Device and method for synthesizing 2,2-bipyridyl
A bipyridine and pyridine technology, applied in the field of synthesis of 2,2-bipyridine, can solve the problems of high separation pressure, low utilization rate of pyridine, high cost, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1-4
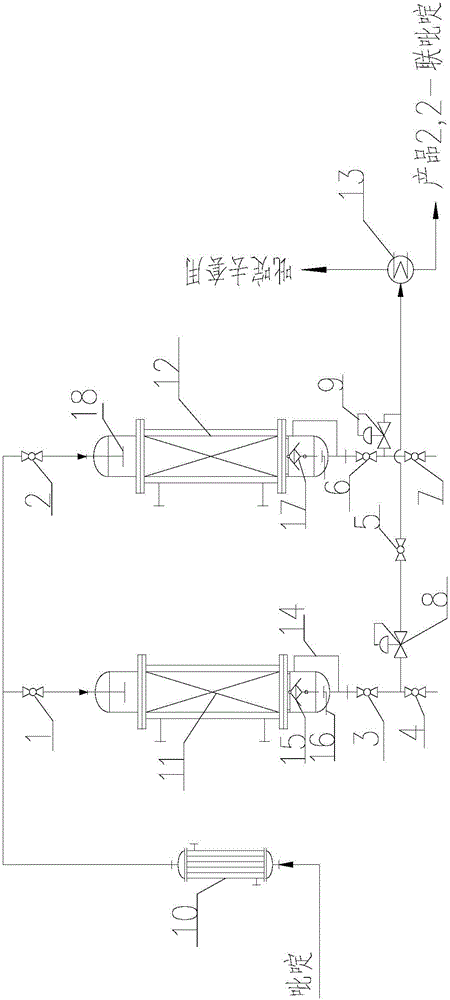
[0023] The 2,2-bipyridine synthesis process is that pyridine is transported by a pump, passes through a preheater, is heated to the reaction temperature, enters the reactor, passes through a liquid distributor, and is evenly washed onto the catalyst fixed bed Raney nickel on the catalyst bed, pyridine Liquid space velocity 2~4h -1 After the reaction, pyridine flows down through the catalyst support plate, and is discharged through the regulating valve. The reaction liquid enters the evaporator to distill the pyridine, recover and use it mechanically, and the residue is 2,2-bipyridine. The reaction conditions of Examples 1-4 and the conversion rate of pyridine per pass are shown in Table 1.
[0024] Table 1 Experimental data of 2,2-bipyridine synthesis
[0025] Example
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More