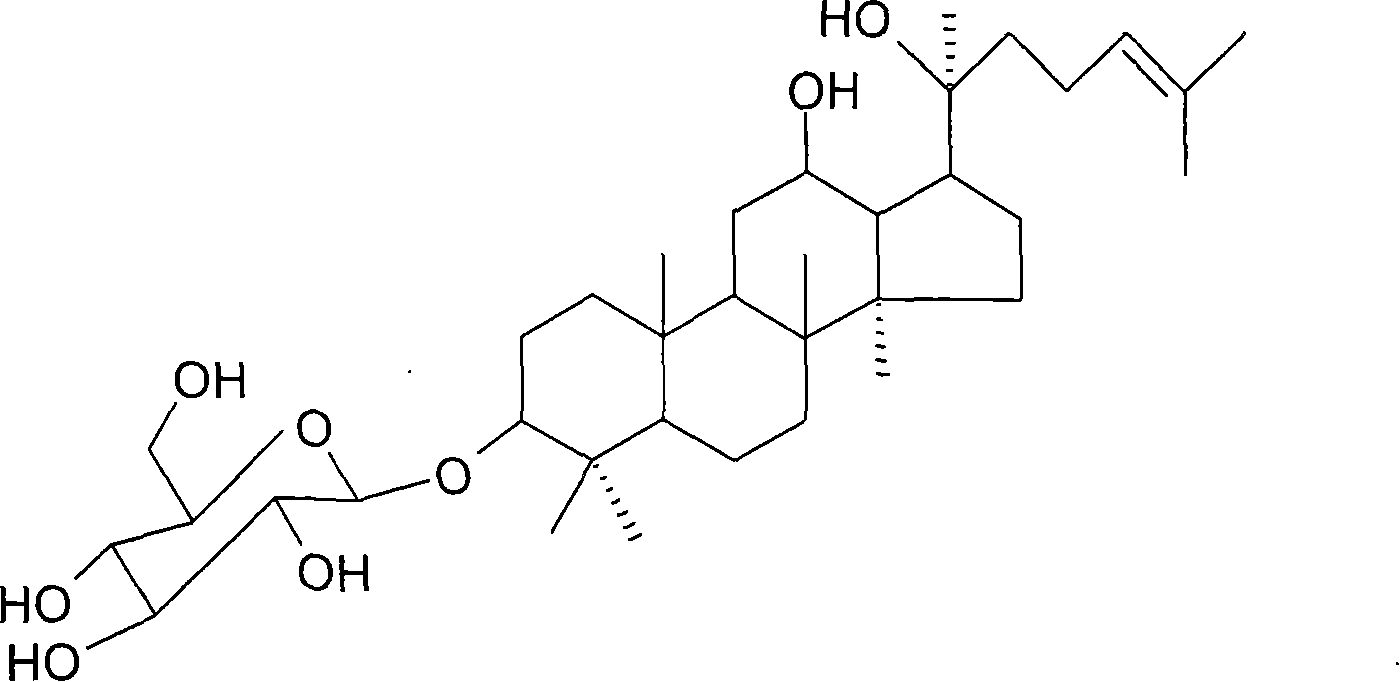
Application of 20(S)-ginsenoside Rh2 compound in preparing anti-fatigue medicament
A technology of ginsenosides and compounds is applied in the application field of cancer-related fatigue drugs, which can solve the problems of unrelieved fatigue and lack of fatigue.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0172] Preparation of 20(S)-ginsenoside Rh2 ordinary capsules
[0173] Prescription: 20(S)-Ginsenoside Rh2 25mg
[0174] Lactose 170mg
[0175] Carboxymethyl Starch Sodium 5mg
[0176]5% povidone (K30) in 70% ethanol solution
[0177] Take 20(S)-ginsenoside Rh2, lactose, and sodium carboxymethyl starch through a 80-mesh sieve and mix evenly, add 5% povidone K30 in 70% ethanol solution to make a soft material, pass through a 40-mesh sieve to granulate. 50 ~ 60 ℃ oven blast drying. The dry granules are sieved through a 40-mesh sieve. Fill No. 1 capsules according to the prescribed amount, each containing 20(S)-ginsenoside Rh2 25mg.
Embodiment 2
[0179] Preparation of 20(S)-ginsenoside Rh2 ordinary capsules
[0180] Prescription: 20(S)-Ginsenoside Rh2 25mg
[0181] Microcrystalline Cellulose 170mg
[0182] Crospovidone 5mg
[0183] 5% povidone (K30) in 70% ethanol solution
[0184] Take 20(S)-ginsenoside Rh2, microcrystalline cellulose, and cross-linked povidone through a 80-mesh sieve and mix evenly, add an appropriate amount of 5% povidone K30 in 70% ethanol solution to make a soft material, pass through a 40-mesh sieve to granulate . 50 ~ 60 ℃ oven blast drying. The dry granules are sieved through a 40-mesh sieve. Fill No. 1 capsules according to the prescribed amount, each containing 20(S)-ginsenoside Rh2 25mg.
Embodiment 3
[0186] Preparation of 20(S)-ginsenoside Rh2 ordinary capsules
[0187] Prescription: 20(S)-Ginsenoside Rh2 25mg
[0188] Povidone (K30) 175mg
[0189] Proper amount of ethanol
[0191] Take 20(S)-ginsenoside Rh2, povidone, add appropriate amount of ethanol and heat to dissolve, put the rotary evaporator to remove ethanol, cool, pulverize, pass through a 40-mesh sieve, add the prescribed amount of magnesium stearate, and mix well. Fill No. 1 capsules according to the prescribed amount, each containing 20(S)-ginsenoside Rh2 25mg.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More