A derivative of 3,4-dithiohydroxyhexyl methacrylate and its preparation method and application
A technology of dithiohydroxyhexyl ester and methacrylic acid, which is applied in the field of methacrylate-1-hexanol) and its preparation, can solve problems such as inconvenient operation and complex technical requirements for PDMAEMA modification, and achieve improved transfection effect of effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
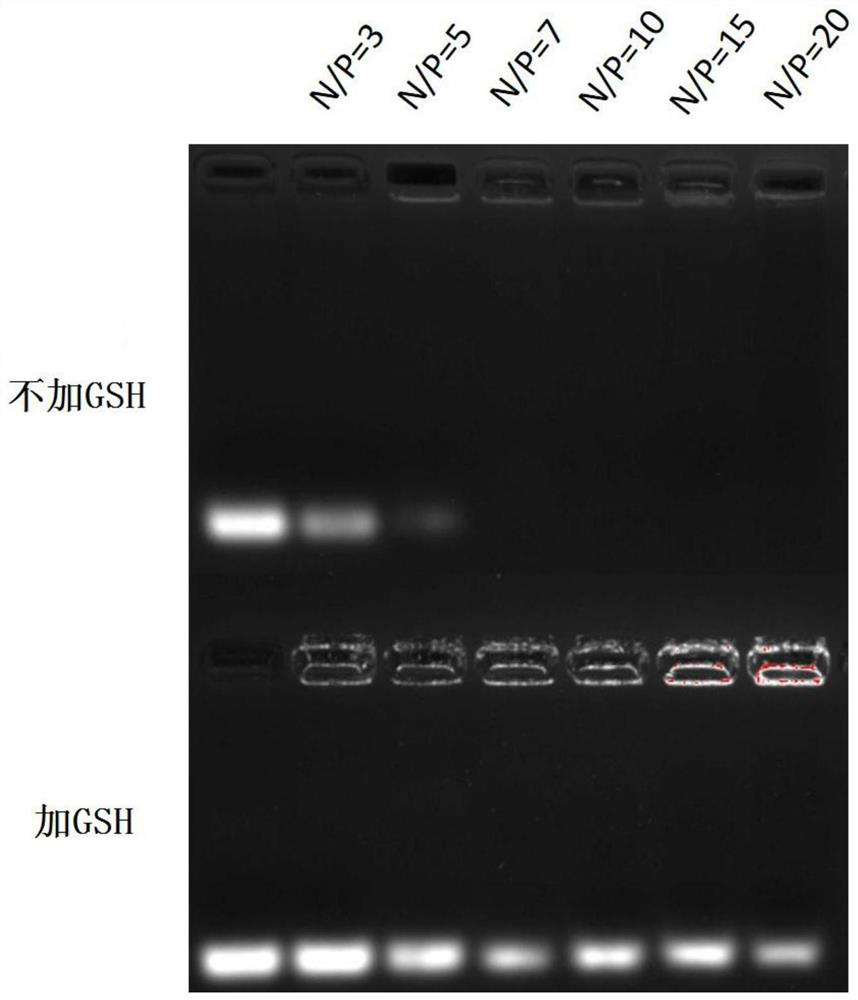
Examples
Embodiment 1
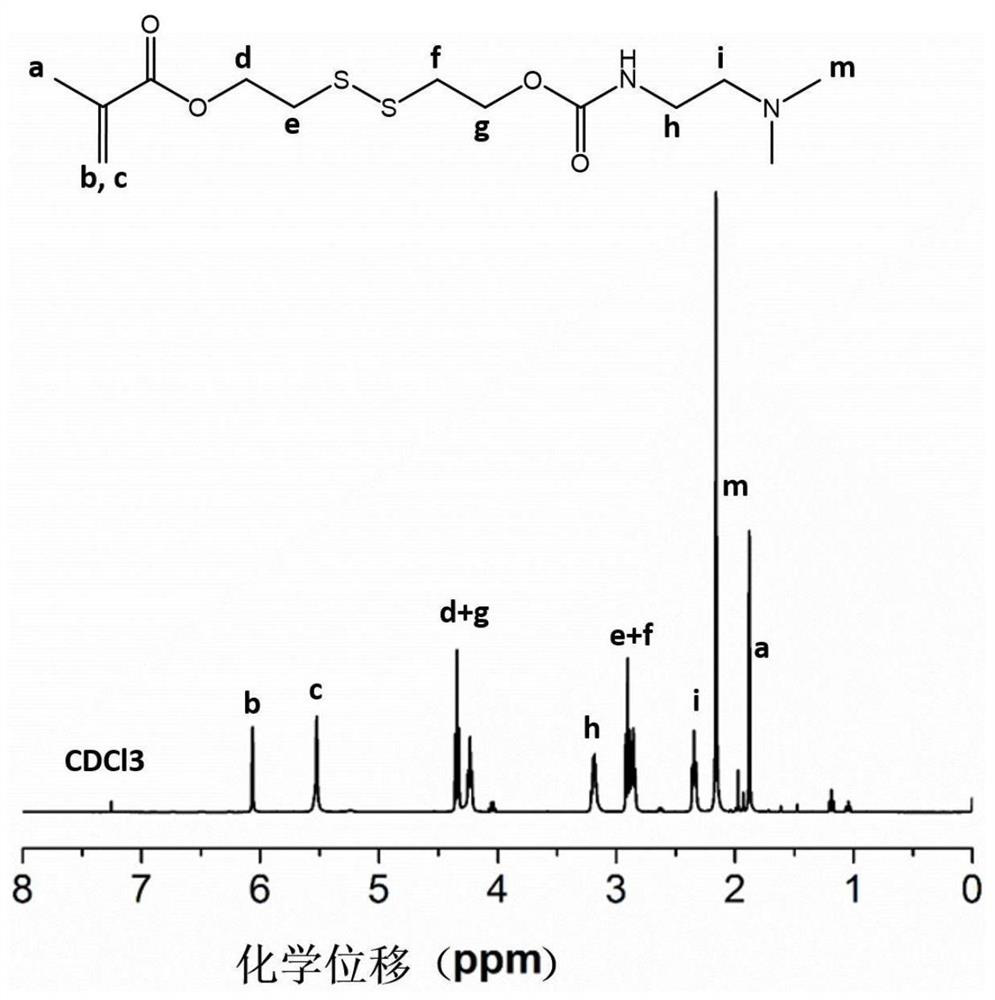
[0024] Add 30mmol of 3,4-dithiohydroxyhexyl methacrylate, 30mmol of benzyl p-nitrochloroformate and 30mmol of triethylamine into the reactor, dissolve them in tetrahydrofuran, react at room temperature for 12h, and terminate the reaction. Filter with NaHCO 3 solution and saturated NaCl solution, and after rotary evaporation, a light yellow oily liquid crude product was obtained, which was purified by silica gel column chromatography to obtain an intermediate product 24mmol methacrylic acid (3,4-dithio-6-(p-nitrobenzylcarbonic acid Ester)-1-hexanol) ester;
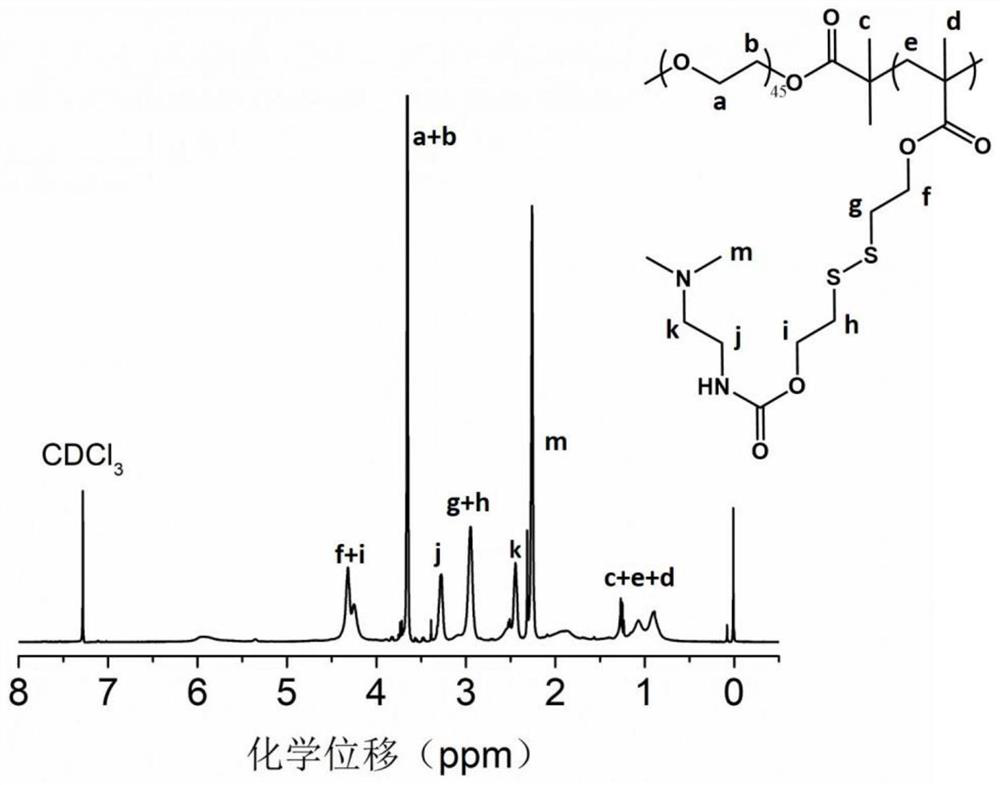
