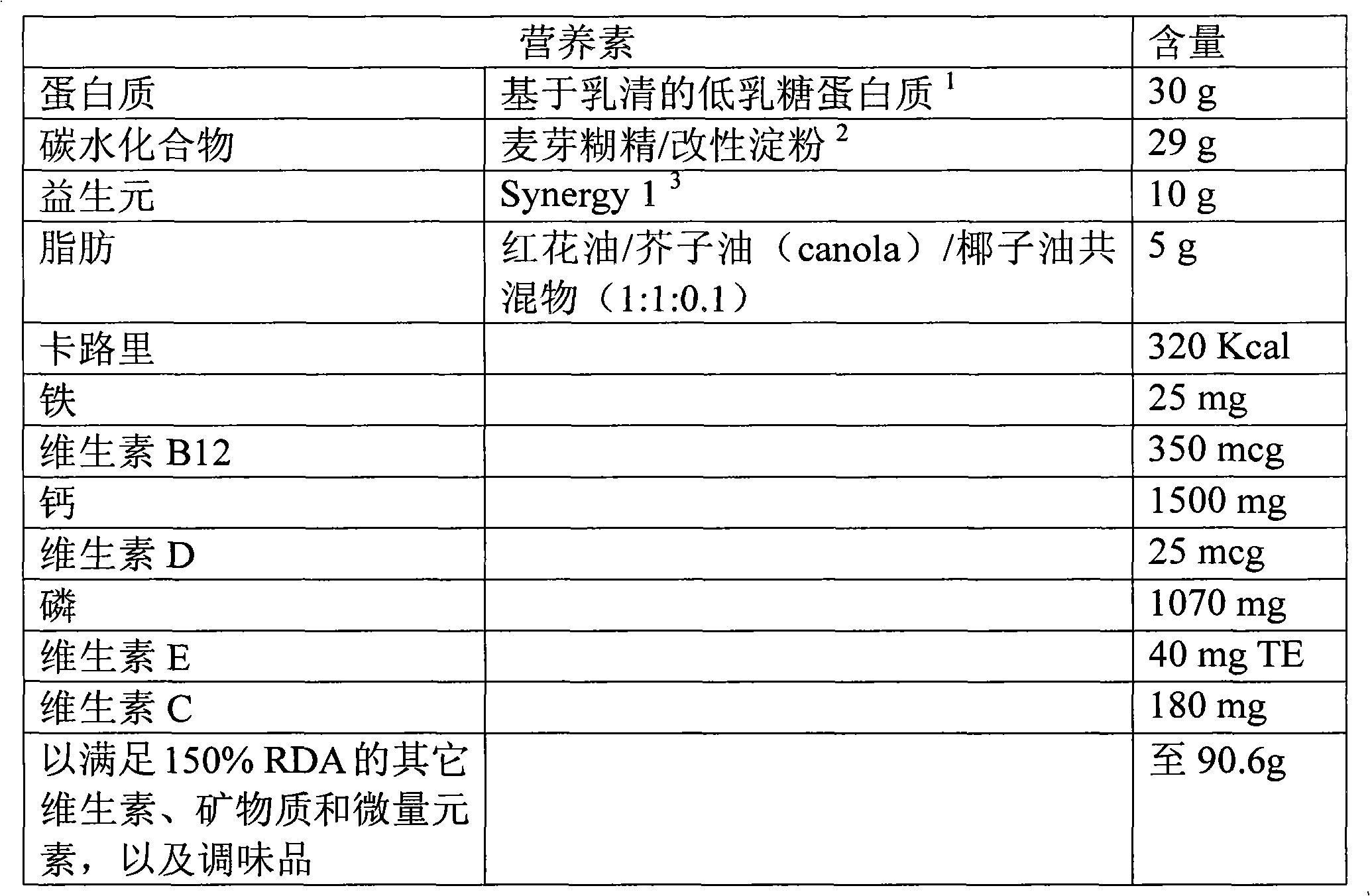
[0082] This example describes a preferred nutritional composition in powder form for use in the treatment of bariatric surgery patients.
[0083]
[0084] 1 Lactose content is about 0.25%
[0085] 2 MD:MS is about 3.3:1
[0086] 3 For example Orafti (about 50% short chain fructooligosaccharides and 50% long chain inulin)
[0087] As stated, the total weight of the composition was 90.6 g. This represents the amount administered to the patient per day. The product is packaged as two sachets, each containing 45.3 g of the formulation. Mix each sachet with water to obtain a liquid product with a volume of 125-135 mL. The patient takes the liquid product two times a day.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
A nutritional composition particularly suitable for use by morbidly obese patients pre and post bariatric surgery comprises 20-60 wt% protein source, 15 -55wt% carbohydrate, 10-20 wt% soluble dietary fibres, less than 10 wt% fat and micronutrients. The micronutrients comprise 0.75-3 wt% calcium, 0.5-1.8 wt% phosphorus, 12.5-100 mg / 100g of iron, 10-100 mcg / 100g vitamin D,and 175-560 mcg / 10Og vitamin B12 the wt% being based on the dry weight of the total composition.
Description
technical field [0001] The present invention relates to nutritional compositions which are particularly suitable for use before and after bariatric surgery in morbidly obese patients. When used in patients on a calorie-restricted diet in the pre-bariatric surgery phase, the product helps reduce liver size and further helps maintain bone health and optimal nutritional needs in post-bariatric surgery patients . [0002] The composition contains protein, digestible carbohydrates, dietary fiber, specific micronutrients, and preferably contains no or very low amounts of fat. Background technique [0003] The rising rate of obesity is a major health problem affecting millions of people in the Western world. Bariatric surgical procedures including gastric bypass represent a successful way to achieve significant weight loss in morbidly obese individuals. After surgery, it is estimated that up to one third of patients suffer from nutritional deficiencies that cannot be overcome by...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): A23L1/29A23L1/304A23L1/302A23L1/303A23L1/305A23L1/308A61K35/20A23L33/00A23L33/15A23L33/155
CPCA23V2002/00A23L1/303A23L1/302A23L1/308A23L1/296A23L1/304A23L1/305A23L1/3056A23L33/15A23L33/155A23L33/16A23L33/17A23L33/19A23L33/21A23L33/40A61P3/00A61P43/00A23V2250/54246A23V2250/54252A23V2250/5062A23V2250/706A23V2250/7106A23V2250/1592A23V2250/1578A23V2250/1618
Inventor J·E·兰福德F·F·塔库鲁库拉C·L·帕特森S·H·吉芬F·J·布罗迪
Owner NV NUTRICIA