Polypeptide, polypeptide derivative, medicinal salt of polypeptide and pharmaceutical composition
A technology of peptide derivatives and compositions, applied in the direction of drug combinations, microorganisms, biochemical equipment and methods, etc., can solve the problems of short plasma half-life, achieve long plasma half-life, good stability, and improve the function of islets of pancreas
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0128] Example 1 Production of Polypeptides
[0129] This example describes the production of polypeptides
[0130] All peptides used in this study were synthesized using 9-fluorenylmethyl chloroformate (Fmoc) solid-phase synthesis. Simply put: Dissolve a weighed amount of 2-chlorotrityl chloride resin (1.6mmol / g) in dichloromethane (DCM). For C-terminally amidated peptides of interest, use Rink amide resin instead of 2-chlorotrityl chloride resin. For coupling reactions in the presence of hydroxybenzotriazole (Sigma Chemicals, Inc., St. Louis, MO, USA) in dimethylformamide (DMF), preactivated Fmoc-amino acids were used. The entire synthesis process uses an excess of amino acids. Deprotection of the Fmoc group in 20% piperidine in DMF leads to chain extension reactions. When the chain extension reaction was completed, the Fmoc protecting group was removed from the N-terminus of the polypeptide using DMF containing 25% piperidine, and then washed four times with DMF solutio...
Embodiment 2
[0134] Example 2 The promoting effect of polypeptide on cell proliferation
[0135] This example describes the effects of polypeptides on the growth of pancreatic cells.
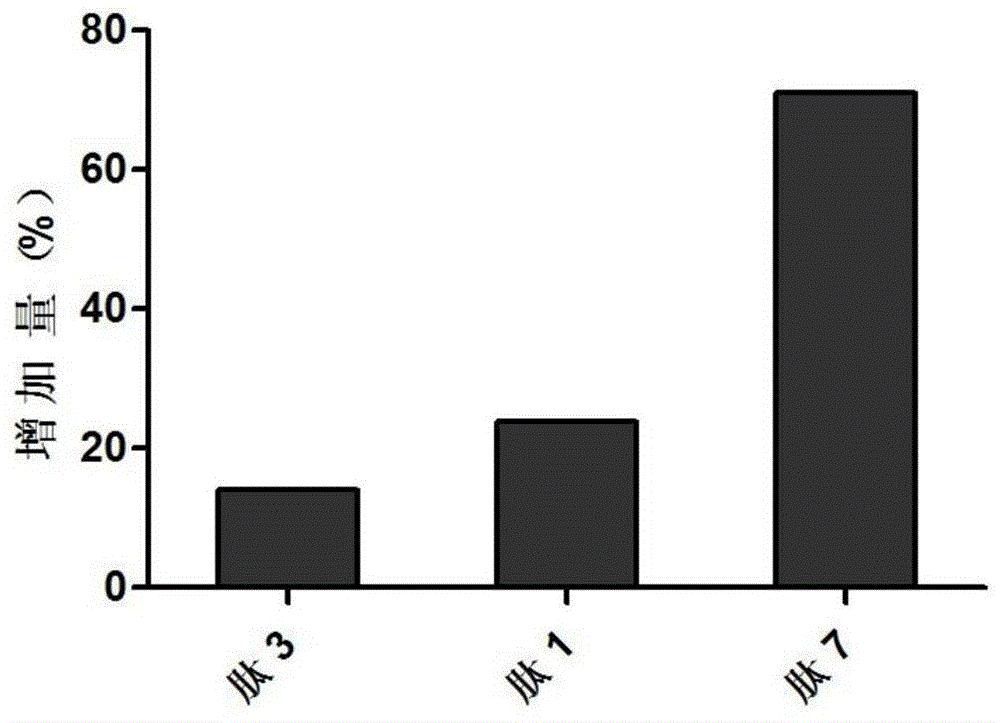
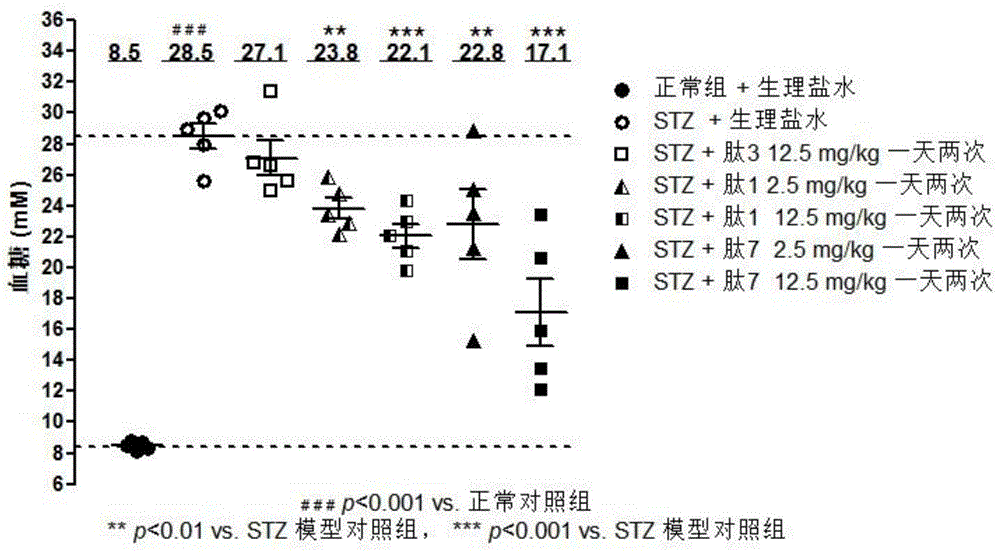
