Type I clopidogrel hydrogen sulfate salt preparation method
A technology of clopidogrel hydrogen sulfate and clopidogrel free base, which is applied in the field of preparation of type I clopidogrel hydrogen sulfate, and can solve the problems of uncontrollable crystal form purity, inability to prepare, and increased toxic and side effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
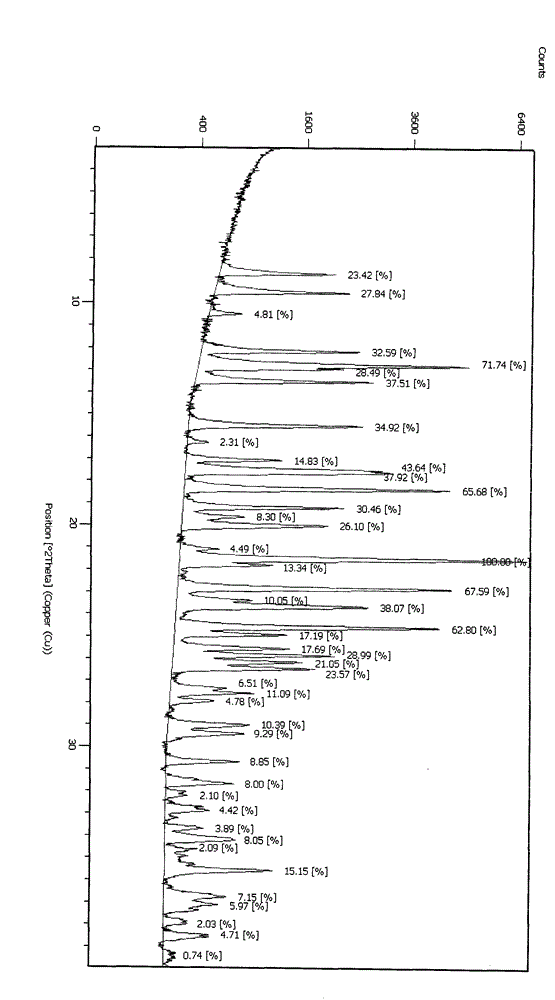
Embodiment 1
[0029] Mix clopidogrel salt with dichloromethane, add water, add ammonia water to the solvent, and stir for 0.5 hours to generate the free base of clopidogrel, measure the pH value of the aqueous phase to be 7 to 8, and let it stand for stratification. The aqueous phase was extracted once more with dichloromethane, the organic phases were combined, and the solvent was recovered to dryness after washing with water; 5 times the amount of a mixed solvent of ethyl acetate and acetone was added to the clopidogrel free base obtained after concentration, and ethyl acetate and acetone The volume ratio of clopidogrel is 15:1, stir to completely dissolve the clopidogrel free base, cool down to -15°C, add 10% of the theoretically generated clopidogrel hydrogen sulfate amount as a seed crystal, according to the moles of sulfuric acid and clopidogrel free base The ratio is 0.9-1.05:1, add the solution of sulfuric acid diluted with organic solvent (diluted by 10 times with sulfuric acid) dro...
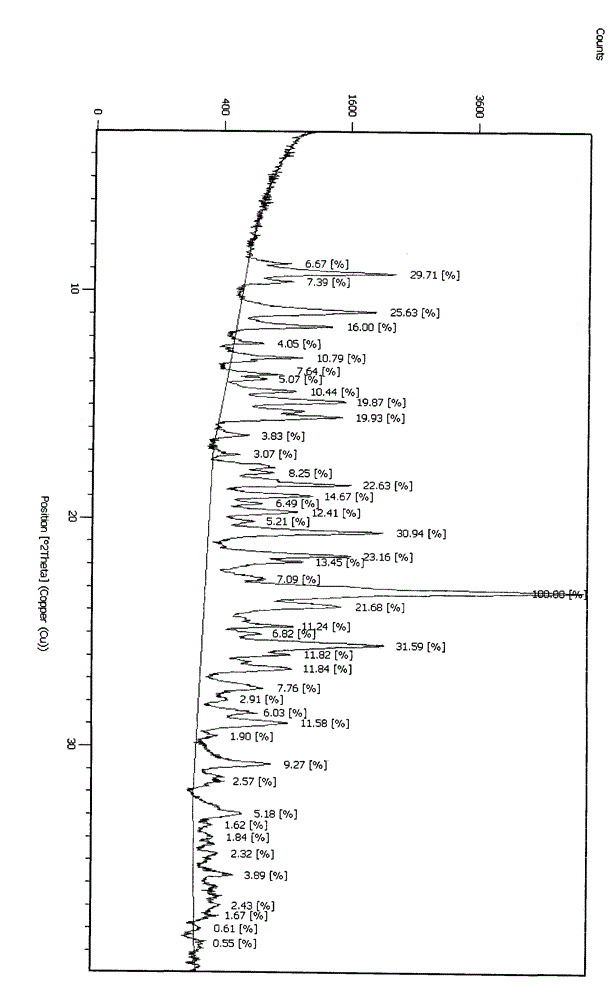
Embodiment 2
[0031] Mix clopidogrel salt with dichloroethane, add water, add sodium carbonate aqueous solution to the solvent, and stir for 1 hour to generate clopidogrel free base, measure the pH value of the aqueous phase to be 7-8 when static Separate the layers, extract the aqueous phase once with an aqueous