Application of fidaxomicin in the preparation of medicines for treating related diseases and/or symptoms caused by dengue virus infection
A technology of dengue virus and fidaxomycin, applied in the field of medicine, can solve problems such as lack of effective drugs, and achieve the effects of high safety, strong binding ability, and high inhibition of dengue virus activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0019] Example 1: Inhibition of dengue virus (DENV) activity by fidaxomicin at the cellular level
[0020] Virus strain: Dengue virus DENV2 (NGC), cell line: A549
[0021] Detection method:
[0022] Fidaxomycin antiviral half effective dose (50% Effective Concentration, EC 50 ): DMSO, 6, 9, 12, 18, 24 μM Fidaxomycin saturated cells 1 hour in advance, 2 hours after virus infection, changed to virus-free medium containing corresponding concentration of drugs for 48 hours; collected cell supernatant, detected by plaque assay Plaque formation inhibition rate of different doses of fidaxomicin group relative to solvent group (DMSO) after virus infection.
[0023] Inhibition rate (%) = (1-the number of virus plaques in the administration group / the number of plaques in the solvent control group) 100%, calculated using the Forcast formula of EXCEL2013, when the inhibition rate is equal to 50%, the corresponding fidaxomicin Concentration, as EC 50 . The average value was obtained f...
Embodiment 2
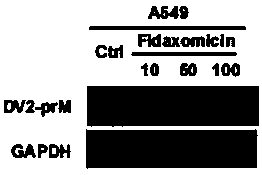
[0027] Example 2 The inhibitory activity of Fidaxomycin to DENV2-prM protein in A549 cell line
[0028] Virus strain: Dengue virus DENV2 (NGC)
[0029] Cell line: A549
[0030] Detection method:
[0031] DMSO, 10, 50, 100 μM Fidaxomycin saturated the cells 1 hour in advance, and 2 hours after the virus infection, the virus-free medium containing the corresponding concentration of drugs was maintained for 48 hours; the cell pellet was collected, and Western blot electrophoresis was used to detect DENV2 in the cells under different treatments The relative expression of (NGC)-prM protein, GAPDH was used as internal reference protein. Such as figure 1 As shown, Fidaxomycin can effectively inhibit the membrane protein of DENV--DENV2-prM protein at a concentration of 10-50 μM. inhibition.
Embodiment 3
[0032] Example 3 Surface Plasmon Resonance Detection of Fidaxomycin and DENV2-NS5 Protein Affinity
[0033] This method adopts GE's Biacore T100 instrument and CM5 chip for experiments. First, the purified DENV2 (NGC)-NS5 protein is amino-coupled to the CM5 chip, and then flows through different concentrations of fidaxomicin. The instrument detects the adsorption on The mass change of the substance on the surface of the chip is calculated to calculate the affinity rate of the compound (K a ) vs. dissociation rate (K d ). Affinity = dissociation equilibrium constant (K D ), this value describes the binding strength between a small molecule and a protein molecule. The biological meaning is that when a small molecule binds to a protein 1:1, let 50% of the small molecule saturate the concentration of the protein molecule. The smaller the value, the stronger the binding. Usually, the affinity between small molecules and proteins calculated by this method is between 1 and 1000 μM...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More