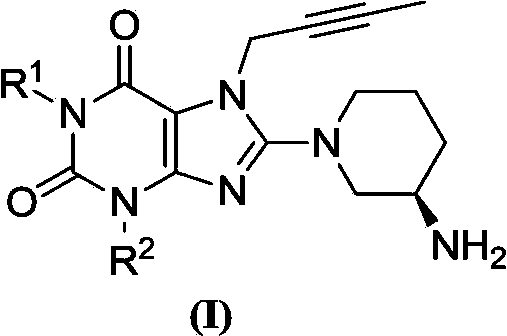
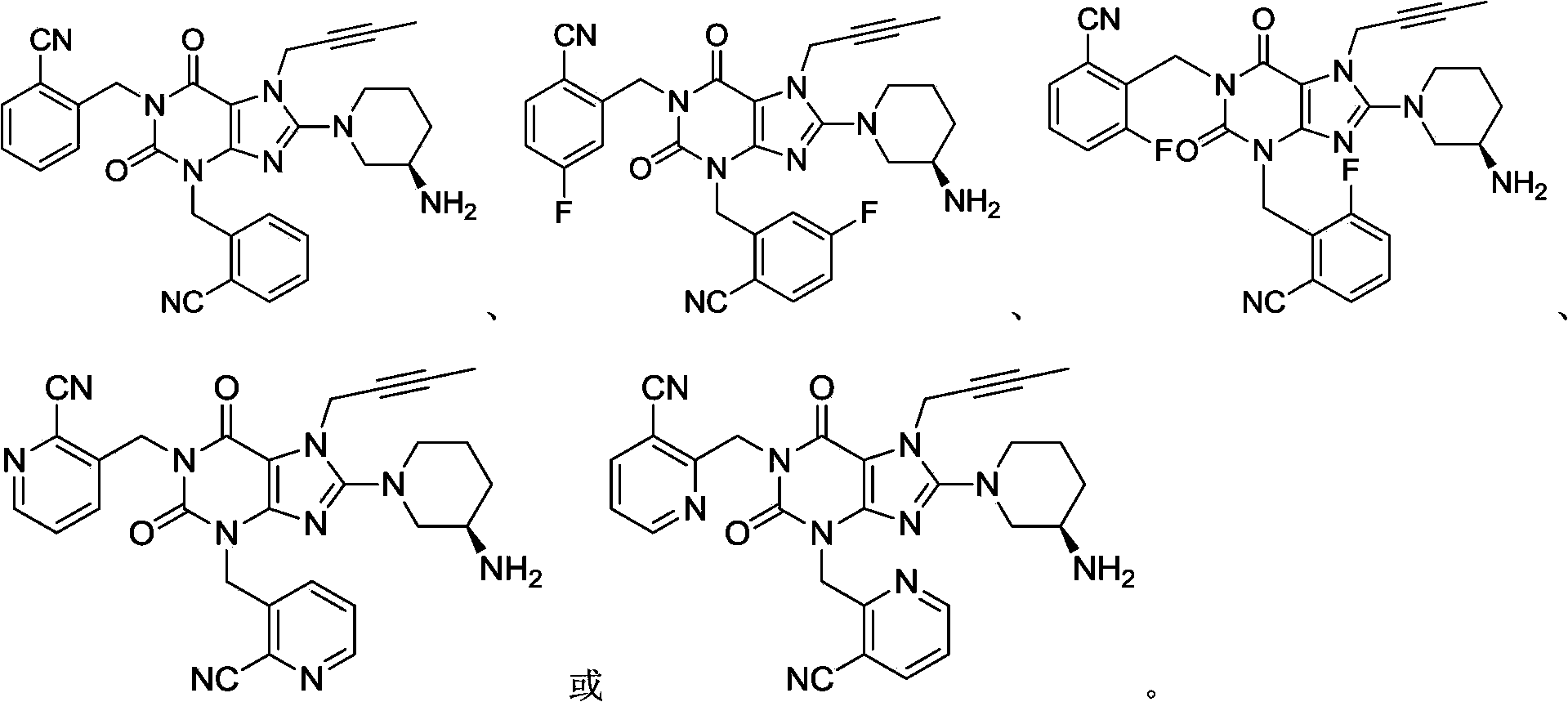
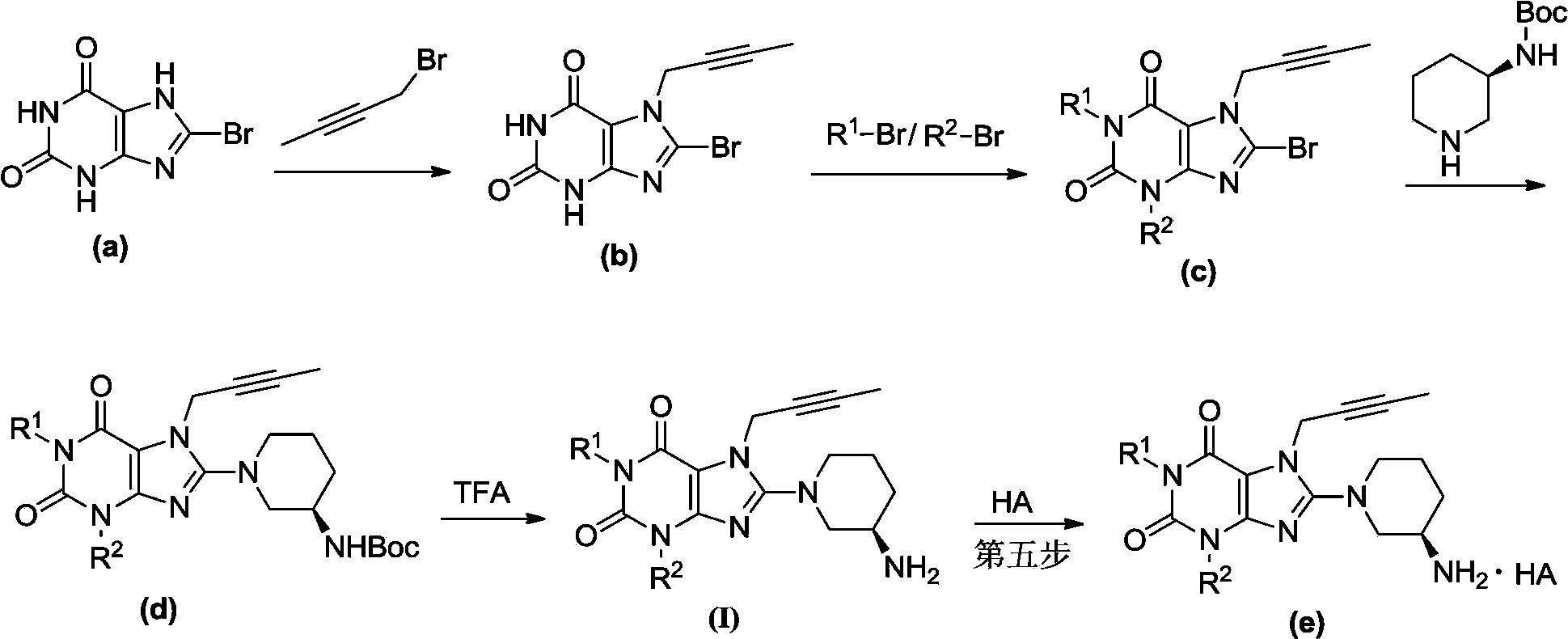
Xanthine derivative
A compound and pharmaceutical technology applied in the field of dipeptidyl peptidase IV inhibitors
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0029] 1-[(benzonitrile-2-yl)methyl]-3-[(benzonitrile-2-yl)methyl]-7-(2-butyn-1-yl)-8-[(R )-3-amino-piperidin-1-yl]-xanthine preparation
[0030]
[0031]
[0032] Preparation of the first step 7-(2-butyn-1-yl)-8-bromo-xanthine
[0033] Using a known method, 8-bromoxanthine (2.31g, 10mmol) was dissolved in N,N-dimethylformamide (20ml), and N,N-diisopropylethylamine (1.42g, 11mmol) was added . 1-Bromo-2-butyne (1.46g, 11mmol), reacted at room temperature for 6h, followed by thin-layer chromatography, which showed that the raw materials were completely consumed. Slowly add 200ml of water to the stirred reaction system, the off-white solid particles are separated out in the solution, after fully stirring for 30min, filter, wash with water, and dry to obtain compound 7-(2-butyn-1-yl)-8-bromo-xanthine 1a ( 2.1 g, yellow solid), yield: 74.2%.
[0034] MS m / z(ESI):283,285[M+1]
[0035] The second step 1-[(benzonitrile-2-yl)methyl]-3-[(benzonitrile-2-yl)methyl]-7-(2-butyn-1-...
Embodiment 2
[0046] 1-[(4-fluoro-benzonitrile-2-yl)methyl]-3-[(4-fluoro-benzonitrile-2-yl)methyl]-7-(2-butyne-1- Preparation of -8-[(R)-3-amino-piperidin-1-yl]-xanthine
[0047]
[0048]
[0049] The first step is the same as the first step in embodiment 1
[0050] The second step 1-[(4-fluoro-benzonitrile-2-yl)methyl]-3-[(4-fluoro-benzonitrile-2-yl)methyl]-7-(2-butyne Preparation of -1-yl)-8-bromo-xanthine
