Synthetic method of atorvastatin calcium intermediate
A technology of atorvastatin calcium and synthesis method, applied in microorganism-based methods, biochemical equipment and methods, microorganisms and other directions, can solve problems such as long steps, and achieve the effects of shortening steps, high yield and reducing costs
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0016] The present invention will be described in detail below in conjunction with embodiments.
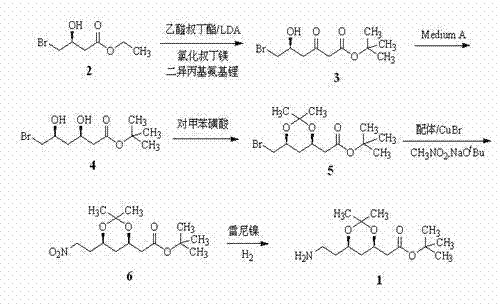
[0017] Step 1) Add THF (50 ml) into a 1 L three-necked flask, and add (3 S )-4-bromo-3-hydroxybutyric acid ethyl ester (purity 97%, 15.7 g, 75 mmol) and tert-butyl acetate (17.4 g, 150 mmol), continue to stir under argon protection at 0-5 ℃, dropwise Add 1 mol / L toluene-THF solution of tert-butylmagnesium chloride (1:2.5, 75 ml), and continue stirring at 5°C for 30 min. Add 1 mol / L lithium diisopropylamide in n-hexane (240 ml) dropwise, and continue stirring at the same temperature for 16 h after the drop. Separately take concentrated hydrochloric acid (60.4 g), water (31.3 g) and ethyl acetate (50 ml), stir and mix, add to the above reaction solution, stir for 10 min, then pour into a 2 L separatory funnel and let stand to separate. layer, the organic layer was washed with water (100 ml × 2) and concentrated under reduced pressure to obtain a reddish-brown oil (5 S )-tert-buty...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More