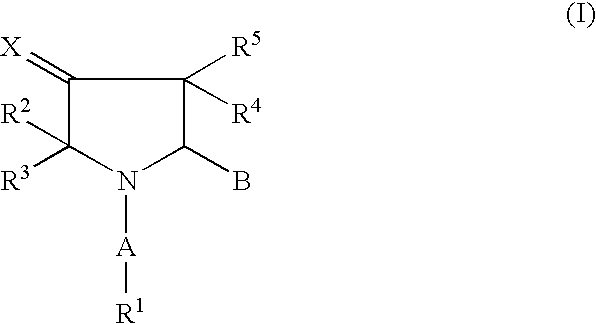
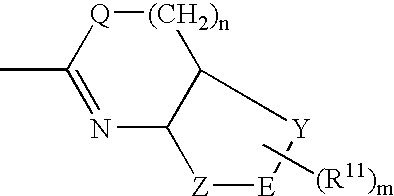
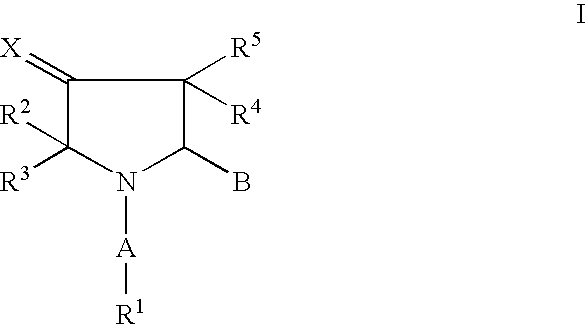
Pharmaceutically active pyrrolidine derivatives
a technology of pyrrolidine and derivatives, applied in the field of pyrrolidine derivatives, can solve the problems of significant problems in the management of preterm labor, undesired pregnancies, and perinatal morbidity and mortality
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
General Procedure for the Saponification of the Olefin-Type Proline Methyl Esters, Such as Intermediates 3-6
[0372]A solution of sodium hydroxide (4.5 g, 112 mmol) in water (70 ml) was added to the relevant proline olefin methyl ester (66 mmol) in 3:1 dioxane:water (500 ml) and the reaction stirred for 3 h. The reaction mixture was then washed with diethyl ether (2×50 ml), and the aqueous phase acidified to pH 2 (0.1N HCl) and extracted into ethyl acetate. The ethyl acetate layer was then dried over magnesium sulfate, filtered and the solvent was then removed in vacuo to give the desired product in near quantitative yields as an oil which was used without further purification.
example 2
General Protocol for the Solution-Phase Synthesis of Oximether Pyrrolidine Derivatives of General Formula Ia (Scheme 1)
Method A: e.g. (2S,4EZ)-1-([1,1′-biphenyl]-4-ylcarbonyl)-N-(2-methoxyethyl)-4-(methoxyimino)-2-pyrrolidinecarboxamide
a) Protocol for the Formation of the Amide Bond
[0373]A solution was made containing the central building block, e.g. (2S,4EZ)-1-(tert-butoxycarbonyl)-4-(methoxyimino)-2-pyrrolidinecarboxylic acid (Intermediate 7) (1.5 g, 5.8 mmol), an amine or an amine salt, e.g. 2-methoxy-ethylamine (0.51 ml, 5.81 mmol) and DMAP (780 mg, 5.8 mmol) in DCM (30 ml). At 0° C., EDC (1.1 g, 5.8 mmol) was slowly added portion-wise. The reaction was slowly allowed to reach room temperature and was stirred overnight. The DCM was evaporated and the crude purified by column chromatography using EtOAc (100%) to collect the desired product, e.g. tert-butyl (2S,4EZ)-2-{[(2-meth-oxyethyl)amino]carbonyl}-4-(methoxyimino)-1-pyrrolidinecarboxylate (1.5 g, 80%) as a colourless oil.
[037...
example 3
(2S,4EZ)-1-([1,1′-biphenyl]-4-ylcarbonyl)-N-[2-(diethylamino)ethyl]-4-{[(4-methoxybenzyl)oxy]imino}-2-pyrrolidinecarboxamide
[0385]Following the general method as outlined in, Example 2, starting from (2S,4EZ)-1-(tert-butoxycarbonyl)-4-{[(4-methoxybenzyl)oxy]imino}-2-pyrrolidinecarboxylic acid, [1,1′-biphenyl]-4-carboxylic acid, and N1,N1-diethyl-1,2-ethanediamine the title compound was obtained after column chromatography as an off-white solid as a mixture of E / Z-isomers.
[0386]1H NMR (400 MHz, CDCl3); 1.05-1.15 (m, 6H), 2.7-2.8 (m, 1H), 2.9-3.2 (m, 6H), 3.4 (m, 1H), 3.6 (s, 3H), 4.0-4.1 (m, 1H), 4.3-4.4 (m, 1H), 3.75 (m, 1H), 3.8 (m, 2H), 6.65 (m, 2H), 7.0-7.1 (m, 2H), 7.2-7.3 (m, 3H), 7.35-7.45 (m, 6H), 8.8 (sibr, 0.5H). M+(APCI+); 543.
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentrations | aaaaa | aaaaa |
| enantiomeric excess | aaaaa | aaaaa |
| enantiomeric excess | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More