[0136] Cell proliferation was measured by bromodeoxyuridine (BrdU)-labeled ELISA. Briefly, a rat pancreatic ductal ARIP cell (ATCC (American Type Culture Collection), Manassas, VA, USA) was cultured in 10% fetal bovine serum (FBS, Fetal Bovine Serum, Thermo Fisher Inc., Waltham, MA, USA), in F-12K medium (GIBCO-BRL, Gaithersburg, MD, USA) with 100 μg / ml streptomycin and 100 μg / ml penicillin. ARIP cells were inoculated into 96-well culture plates at a density of 8000 or 0 (blank control) cells / well, and 50 μl of cell culture medium was added to culture overnight for use in experiments. The next day, use serum-free medium to replace the original medium, and 50 μl of serum-free cell culture medium containing a series of concentrations of the test polypeptide (final concentrations are 10 μM, 5 μM, 1 μM, 500 nM...
Embodiment 3
[0142] Example 3 Peptide Stability Experiment
[0143] This example describes stability experiments of peptides under various conditions.
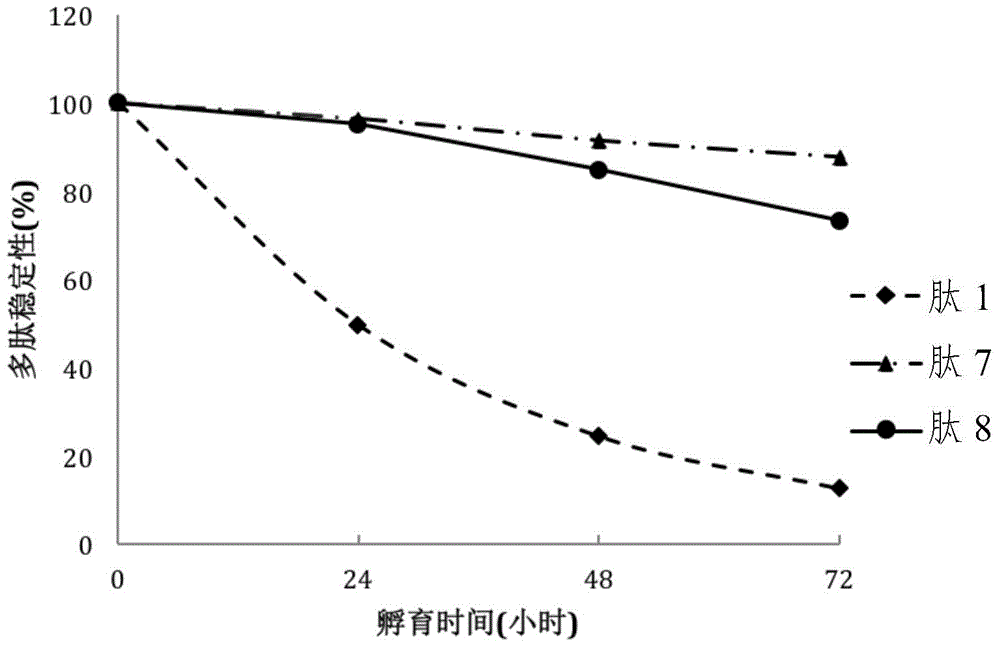
[0144]Accurately weigh a certain amount of the selected peptide, dissolve it in distilled water to a concentration of 5 mg / mL, and use it as a stock solution to examine the stability of the peptide in the culture medium. Use F-12K medium (GIBCO-BRL, Gaithersburg, MD, USA) to dilute the stock solution to 0.25 mg / mL as a working solution. Transfer each 100 µL of working solution to a separate vial. After the vials were placed in a 37°C incubator for 0, 24, 48 and 72 hours, quantitative analysis was performed using HPLC.
[0145] figure 2 and Table 6 shows the stability of the compounds in the medium. figure 2 In particular, a comparison of the stability in culture medium of INGAP-PP (peptide 1) and selected peptides, peptides 7 and 8 (see Table 2) is shown.
[0146] Table 6
[0147] polypeptide
0h
24h
48h
72h...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More