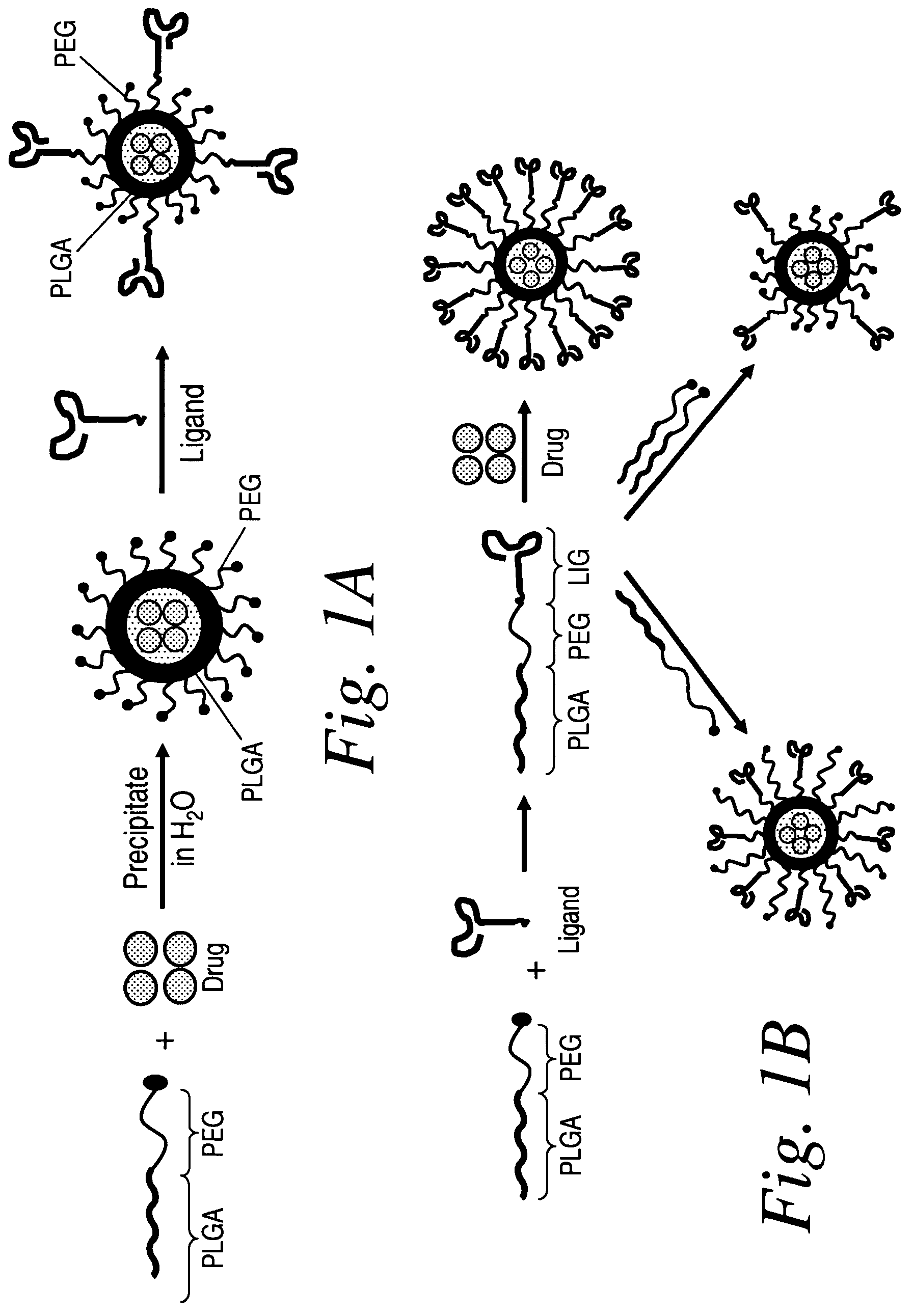
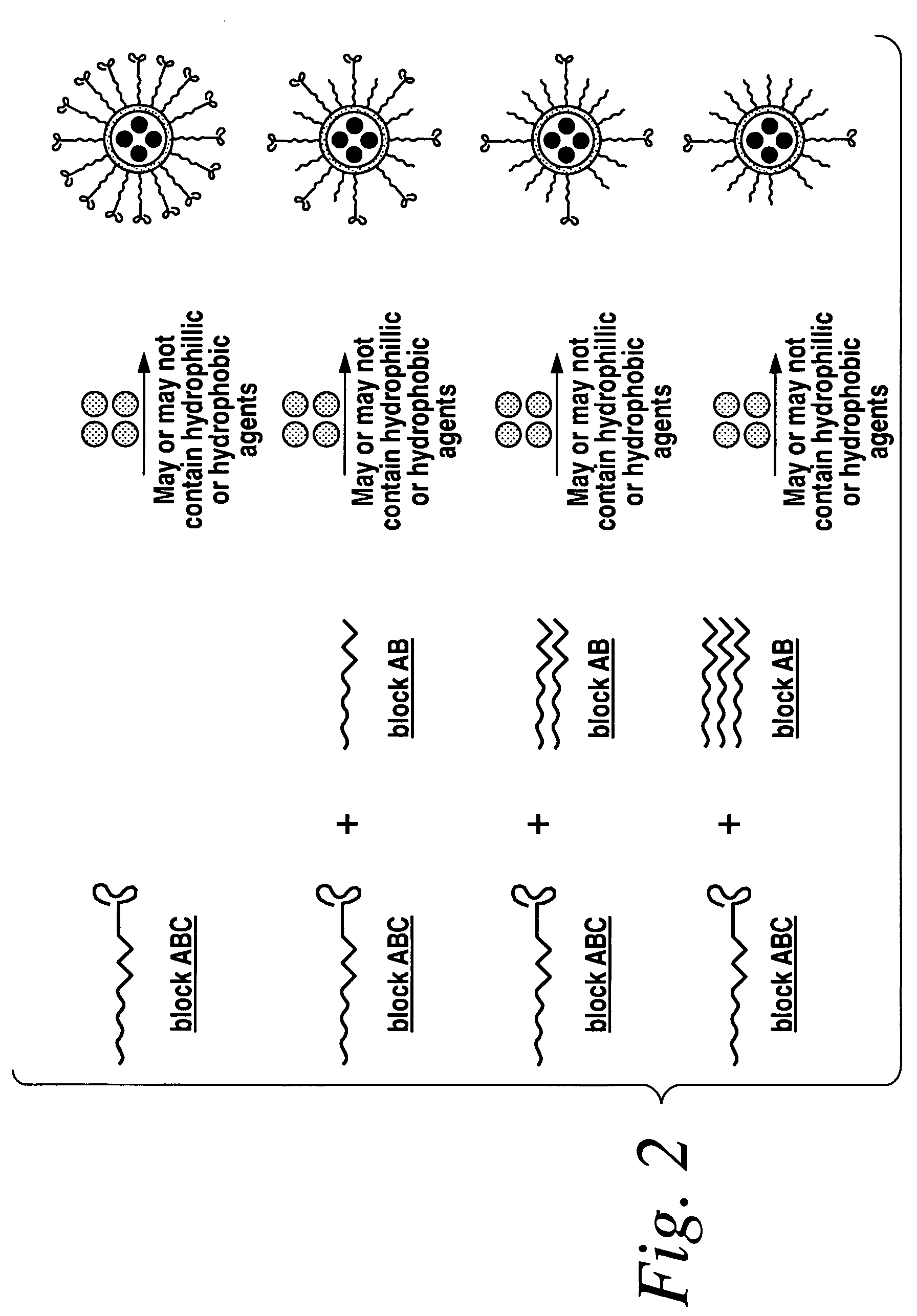
Poly(amino acid) targeting moieties
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Amphiphilic Nanoparticle with Aptamer
[0238]In one embodiment, the A10 RNA aptamer which binds to the Prostate Specific Membrane Antigen (PSMA) on the surface of prostate cancer cells is conjugated to DSPE (1,2-Distearoyl-sn-glycero-3-phosphoethanolamine)-PEG-COOH using EDC / NHS chemistry with a conjugate concentration of 0.7 mg / mL. 0.21 mg of this DSPE-PEG-aptamer bioconjugate is mixed with 0.07 mg lecithin in 2 mL aqueous solution containing 4% ethanol. 1 mg poly(D,L-lactic-co-glycolic acid) (PLGA, Mw=100 kD) is dissolved in 1 mL tetrahydrofuran (THF) solvent, to which 5% docetaxel of the mass of PLGA is added. This PLGA solution is then mixed with the aqueous solution of lecithin / DSPE-PEG-Aptamer. These mixtures are vortexed for 3 minutes, followed by stirring for 2 hours. In order to remove all organic solvents, these mixtures are then dialyzed for another 4 hours against PBS buffer. This procedure would yield nanoparticles targeting to prostate cancer cells expressing PSMA antige...
example 2
Amphiphilic Nanoparticle with CREKA
[0242]The peptide CREKA is conjugated to DSPE-PEG-Maleimide before formulating nanoparticles using the protocol of Example 1. This peptide will target the delivery and uptake of the nanoparticles to extracellular basement membranes which are exposed under the leaky endothelial layer covering atherosclerotic plaques.
example 3
Amphiphilic Nanoparticle with AXYLZZLN
[0243]The peptide AXYLZZLN, or conservative variants or peptidomimetics thereof, wherein X and Z are variable amino acids, can be conjugated to DSPE-PEG-Maleimide before formulating nanoparticles using the protocol of Example 1. This peptide will target the delivery and uptake of the nanoparticles to extracellular basement membranes which are exposed under the leaky endothelial layer covering atherosclerotic plaques.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Molecular weight | aaaaa | aaaaa |
| Ratio | aaaaa | aaaaa |
| Therapeutic | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More