[0025] Add 20mmol methacrylic acid (3,4-dithio-6-(p-nitrobenzyl carbonate)-1-hexanol) ester and 20mmol N,N-dimethylaminoethylenediamine respectively in the reactor, Dissolve with tetrahydrofuran, react at room temperature for 24h, terminate the reaction, and use NaHCO 3 solution and saturated NaCl solution, and after rotary evaporation, a yellow oily liquid crude product was obtained, which was purified by silica gel chro...
Embodiment 2
[0027] Add 20mmol of 3,4-dithiohydroxyhexyl methacrylate, 30mmol of benzyl p-nitrochloroformate and 30mmol of triethylamine into the reactor, dissolve them in tetrahydrofuran, react at room temperature for 6h, and terminate the reaction. Filter with NaHCO 3 solution and saturated NaCl solution, and after rotary evaporation, the light yellow oily liquid crude product was obtained, and the intermediate product 18mmol methacrylic acid (3,4-dithio-6-(p-nitrobenzylcarbonic acid) was purified by silica gel column chromatography Ester)-1-hexanol) ester;
[0028] Add 15mmol methacrylic acid (3,4-dithio-6-(p-nitrobenzyl carbonate)-1-hexanol) ester and 20mmol N,N-dimethylaminoethylenediamine respectively in the reactor, Dissolve in tetrahydrofuran, react at room temperature for 6h, terminate the reaction, and use NaHCO 3 solution and saturated NaCl solution, and after rotary evaporation, a yellow oily liquid crude product was obtained, which was purified by silica gel column chromatog...
Embodiment 3
[0030]Add 15mmol of 3,4-dithiohydroxyhexyl methacrylate, 20mmol of benzyl p-nitrochloroformate and 30mmol of triethylamine into the reactor, dissolve them in 150mL of tetrahydrofuran, react at room temperature for 12h, and terminate the reaction , filtered, with NaHCO 3 solution and saturated NaCl solution, and after rotary evaporation, the crude product of light yellow oily liquid was obtained, and the intermediate product 14mmol methacrylic acid (3,4-dithio-6-(p-nitrobenzylcarbonic acid) was purified by silica gel column chromatography Ester)-1-hexanol) ester;
[0031] Add 10mmol methacrylic acid (3,4-dithio-6-(p-nitrobenzyl carbonate)-1-hexanol) ester and 20mmol N,N-dimethylaminoethylenediamine respectively in the reactor, Dissolve with 100mL tetrahydrofuran, react at room temperature for 12h, terminate the reaction, and use NaHCO 3 solution and saturated NaCl solution, and after rotary evaporation, a yellow oily liquid crude product was obtained, which was purified by si...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More