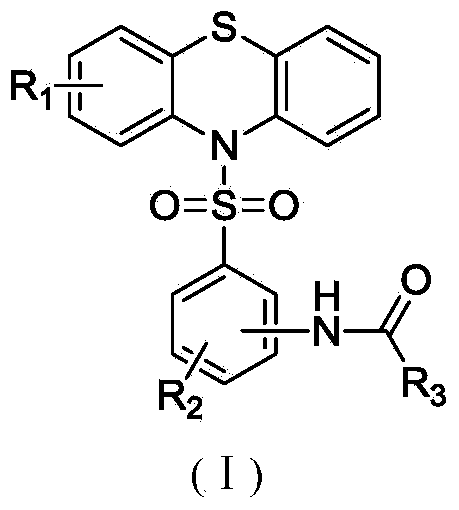
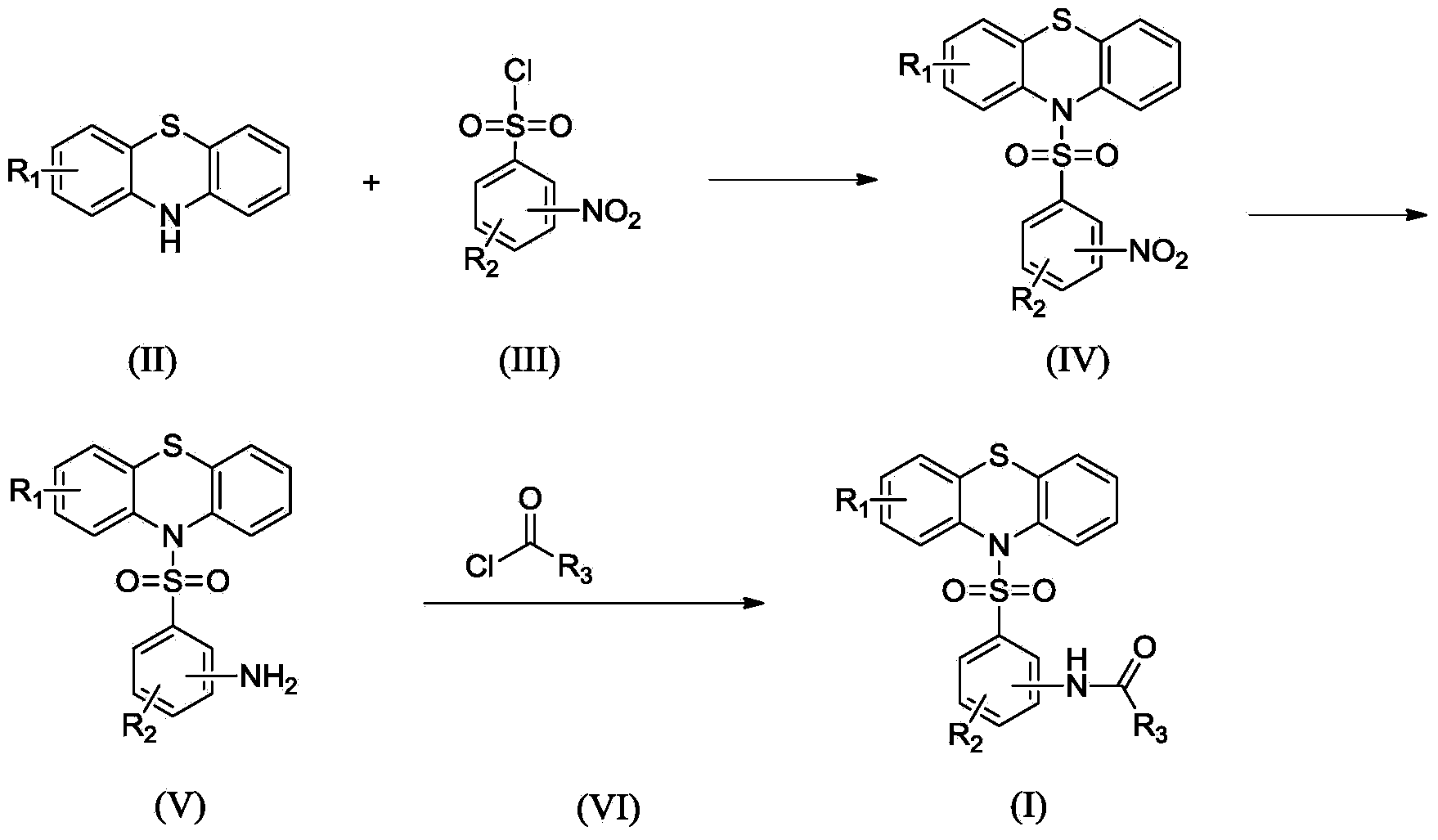
N-substituted benzenesulfonyl phenothiazine compounds as well as preparation method and use thereof
A compound and mono-substituted technology, applied in the field of medicine, can solve the problems of insufficient activity, side effects and physicochemical properties of benzoazepines
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0057] Preparation of IV-1
[0058]
[0059] II-1 (100g, 502mmol) was placed in a 1000mL reaction flask, and CH was added 2 Cl 2 (500mL) stirred to make it dissolve, added triethylamine (76g, 750mmol), stirred under ice-water bath, added intermediate III-1 (121.9g, 550mmol) in batches, kept the temperature and stirred for 1h, TLC detection showed that the reaction was finished ( Developing agent ethyl acetate:petroleum ether=1:3).
[0060] The reaction solution was poured into 500ml of cold water, fully shaken to separate the layers, and the organic layer was separated and washed three times in succession. The organic layer was dried over anhydrous sodium sulfate and left overnight. After filtration, the solvent was evaporated to dryness under reduced pressure to obtain a light yellow solid. The resulting product was recrystallized from ethanol to obtain 173 g of a light yellow solid. The purity is 97.9% (HPLC normalization method), and the yield is 89.6%. ESI-MS ([...
Embodiment 2
[0062] Preparation of IV-2
[0063]
[0064] II-1 (20g, 100mmol) was placed in a 250mL reaction flask, and CHCl was added 3 (100mL) was stirred to dissolve, added pyridine (8.7g, 110mmol), stirred and dissolved at 50°C, added intermediate III-2 (23.3g, 105mmol) in batches, kept stirring at the temperature for 3h, TLC detection showed that the reaction was complete (Developer ethyl acetate:petroleum ether=1:3).
[0065] The reaction solution was poured into 100ml of cold water, fully shaken to separate the layers, and the organic layer was separated and washed three times in succession. The organic layer was dried over anhydrous sodium sulfate and allowed to stand overnight. After filtration, the solvent was evaporated to dryness under reduced pressure to obtain a light yellow solid crude product. The resulting crude product was purified by silica gel column chromatography to obtain 34.8 g of a pale yellow solid. The purity is 98.2% (HPLC normalization method), and the...
Embodiment 3
[0067] Preparation of IV-3
[0068]
[0069] Put II-2 (20g, 86mmol) in a 250mL reaction flask, add pyridine (60mL), stir to dissolve, stir under ice-water bath, add intermediate III-3 (20.3g, 86mmol) in batches, keep the temperature and stir for 4h , TLC detection showed that the reaction was complete (developing agent ethyl acetate:petroleum ether=1:3).
[0070] The reaction solution was poured into 300ml of cold water, stirred, and solids were precipitated. After filtering, the filter cake was washed with water and dried to obtain a yellow crude product. The crude product was recrystallized from ethanol to obtain 34.7 g of a light yellow solid. The purity is 98.4% (HPLC normalization method), and the yield is 93.3%. ESI-MS ([M+H]+): 433.0.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More