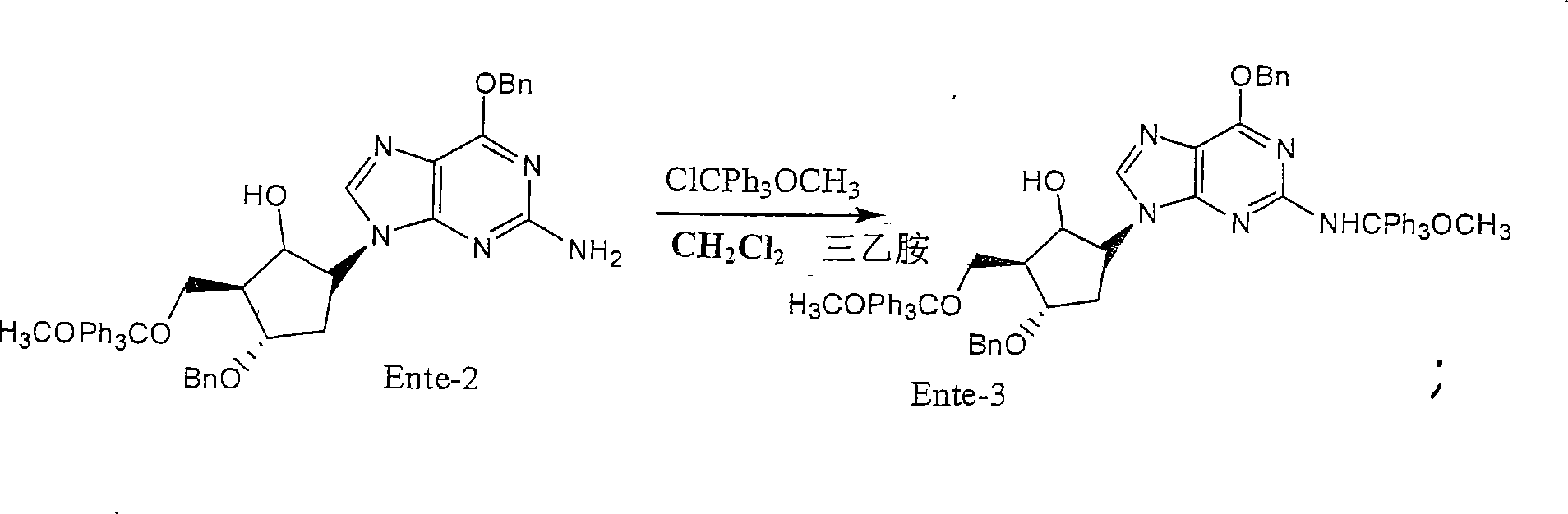
Method for synthesizing entecavir
A technology of entecavir and a synthetic method, applied in directions such as organic chemistry, can solve problems such as high cost, low yield, complicated reaction, etc., and achieve the effects of low production cost, convenient operation and simple process
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
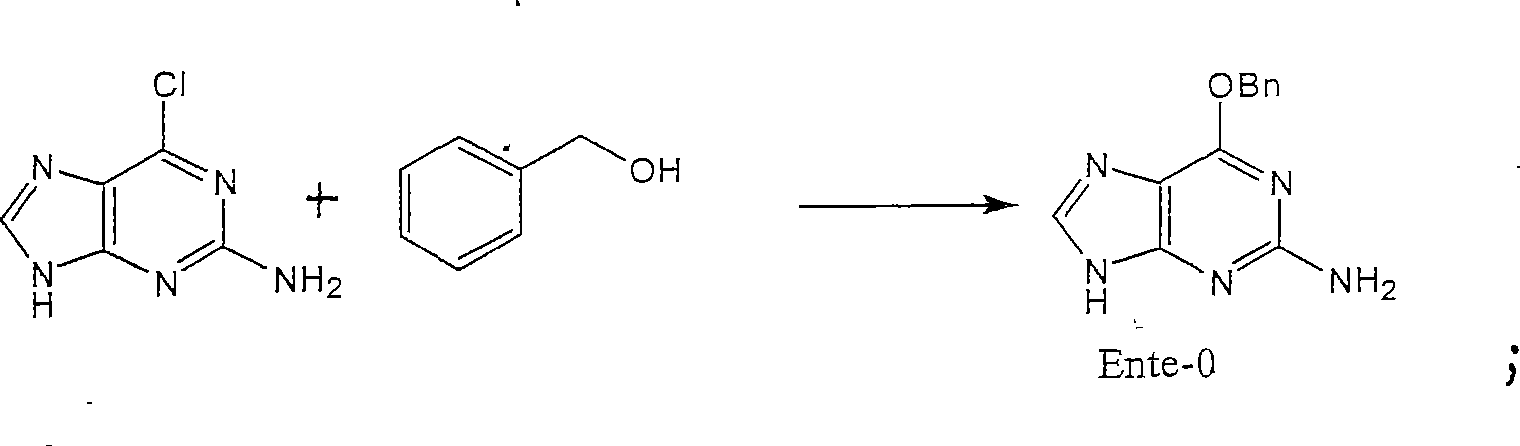
[0029] 1. Preparation of Ente-0:
[0030] Ente-0 is prepared by reacting 2-amino-6-chloropurine with benzyl alcohol in the presence of NaH and 1,4-dioxane. The reaction formula is:
[0031]
[0032] Specific operation: In the reaction flask, add 400g NaH and 12L 1,4-dioxane, dropwise add 1040g benzyl alcohol, stir at