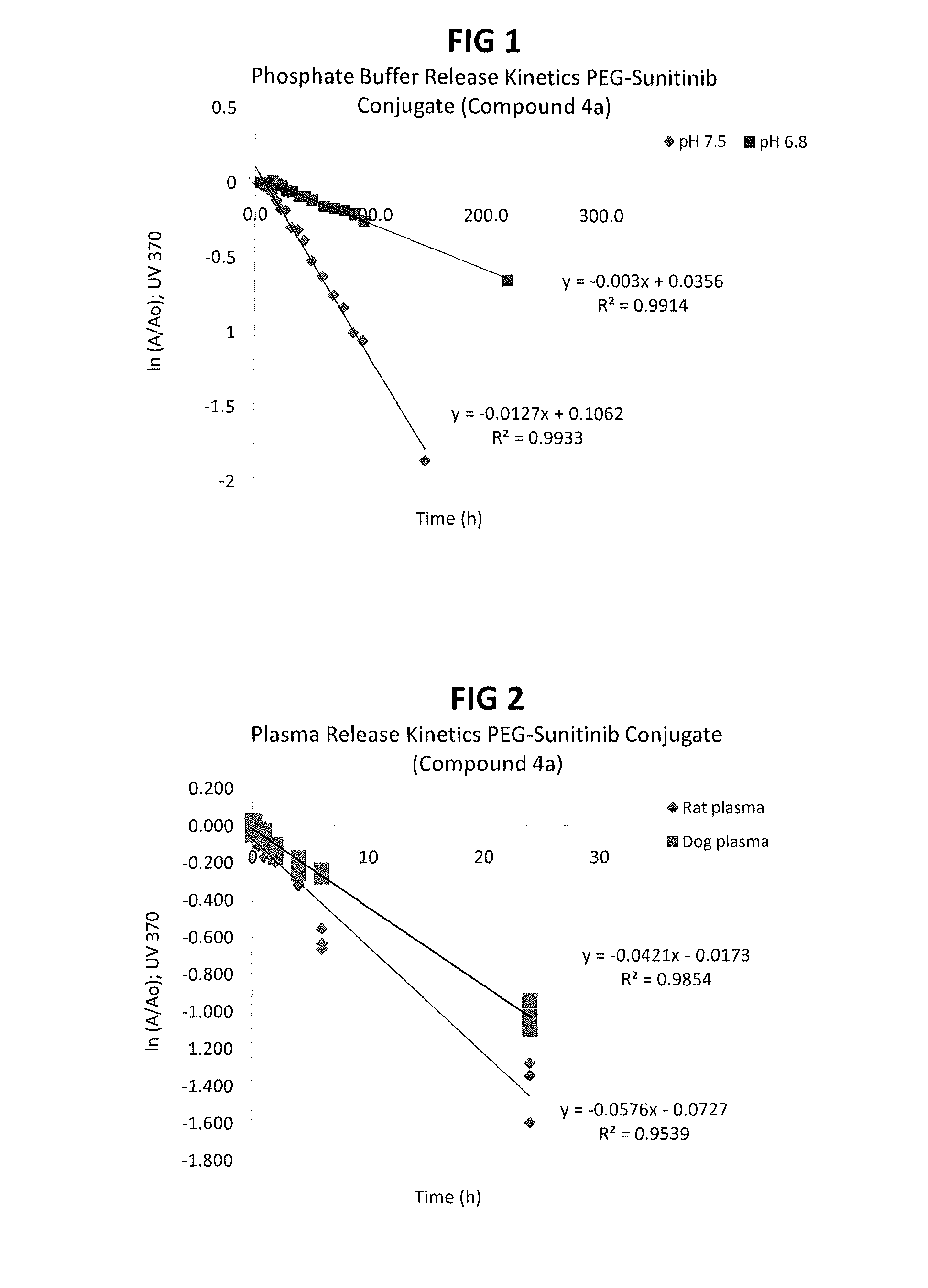
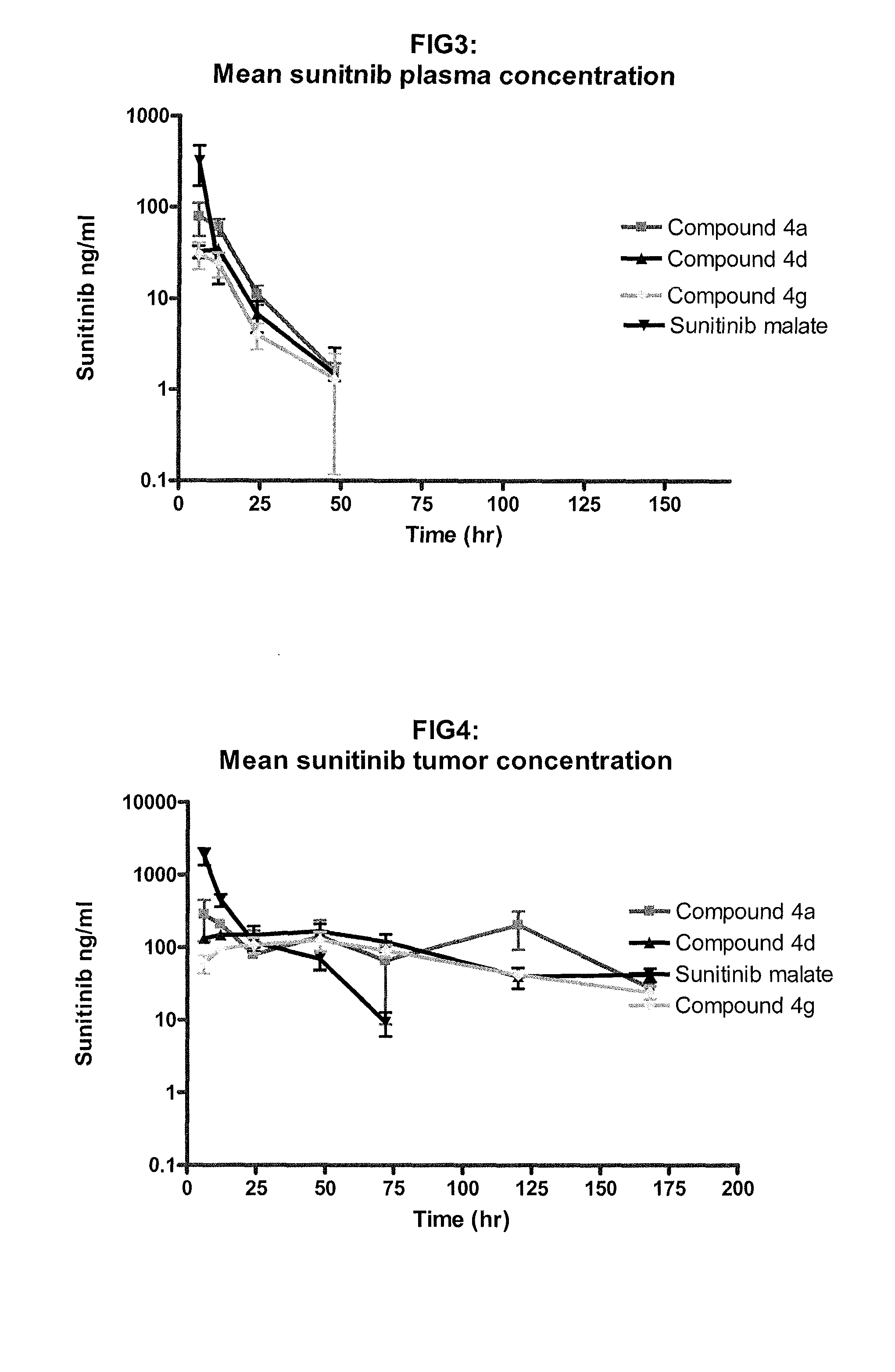
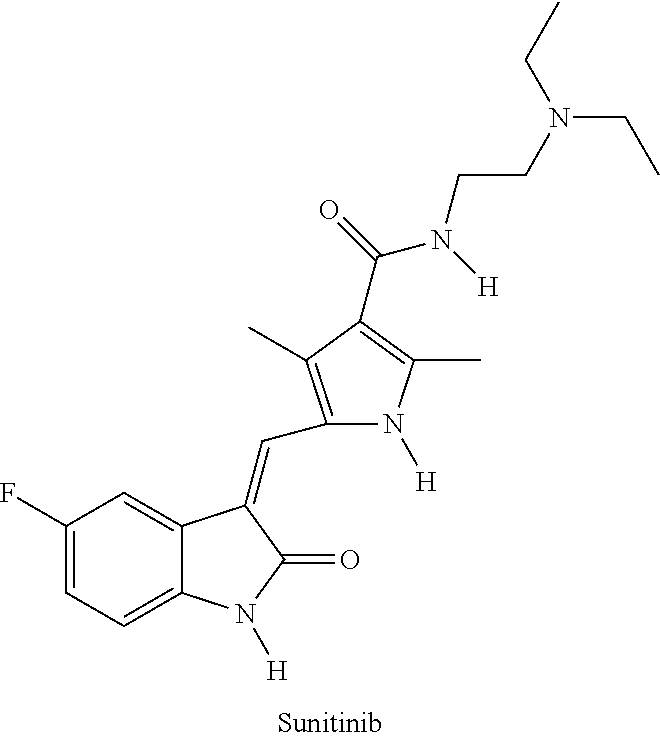
Polymer-sunitinib conjugates
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Synthesis of Amino-Diethyleneglycol Linked PEG-Sunitinib Conjugates
[0205]This example references one or more of the following compounds.