solution of sodium carbonate, combine the organic phases, and recycle the solvent to dryness after washing with water; add 10 times the amount of a mixed solvent of ethyl acetate and acetone to the clopidogrel free base obtained after concentration, The volume ratio of ethyl acetate to acetone is 5:1, stir to completely dissolve the clopidogrel free base, cool down to 15°C, add 0.5% of the theoretically generated clopidogrel hydrogen sulfate seed crystals, follow the sulfuric acid and clopidogrel The molar ratio of the free base is 0.9~1.05:1. Add the solution of sulfuric acid (diluted 20 times by sulfuric acid) diluted with organic solvent to the solution dropwise. After dropping, ...
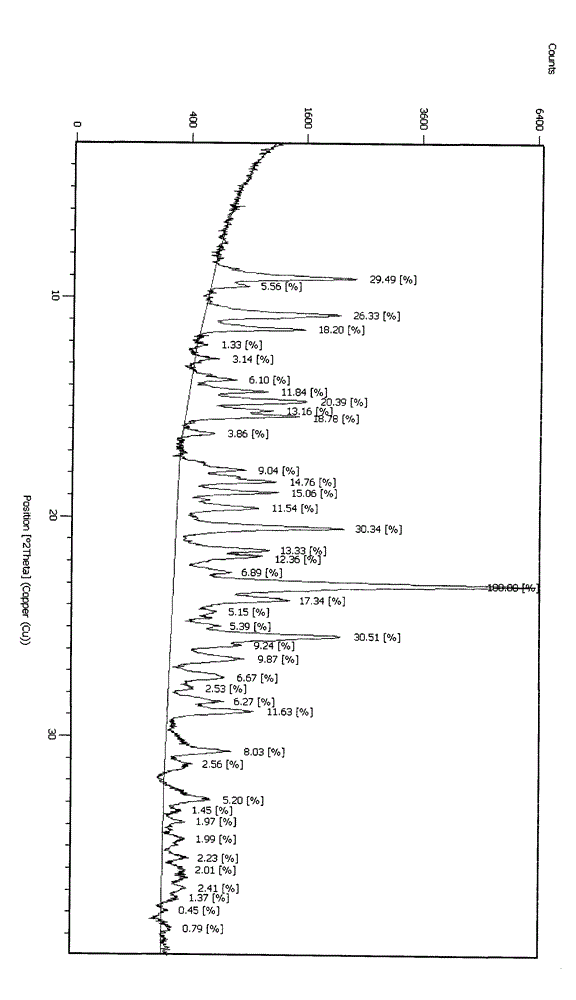
Embodiment 3
[0033] Mix clopidogrel salt with chloroform, add water, add an aqueous solution of potassium carbonate to the solvent, and stir for 0.8 hours to generate the free base of clopidogrel, measure the pH value of the water phase to 7-8 and let it stand Separate the layers, extract the aqueous phase once with chloroform, combine the organic phases, wash with water and recover the solvent to dryness; add 8 times the amount of a mixed solvent of ethyl acetate and acetone to the clopidogrel free base obtained after concentration, ethyl acetate The volume ratio of ester to acetone is 8:1, stir to completely dissolve the clopidogrel free base, cool down to 0°C, add 1% of the theoretically generated clopidogrel bisulfate amount as seed crystals, follow the formula of sulfuric acid and clopidogrel free base The molar ratio of the solution is 0.9~1.05:1, add dropwise the sulfuric acid solution diluted by organic solvent (10 times dilution of sulfuric acid) into the solution, after dropping, ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More