Anti-MUC1 CAR-T cell and its preparation method and application
A technology of cells and lymphocytes, applied in the field of gene-modified cells and tumor treatment, to achieve the effect of high-efficiency tumor-killing activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0043] Preparation of Anti MUC1 CAR-T cells of the present invention
[0044] 1. Construction of lentiviral expression vector
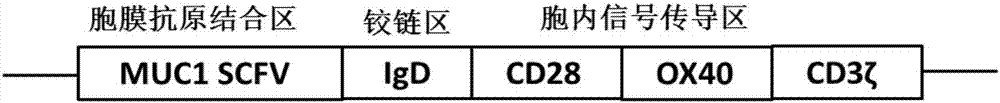
[0045] Through conventional genetic engineering methods, the SCFV-IgD-CD28-OX40-CD3ζ fusion protein gene sequence was synthesized, and the SCFV-IgD-CD28-OX40-CD3ζ fusion protein gene sequence was cloned into a lentiviral expression vector to obtain SCFV-IgD-CD28 –OX40–CD3ζ lentiviral expression vector pGreen puro-CAR. Wherein, each sequence of the SCFV-IgD-CD28-OX40-CD3ζ fusion protein is as described above.
[0046] 2. Lentiviral packaging
[0047] 1) Culture 293T cells in 1640 medium with a mass fraction of 10 wt% fetal bovine serum (fetal bovine serum, FBS for short);
[0048] 2) 293T cells were treated with 3x 10 5 / cm 2 Transfer the density to a culture dish with a diameter of 15cm and cultivate it for 20h to ensure that the cell confluence is 80-90% during transfection;
[0049] Replace the medium with serum-free 1640 medium and set aside;...
Embodiment 2
[0063] The in vitro anti-tumor effect of the Anti MUC1 CAR-T cells prepared in Example 1 was evaluated, including the following steps:
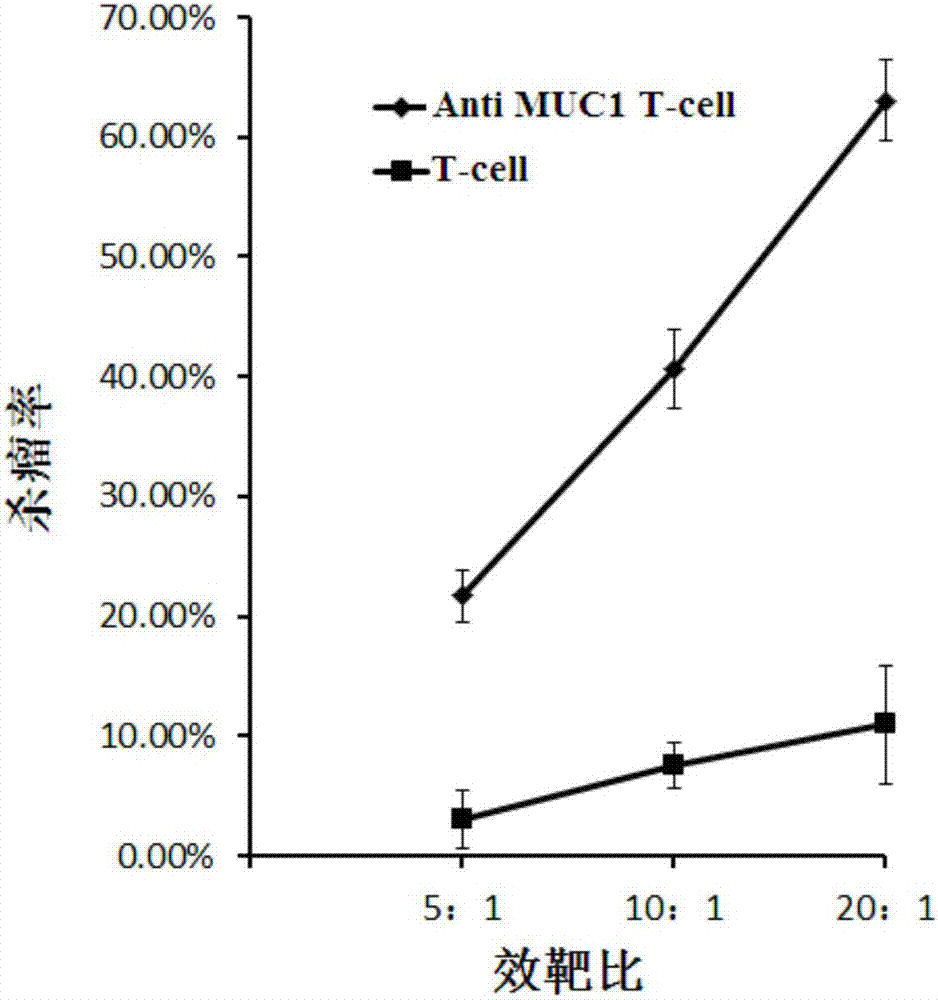
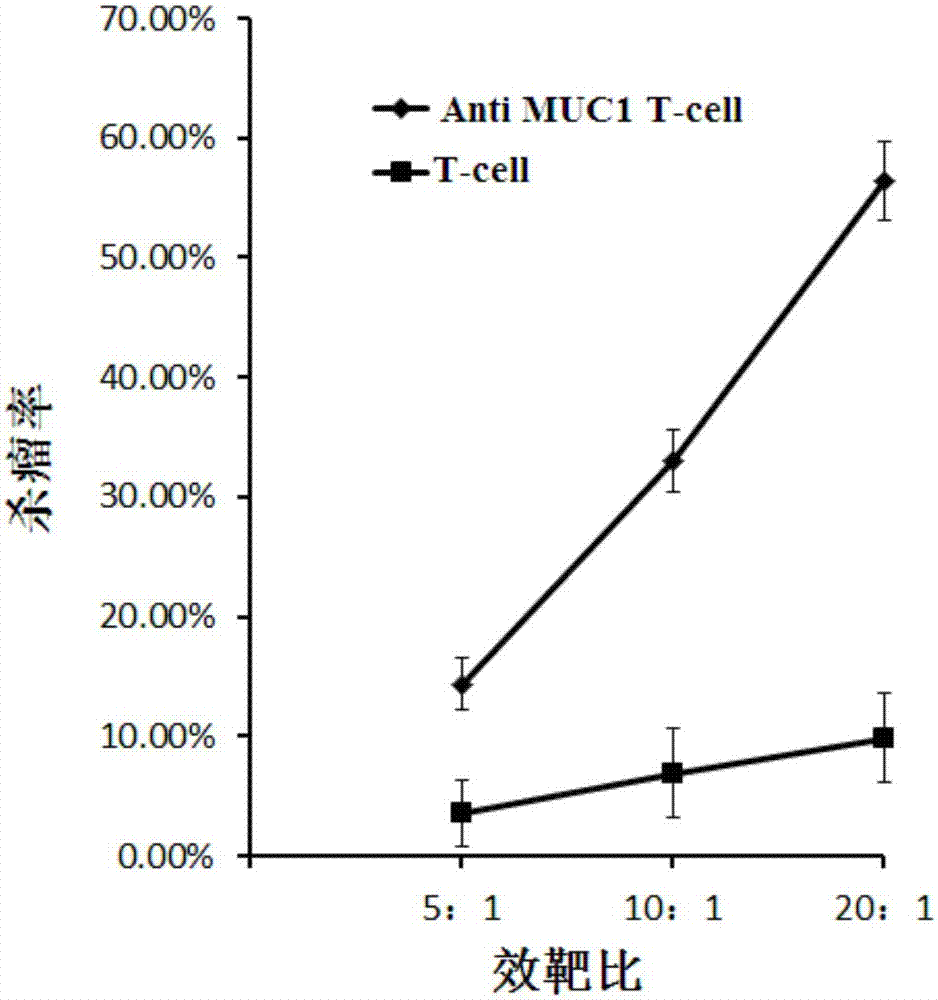
