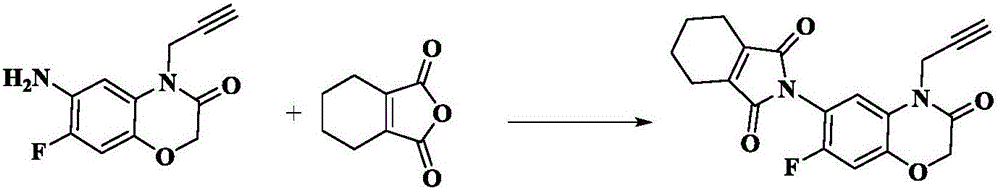
Synthesis method of 7-fluorine-6-amino-4-(2-propargyl)-1,4-benzoxazine-3(4H)-ketone derivative
A synthesis method and a benzoxazine technology, applied in the field of organic synthesis, can solve the problems of high price, low product purity, high price, etc., and achieve the effects of low cost, easily available raw materials, and low price
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0029] A method for synthesizing 7-fluoro-6-amino-4-(2-propargyl)-1,4-benzoxazin-3(4H)-one derivatives, the specific operations are:
[0030] 23.2g of 95% 6-amino-7-fluoro-4-propynyl-1,4-benzoxazin-3(4H)-one, 15.9g of 3,4,5,6-tetrahydrophthalic anhydride, Add 0.6g of acetic acid and 1.96g of potassium acetate into 100mL of toluene solvent, reflux and dehydrate at 110°C for 6h, monitor the reaction progress by HPLC, after the reaction is completed, remove the solvent under reduced pressure, beat the obtained filter cake with ethyl acetate, filter and dry , 32.1 g of diflufenamide was obtained with a purity of 99.5% and a yield of 91%. The product was confirmed by NMR and LC-MS.
Embodiment 2
[0032] A kind of synthetic method of 7-fluoro-6-amino-4-(2-propargyl)-1,4-benzoxazin-3(4H)-one derivative, the operation of this embodiment is the same as that of Example 1 , the only difference is: replace potassium acetate with sodium acetate. After the reaction was completed, 31.8 g of flufenazim was obtained, with a purity of 99.2% and a yield of 90%.
Embodiment 3
[0034] A kind of synthetic method of 7-fluoro-6-amino-4-(2-propargyl)-1,4-benzoxazin-3(4H)-one derivative, the operation of this embodiment is the same as that of Example 1 , the only difference is: Potassium acetate is replaced by potassium carbonate. After the reaction was completed, 31.7 g of flufenflume was obtained, with a purity of 99.5% and a yield of 90%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More