Pharmaceutical composition for treating epilepsy
A composition and drug technology, applied in the field of pharmaceutical compositions for the treatment of epilepsy, can solve problems such as weight loss, influence on cognition, etc., and achieve the effects of improving symptoms, promoting awakening, and good curative effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0021] Embodiment 1, the preparation of medicine of the present invention
[0022] Recipe: Bupleurum bupleurum 10g, calamus 14g, Uncaria 21g, keel 21g, oyster 21g, peony root 21g, windproof 7g, fried silkworm 10g, earthworm 10g, licorice 3g.
[0023] Preparation method: take the above-mentioned herbs, add water to decoct, decoct twice in total, add 12 times v / w, ml / g of raw medicinal materials for decoction each time, and decoct for 2 hours, combine the two decoction liquids to obtain the product.
Embodiment 2
[0024] Embodiment 2, the preparation of medicine of the present invention
[0025] Recipe: 20g Bupleurum bupleurum, 26g Shichangpu, 39g Uncaria, 39g keel, 39g oyster, 39g white peony, 13g Fangfeng, 20g fried silkworm, 20g earthworm, 7g licorice.
[0026] Preparation method: take the above-mentioned herbs, add water to decoct, decoct 3 times in total, add 6 times the amount of raw medicinal materials v / w, ml / g each time and decoct for 1 hour, combine the two decoction liquids to obtain the product.
Embodiment 3
[0027] Embodiment 3, the preparation of medicine of the present invention
[0028] Recipe: 15g Bupleurum bupleurum, 20g Shichangpu, 20g Uncaria, 30g keel, 30g oyster, 20g white peony, 10g Fangfeng, 15g fried silkworm, 15g earthworm, 5g licorice.
[0029] Preparation method: take the above-mentioned herbs, add water to decoct, decoct twice in total, each time add 8 times the amount of raw medicinal materials v / w, ml / g to decoct for 1.5 hours, combine the two decoction liquids to obtain the product.
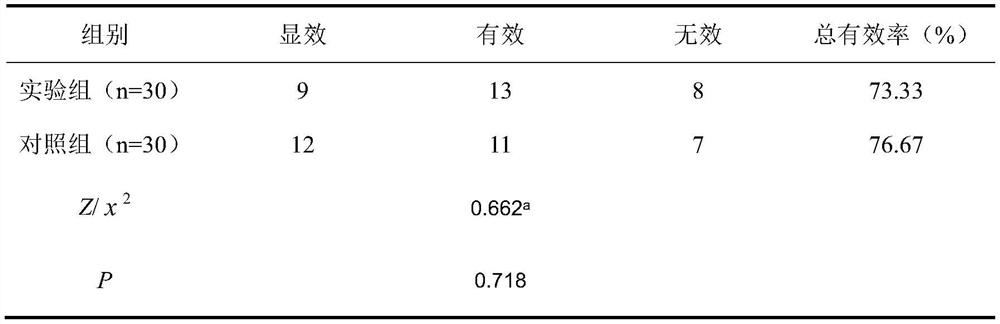
[0030] The beneficial effects of the present invention are illustrated below through test examples.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More