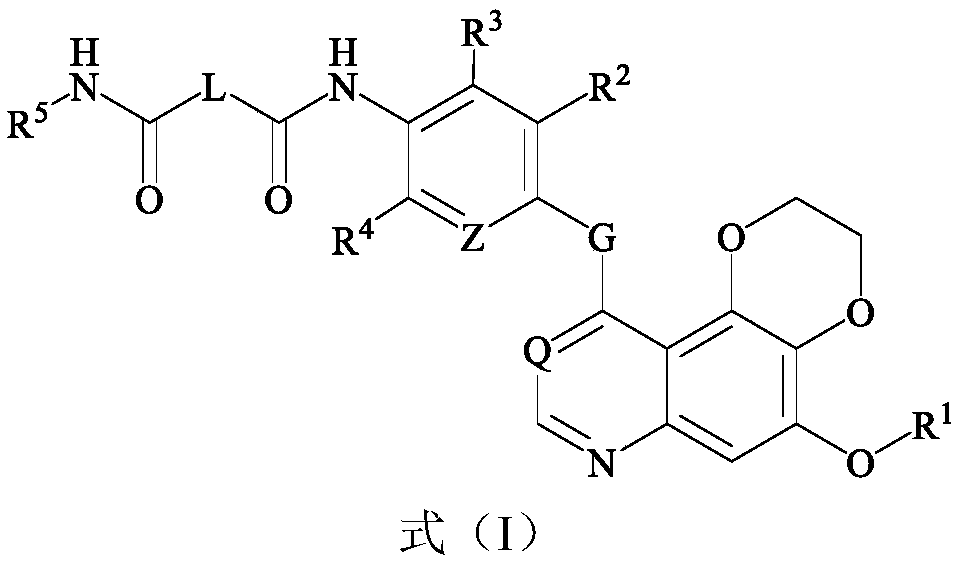
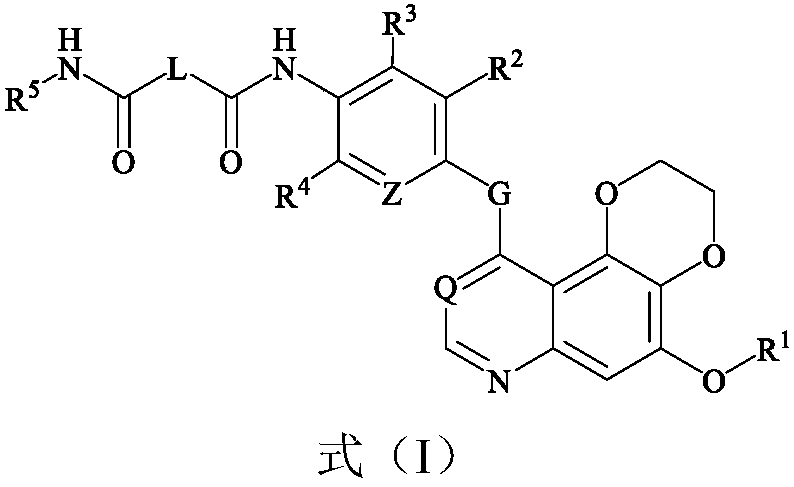
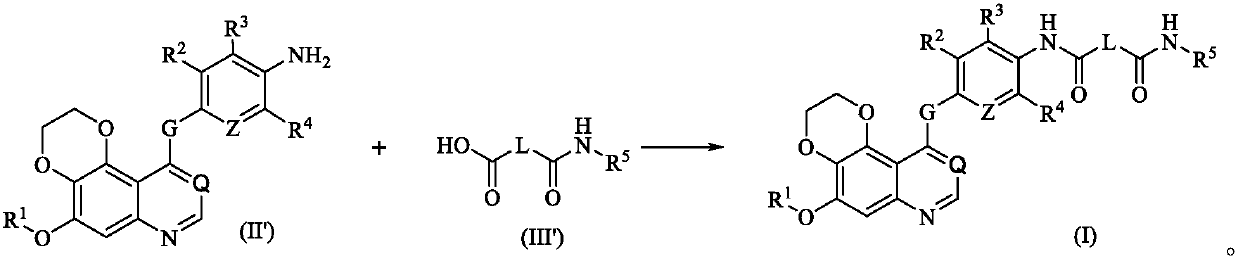
Dioxane quinoline compound and preparation method and application thereof
A compound and hydrate technology, applied in the field of dioxane quinoline compounds, can solve problems such as side effects, limited access to drugs, drug resistance, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0123] Example 1. N-(4-fluorophenyl)-N-(4-((5-methoxy-2,3-dihydro-[1,4]dioxalo[2,3-f] Preparation of -quinolin-10-yl)oxy)phenyl)cyclopropane-1,1-dicarboxamide
[0124]
[0125] Step 1): Mix 10-chloro-5-methoxy-2,3-dihydro-[1,4]dioxano[2,3-f]quinoline (251 mg, 1 mmol) with p-nitrophenol (139mg, 1mmol) was placed in a reaction flask, added chlorobenzene, heated to reflux and stirred until the reaction was complete. Suction filtration after cooling, the obtained solid was washed with potassium carbonate aqueous solution to give light yellow solid (5-methoxy-10-(4-nitrophenoxy)-2,3-dihydro-[1,4]diox Alkano[2,3-f]quinoline) 250 mg, yield 71%. MS:355[M+H] + .