room temperature for 1h (hour), add 800g 2-amino-6-chloropurine, and complete the addition , heat up to reflux, react for 48 hours, stop heating, add the reaction solution into 50L ice water, stir to precipitate solids, filter to remove solids, keep the filtrate at 10°C, adjust the pH of the filtrate to 6-7 with glacial acetic acid, precipitate solids, and filter , wash the filter cake with methanol, and dry the solid to obtain Ente-0.
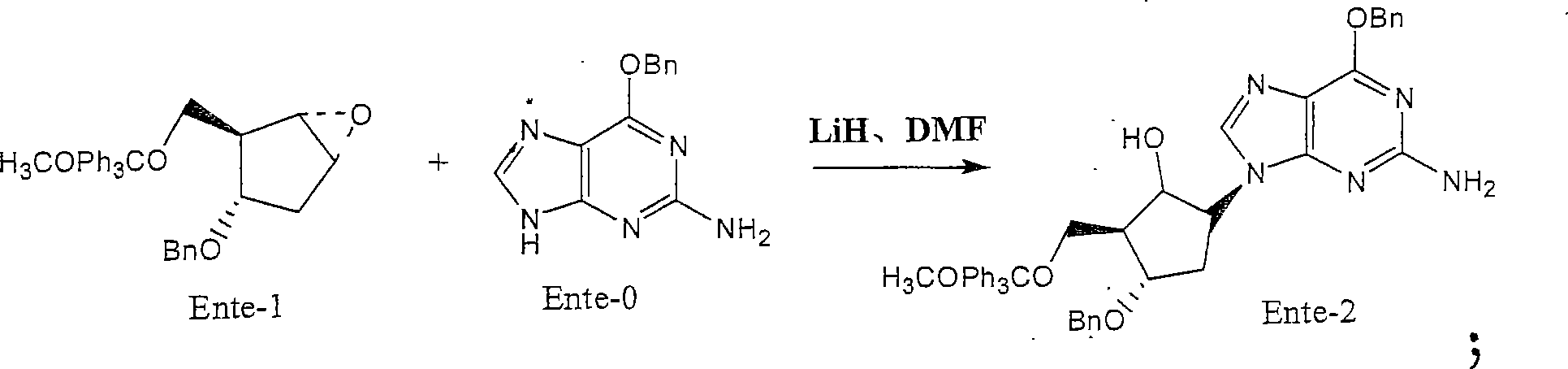
[0033] 2. Preparation of Ente-2:
[0034] Reaction of Ente-1 and Ente-0 in the presence of LiH and DMF to prepare Ente-2, the reaction formula:
[0035]
[0036] Feeding:
[0037] Name Feeding
[0038] Ente-0 720g
[0...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More