Method for preparing wall breaking decoction pieces with radix astragali
A technology for broken-wall decoction pieces and astragalus, which is applied in the field of preparation of broken-wall decoction pieces of astragalus, can solve the problems such as no treatment method provided for microbial limits, rapid dissolution of unfavorable medicinal components, unsuitable medicinal materials, etc. Moderate tightness, easy drying effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
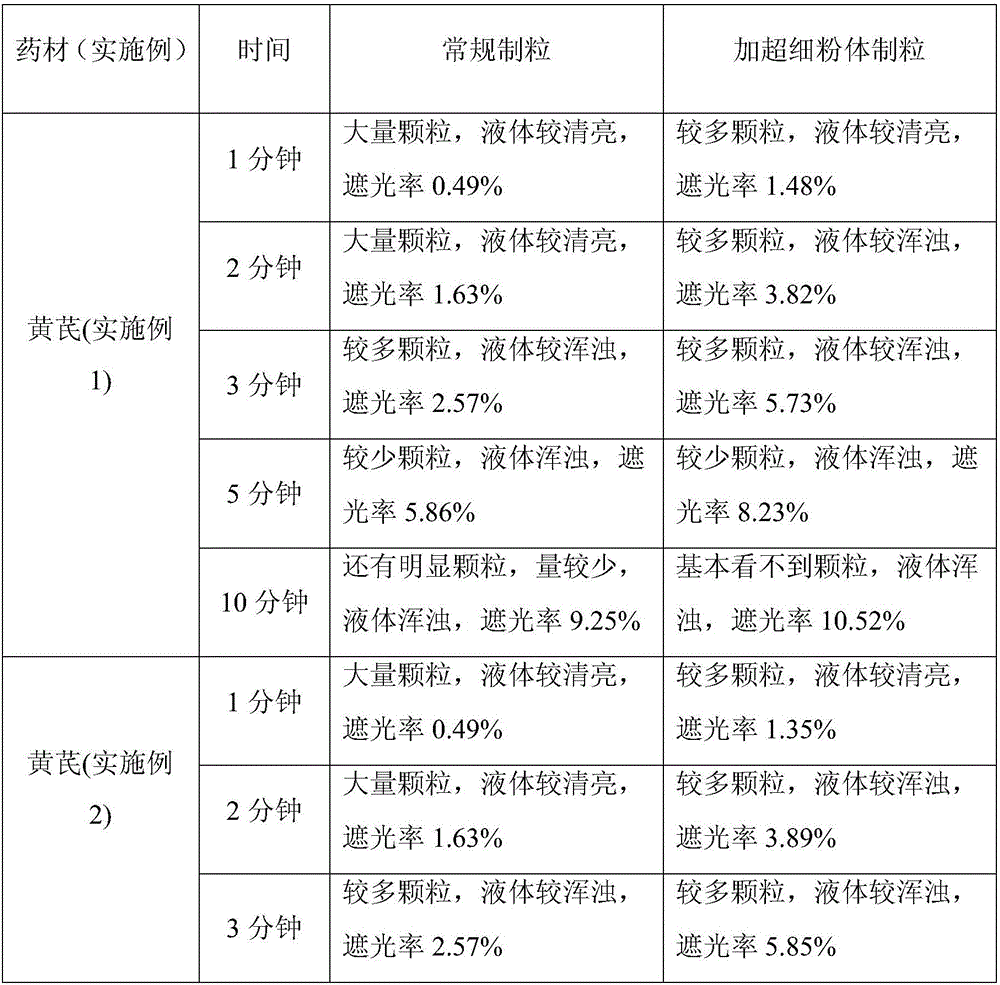
Embodiment 1
[0038] Astragalus Broken Wall Decoction Pieces
[0039] 1) Medicinal material processing: pick and clean / process the Astragalus medicinal material, dry the medicinal material until the water content of the medicinal material is below 10%, and crush the medicinal material into small particles of about 10 mesh with a mechanical crusher of 10 mesh screen; further coarsely crush the medicinal material Reach about 100 mesh;
[0040] 2) Intermittent sterilization: Put the coarse powder of Astragalus membranaceus in a constant temperature and humidity box with a thickness of about 10mm, set the temperature at 65°C (the actual temperature is 60-70°C), and keep it dry and sterilized for 8 hours; The temperature is 25°C and the relative humidity is 75% for 12 hours; then the temperature of the constant temperature and humidity box is adjusted to 35°C and the relative humidity is 88% for 24 hours; the set temperature is changed to 75°C (the actual temperature is 70-80°C), Keep it dry an...