Synthesis of (Z)-2-(diethylamino)ethyl(5-((5-fluoro-2-oxoindolin-3-ylidene)methyl)-2,4-dimethyl-1H-pyrrole-3-carbonyl)carbamic chloride (Compound 1)
[0206]In a 500 mL round-bottomed flask was suspended sunitinib (2.0 g, 5.1 mmol) in THF (200 mL). To this yellow suspension was added triethylamine (11.8 mL, 84 mmol). The suspension was heated in an oil bath with stirring at 60° C. for several minutes to give an orange solution. The solution was cooled for several minutes before transfer to the triphosgene reaction.
[0207](Caution: To prevent release of toxic phosgene gas from either the reaction apparatus or rotary evaporator, the equipment setups were sparged through a sodium hydroxide scrub solution via an over pressure or exhaust port.) In a separate 1 L round-bottomed flask was added triphosgene (1.6 g, 5.4 mmol) in THF (40 mL) to give a colorless s...
example 2
Synthesis of Amino-Propanol Linked PEG-Sunitinib Conjugates
[0222]
Synthesis of (Z)-(tert-butyl 3-hydroxypropylcarbamate) 2-(diethylamino)ethyl(5-((5-fluoro-2-oxoindolin-3-ylidene)methyl)-2,4-dimethyl-1H-pyrrole-3-carbonyl)carbamate (Compound 1b)
[0223]Synthesis was conducted as described in Method A substituting tert-butyl 3-hydroxypropylcarbamate (Compound b) (0.83 mL, 4.8 mmol) to give a orange suspension. Product precipitation in phosphate buffer and extraction were omitted. Purified product yield was 41 mg of yellow powder. HPLC analysis was on a C18 silica column applying an acetonitrile gradient with 0.1% TFA; retention times observed were sunitinib 3.7 minutes and product 9.1 minutes with 99% purity at 370 nm. Analysis by LC-MS ([C31H43FN5O6]+ expected M+H=600.32. found M+H=600.3). 1H-NHR (d6-DMSO): δ (ppm) 0.9 (6H, bm, CH3); 1.3 (9H, s, CH3); 1.5 (2H, m, CH2); 2.3 (3H, s, CH3); 2.4 (3H, s, CH3); 2.6 (2H, bm, CH2); 2.8 (2H, m, CH2); 3.8 (2H, bs, CH2); 4.0 (2H, t, CH2); 6.8 (1H,...
example 3
Synthesis of Hydroxyethylpiperazine Linked PEG-Sunitinib Conjugates
[0227]
Synthesis of (Z)-(tert-butyl 4-(2-hydroxyethyl)piperazine-1-carboxylate) 2-(diethylamino)ethyl(5-((5-fluoro-2-oxoindolin-3-ylidene)methyl)-2,4-dimethyl-1H-pyrrole-3-carbonyl)carbamate (Compound 1c)
[0228]Synthesis was conducted as described in Method A substituting tert-butyl 4-(2-hydroxyethyl)piperazine-1-carboxylate (Compound c) (3.8 g, 17 mmol) in anhydrous tetrahydrofuran (12 mL) and anhydrous acetonitrile (8 mL) to give a orange suspension. Purified product yield was 0.2 g of yellow powder. HPLC analysis was on a C18 silica column applying an acetonitrile gradient with 0.1% TFA; retention times observed were sunitinib 3.4 minutes and product 4.7 minutes with 99% purity at 370 nm. Analysis by LC-MS ([C34H48FN6O6]+ expected M+H=655.36. found M+H=655.3). 1H-NHR (CDCl3): δ (ppm) 1.0 (6H, t, CH3); 1.4 (9H, s, CH3); 2.3 (4H, m, CH2); 2.4 (3H, s, CH3); 2.5 (3H, s, CH3); 2.6 (4H, m, CH2); 2.8 (2H, m, CH2); 3.3 (4H,...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Mass | aaaaa | aaaaa |
| Mass | aaaaa | aaaaa |
| Solubility (mass) | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More