[0064] With breast cancer cell line MCF-7 and pancreatic cancer cell line BxPC-3 stably expressing MUC1 as target cells, T cells infected with lentiviral vector pGreen puro-CAR (i.e., Anti MUC1 CAR-T cells prepared in Example 1 cells) and uninfected T cells to make effector cells, target cells according to the density of 1x 10 5 cells / ml inoculated in 96-well plate, 100 μl per well, added effector cells to target cells according to 5:1, 10:1, 20:1 effect-to-target ratio, placed in 5% CO 2 , cultured in a 37°C incubator for 4 hours, using WST-1 to detect cell viability, and calculate the killing efficiency.
[0065] figure 2 It is a schematic diagram of the killing effect of Anti MUC1 CAR-T cells and uninfected T cells on the target cell MCF-7 (breast cancer) according to the embodiment of the present invention; image 3 It is a schematic ...
Embodiment 3
[0068] The anti-tumor effect of the Anti MUC1 CAR-T cells prepared in Example 1 was evaluated in vivo, including the following steps:
[0069] Take 15 6-week-old female nude mice and inject 5x 10 subcutaneously in the right armpit 6 MCF-7 (breast cancer) cells, when the tumor grows to 60mm 3 Size, tumor model was randomly divided into 3 groups: control group, Anti MUC1 CAR-T cell group, T cell group; control group was injected with normal saline 200ul / time, twice a week; Anti MUC1 CAR-T cell group was injected with tail vein Anti MUC1 CAR-T cells 1×10 7 T cells / time, 2 times a week; T cells in the T cell group were injected into the tail vein 1×10 7 Each time, twice a week; the survival status of the mice within 100 days was counted, and the survival rate curve was made. Figure 4 It is a schematic diagram of the survival period of mice treated with breast cancer (MCF-7) xenografted tumor model mice by Anti MUC1 CAR-T cells of the embodiment of the present invention, contro...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More