Embodiment 2
[0046] Astragalus Broken Wall Decoction Pieces
[0047] 1) Medicinal material processing: the astragalus medicinal material is sorted, cleaned / processed, and dried until the water content of the medicinal material is below 10%, and the medicinal material is crushed into small particles of about 10 mesh with a mechanical crusher of a 10-mesh screen; further coarsely crushing the medicinal material reaches About 100 mesh;
[0048] 2) Intermittent sterilization: Put the coarse powder of Astragalus membranaceus in a constant temperature and humidity box with a thickness of about 5mm, set the temperature at 70°C (the actual temperature is 65-75°C), and keep it dry and sterilized for 4 hours; The temperature is 20°C and the relative humidity is 70% for 15 hours; then the temperature of the constant temperature and humidity box is adjusted to 33°C and the relative humidity is 83% for 8 hours; change the set temperature to 65°C (the actual temperature is 60~70°C), Keep it dry and ste...
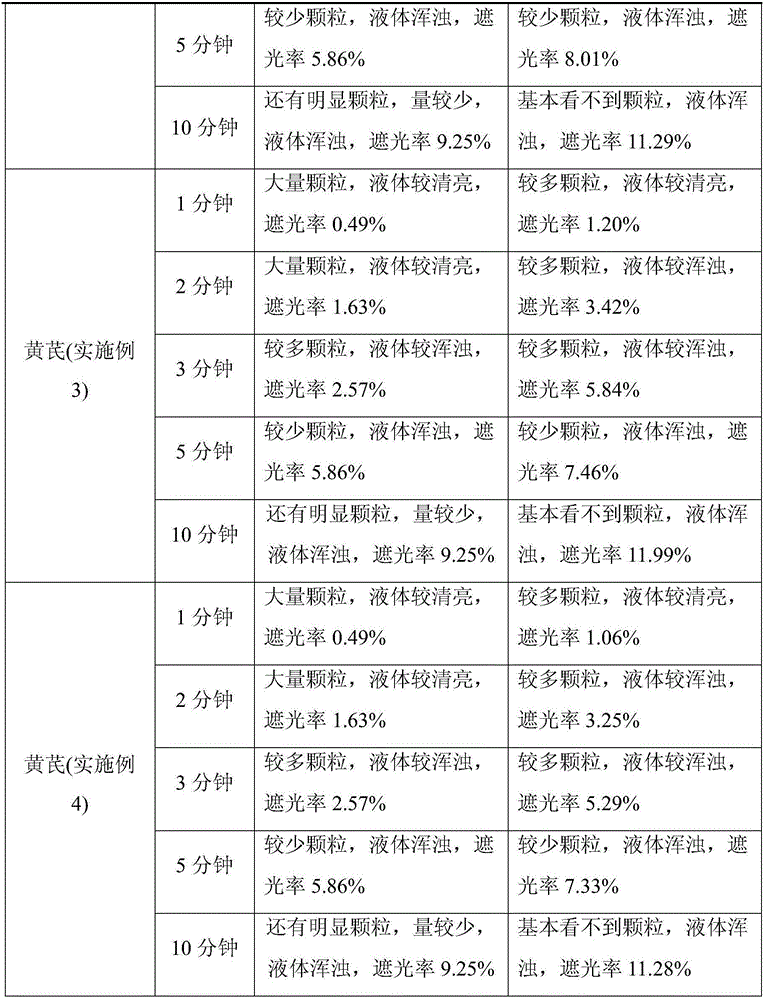
Embodiment 3
[0054] Astragalus Broken Wall Decoction Pieces
[0055] 1) Medicinal material processing: the astragalus medicinal material is sorted, cleaned / processed, and dried until the water content of the medicinal material is below 10%, and the medicinal material is crushed into small particles of about 10 mesh with a mechanical crusher of a 10-mesh screen; further coarsely crushing the medicinal material reaches About 100 mesh;
[0056] 2) Intermittent sterilization: Put the coarse powder of Astragalus membranaceus in a constant temperature and humidity box with a thickness of about 30mm, set the temperature at 75°C (the actual temperature is 70-80°C), and keep it dry and sterilized for 4 hours; The temperature is 25°C and the relative humidity is 75% for 24 hours; then the temperature of the constant temperature and humidity box is adjusted to 40°C and the relative humidity is 80% for 12 hours; Set the temperature at 80°C (the actual temperature is 75-85°C), keep it dry and steriliz...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Thickness | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More