[0126] Step 2): Put the product (250mg, 0.7mmol) obtained in step 1) into a reaction flask, add methanol and Raney nickel (250mg), stir under hydrogen atmosphere until the reaction is complete, filter and concentrate to obtain an off-white solid product (4-((5-methoxy-2,3-dihydro-[1,4]dioxano[2,3-f]quinolin-10-yl)oxy)...
Embodiment 2
[0128] Example 2. N-(3-fluoro-4-((5-methoxy-2,3-dihydro-[1,4]dioxano[2,3-f]-quinoline-10- Preparation of yl)oxy)phenyl)-N-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide
[0129]
[0130] Step 1) 10-(2-fluoro-4-nitrophenoxy)-5-methoxy-2,3-dihydro-[1,4]dioxano[2,3-f]-quinone Preparation of morphine
[0131] Referring to the operation of step 1) of Example 1, the same molar equivalent of 2-fluoro-4 nitrophenol can be used instead of p-nitrophenol. 1 HNMR (400MHz, DMSO-d 6 )δ8.67(d,J=5.0Hz,1H),8.44–8.27(m,1H),8.13–7.93(m,1H),7.19(s,1H),7.07(d,J=4.9Hz,1H ),6.98(t,J=8.7Hz,1H),4.31–4.18(m,2H),4.16–4.06(m,2H),3.95(s,3H); MS:373[M+H] + .
[0132] Step 2) 3-fluoro-4-((5-methoxy-2,3-dihydro-[1,4]dioxano[2,3-f]quinolin-10-yl)oxy) Preparation of aniline
[0133] Reference Example 1 step 2) operation, with the same molar equivalent of 10-(2-fluoro-4-nitrophenoxy)-5-methoxy-2,3-dihydro-[1,4]diox Alkano[2,3-f]quinoline instead of 5-methoxy-10-(4-nitrophenoxy)-2,3-dihydro-[1,4]dioxa...
Embodiment 3
[0136] Example 3. N-(2-fluoro-4-((5-methoxy-2,3-dihydro-[1,4]dioxalo[2,3-f]quinolin-10-yl Preparation of )oxy)phenyl)-N-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide
[0137] Step 1) 10-(3-fluoro-4-nitrophenoxy)-5-methoxy-2,3-dihydro-[1,4]dioxano[2,3-f]quinoline preparation of
[0138] Referring to the operation of step 1) of Example 1, the same molar equivalent of 3-fluoro-4 nitrophenol can be used instead of p-nitrophenol. MS:373[M+H] + .
[0139] Step 2) 2-fluoro-4-((5-methoxy-2,3-dihydro-[1,4]dioxano[2,3-f]quinolin-10-yl)oxy) Preparation of aniline
[0140] Reference Example 1 step 2) operation, with the same molar equivalent of 10-(3-fluoro-4-nitrophenoxy)-5-methoxy-2,3-dihydro-[1,4]dioxin Alkano[2,3-f]quinoline instead of 5-methoxy-10-(4-nitrophenoxy)-2,3-dihydro-[1,4]dioxano[2,3 -f] quinoline. 1 HNMR(400MHz,DMSO-d6)δ8.38(d,J=5.2Hz,1H),7.04(s,1H),6.98–6.91(m,1H),6.89–6.79(m,1H),6.78–6.67 (m,1H),6.37(d,J=5.2Hz,1H),5.14(s,2H),4.43–4.30(m,4H),3.91(s,3H); MS:343[M+...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More