[0051] Using a known method, the compound 7-(2-butyn-1-yl)-8-bromo-xanthine 1a (283mg, 1mmol) was dissolved in N,N-methylformamide (5ml), and potassium hydroxide was added (140mg, 2.5mmol) was added with 2-cyano-5-fluorobenzyl bromide (535mg, 2.5mmol) while stirring, and reacted overnight at room temperature. TLC followed the reaction, showing that the raw materials were completely consumed, and slowly added 50ml of water, a light yellow solid precipitated out, stirred thoroughly for 30 minutes, filtered, washed with water, and dried to obtain compound 1-[(4-fluoro-benzonitri...
Embodiment 3
[0061] 1-[(3-fluoro-benzonitrile-2-yl)methyl]-3-[(3-fluoro-benzonitrile-2-yl)methyl]-7-(2-butyne-1- Preparation of -8-[(R)-3-amino-piperidin-1-yl]-xanthine
[0062]
[0063]
[0064] The first step is the same as the first step in embodiment 1
[0065] The second step 1-[(3-fluoro-benzonitrile-2-yl)methyl]-3-[(3-fluoro-benzonitrile-2-yl)methyl]-7-(2-butyne Preparation of -1-yl)-8-bromo-xanthine
[0066] Using a known method, the compound 7-(2-butyn-1-yl)-8-bromo-xanthine 1a (283mg, 1mmol) was dissolved in N,N-dimethylformamide (5ml), added with hydrogen Potassium (140mg, 2.5mmol) was added with 2-bromomethyl-3-fluorobenzocyanide (535mg, 2.5mmol) while stirring, and reacted overnight at room temperature. The reaction was tracked by thin-layer chromatography, which showed that the raw materials were completely consumed. Slowly add 50ml of water, a light yellow solid precipitates out, stir thoroughly for 30min, filter, wash with water, and dry to obtain the compound 1-[(...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap