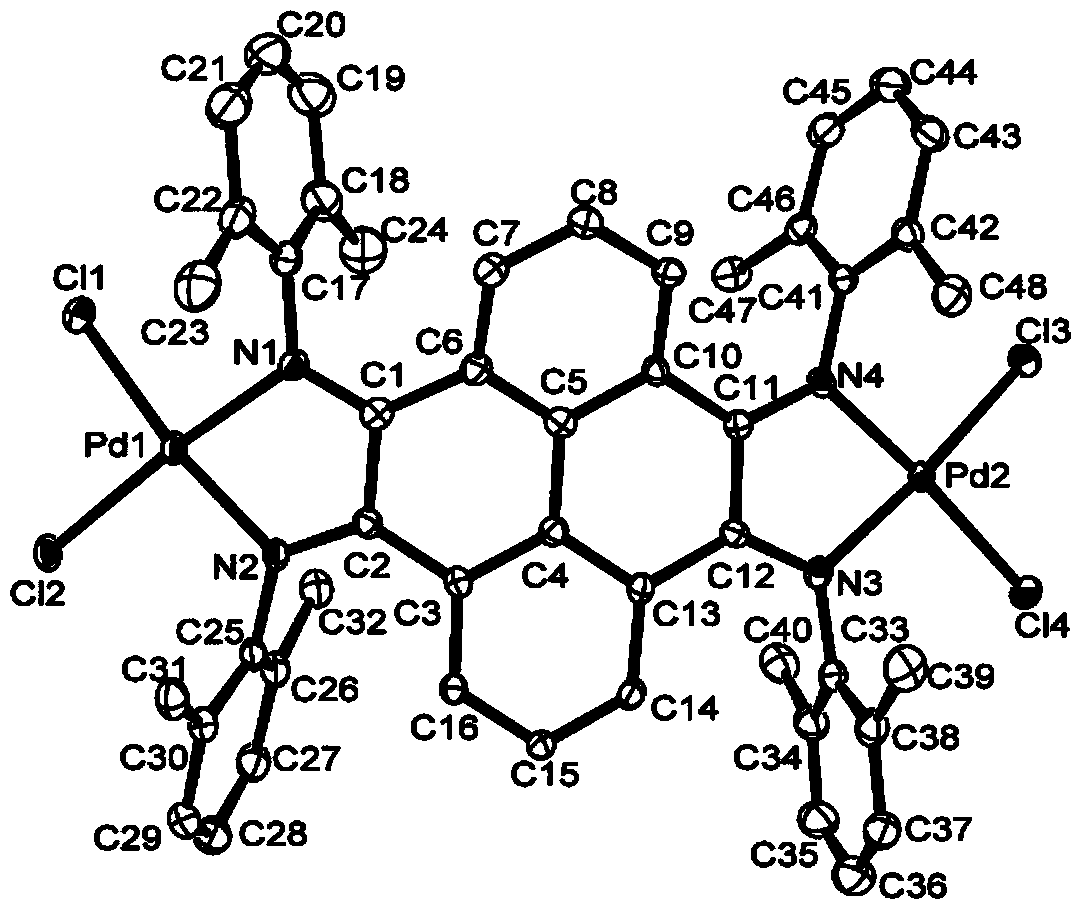
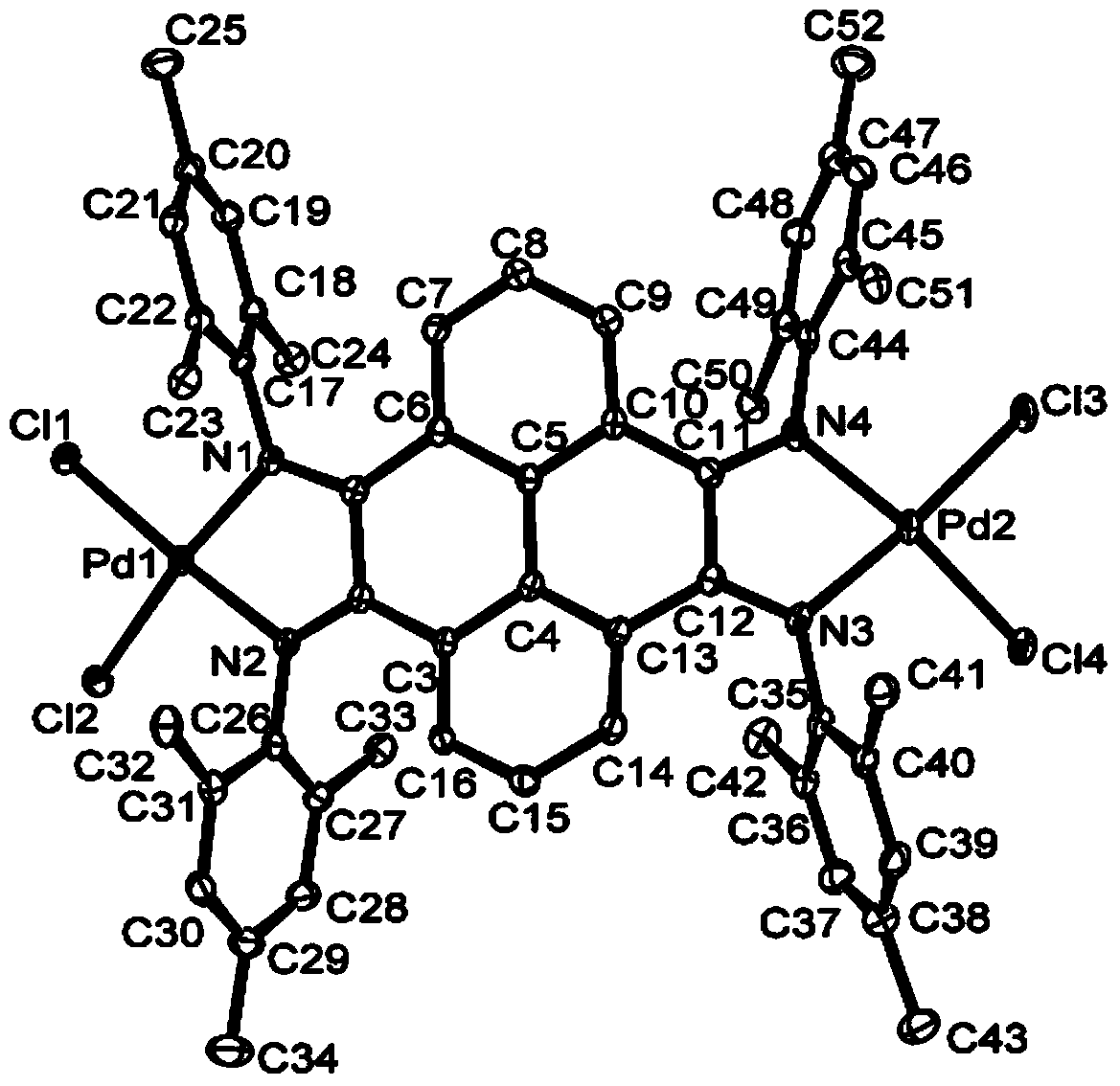
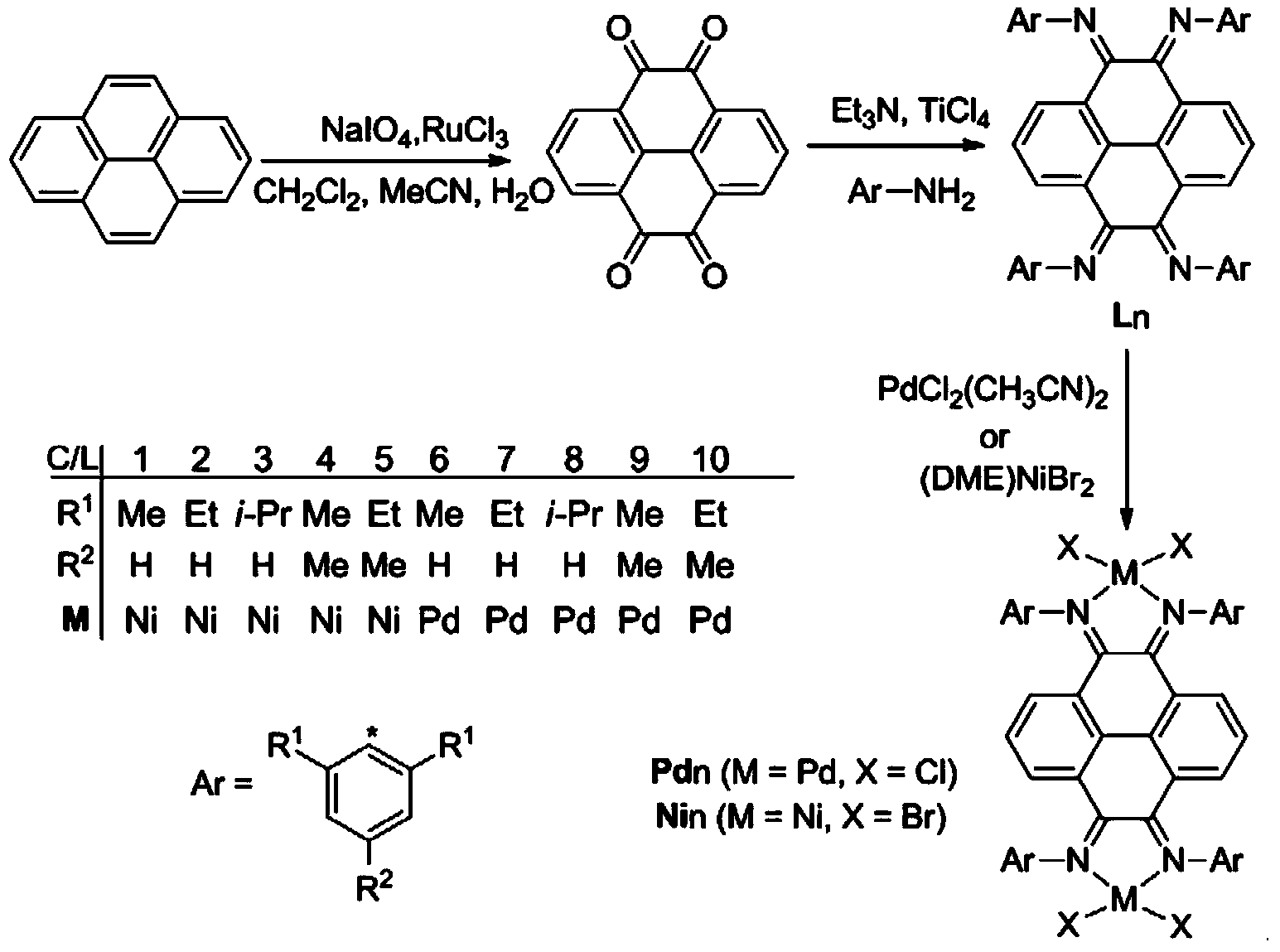Pyrene-4,5,9,10-tetra-imine nickel-palladium complex catalyst and preparation method and application thereof
A catalyst and compound technology, which is applied in the direction of preparation of imino compounds, compounds containing elements of Group 8/9/10/18 of the periodic table, nickel organic compounds, etc., can solve the problems of limited metal catalysts, achieve high catalytic activity, Broad industrial application prospects and the effect of increasing catalytic life
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0074] Embodiment 1, preparation of pyrene-4,5,9,10-tetraimine-(2,6-dimethylanilino) [L1] (R 1 is methyl, R 2 for hydrogen)
[0075]
[0076] 3.60g (30mmol) belongs to the compound 2 of formula III, 6-dimethylaniline (R 1 is methyl, R 2 For hydrogen) and 9.08g (90mmol) triethylamine dissolved in 100ml solvent toluene and heated to 90 ° C, then added 3.2ml (30mmol) TiCl 4 , white smoke appeared, and then 1.33 g (5 mmol) of the compound pyrene-4,5,9,19-tetraketone represented by formula II was added. The condensation reaction was carried out for 30 minutes, and the mixture was subjected to column chromatography, and the mixed solvent of petroleum ether and dichloromethane was used as the eluent to elute, and the eluted fraction was detected by thin-layer chromatography to obtain 1.94 g of a red solid, with a yield of 57%.
[0077] 1 H NMR (400MHz, CDCl 3 ,ppm):8.37(d,J=8.0Hz,2H,Ph-H),7.13(t,J=7.6,2H,Ph-H),7.02(d,J=7.6Hz,4H,Ph-H) ,6.92(t,J=7.6,2H,Ph-H),6.82-6.86(m,8H,Ph...
Embodiment 2
[0078] Embodiment 2, preparation of pyrene-4,5,9,10-tetraimine-(2,6-diethylanilino) [L2] (R 1 is ethyl, R 2 for hydrogen)
[0079]
[0080] 4.47g (30mmol) 2,6-dimethylaniline and 9.08g (90mmol) triethylamine were dissolved in 100ml toluene and heated to 90°C, then added 3.2ml TiCl 4 , white smoke appeared, and then 1.33g (5mmol) pyrene-4,5,9,19-tetraketone was added. The condensation reaction was carried out for 30 minutes. The mixture was subjected to column chromatography and eluted with a mixed solvent of petroleum ether and ethyl acetate as the eluent. The eluted fraction was detected by thin-layer chromatography to obtain 0.86 g of a red solid with a yield of 32.0%.
[0081] The structural confirmation data are as follows: 1 H NMR (400MHz, CDCl 3 ,ppm):8.36(d,J=8.0Hz,2H,Ph-H),7.11-7.04(m,10H,Ph-H),6.96-6.83(m,6H,Ph-H),2.52-2.46( m,4H,2×CH 2 ),2.39-2.34(m,4H,2×CH 2 ),1.69-1.62(m,8H,4×CH 2 ),0.87(t,J=7.6Hz,12H,4×CH 3 ),0.73(t,J=7.6Hz,12H,4×CH 3 ). 13 C NMR (100M...
Embodiment 3
[0082] Embodiment 3, preparation of pyrene-4,5,9,10-tetraimine-(2,4,6-trimethylanilino) [L4] (R 1 is methyl, R 2 for methyl)
[0083]
[0084] 4.05g (30mmol) 2,6-dimethylaniline and 9.08g (90mmol) triethylamine were dissolved in 100ml toluene and heated to 90°C, then added 3.2ml TiCl 4 , white smoke appeared, and then 1.33g (5mmol) pyrene-4,5,9,19-tetraketone was added. The condensation reaction was carried out for 30 minutes, and the mixture was subjected to column chromatography, eluting with a mixed solvent of petroleum ether and dichloromethane as an eluent, and the eluted fraction was detected by thin-layer chromatography to obtain 1.69 g of a red solid, with a yield of 45.6%.
[0085] The structural confirmation data are as follows: 1 H NMR (400MHz, CDCl 3 ,ppm):8.29(d,J=7.6Hz,2H,Ph-H),7.12(t,J=7.6Hz,2H,Ph-H),6.88(t,J=7.6Hz,2H,Ph-H ),6.84(s,4H,Ph-H),6.63(s,4H,Ph-H),2.34(s,6H,2×CH 3 ),2.27(s,6H,2×CH 3 ),2.18(s,12H,4×CH 3 ),1.31(s,12H,4×CH 3 ). 13 C NMR (100MH...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com