Synthesis method of dibutyltin dilaurate
A technology of dibutyltin laurate and synthesis method, which is applied in the direction of tin organic compounds, chemical instruments and methods, compounds of group 4/14 elements of the periodic table, etc., and can solve problems such as many process steps, ethanol residue, and long synthesis time , to achieve the effect of simple process, reduced product loss and short time consumption
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
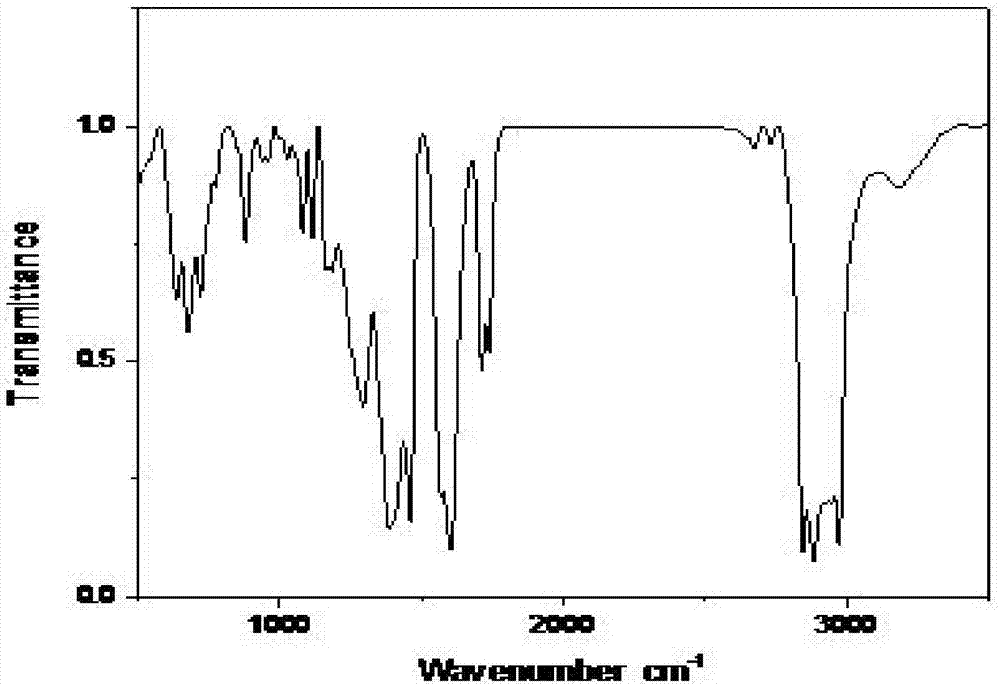
Embodiment 1
[0021] The synthetic method of the present embodiment dibutyltin dilaurate comprises the following steps:
[0022] Step 1, stirring the solid mixture of 100g lauric acid and 62g dibutyltin oxide at 25°C to convert the solid mixture into a liquid mixture;
[0023] Step 2. Heat the liquid mixture obtained in Step 1 to 65°C, and then reflux for 2.5 hours;
[0024] Step 3. Cool the liquid mixture after the reflux reaction in step 2 to 20° C., collect the organic phase after standing and separating, add anhydrous magnesium sulfate to the organic phase to remove water for 9 hours, and then filter to obtain the filtrate It is dibutyltin dilaurate; the amount of anhydrous magnesium sulfate added is 8g of anhydrous magnesium sulfate per 100mL of organic phase.
[0025] This embodiment obtains 153.5g of dibutyltin dilaurate, the theoretical output value of dibutyltin dilaurate calculated according to the reaction equation is 157.9g, and the yield of dibutyltin dilaurate is 97.2%.
[0...
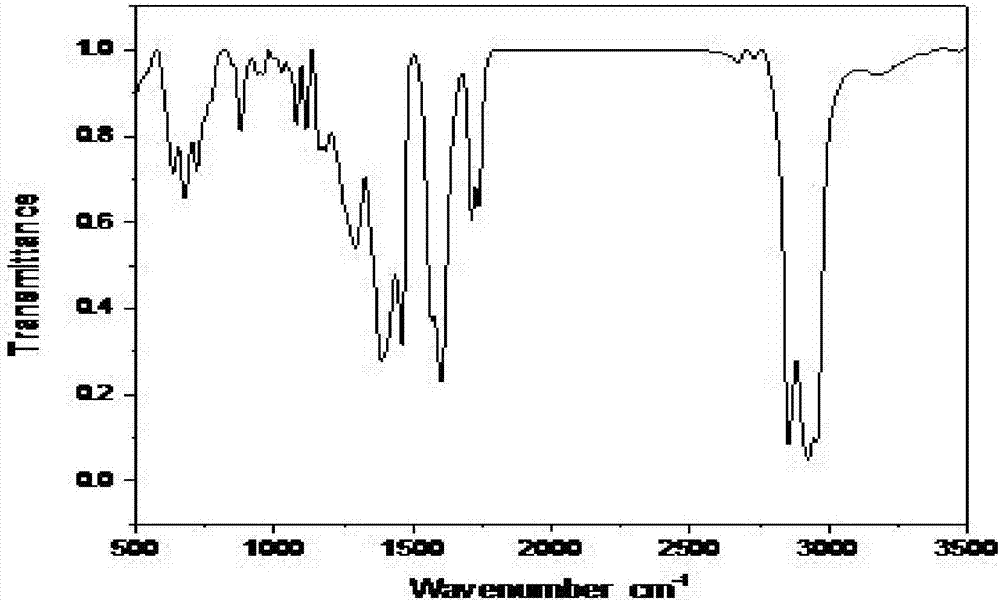
Embodiment 2
[0028] The synthetic method of the present embodiment dibutyltin dilaurate comprises the following steps:
[0029] Step 1, stirring the solid mixture of 176g lauric acid and 109g dibutyltin oxide at 20°C to convert the solid mixture into a liquid mixture;
[0030] Step 2, heating the liquid mixture obtained in Step 1 to 60°C, and then reflux for 2 hours;
[0031] Step 3. Cool the liquid mixture after the reflux reaction in step 2 to 10°C, and collect the organic phase after standing for stratification. Add anhydrous magnesium sulfate to the organic phase to remove water for 8 hours, and then filter to obtain the filtrate It is dibutyltin dilaurate; the amount of anhydrous magnesium sulfate added is 5g of anhydrous magnesium sulfate per 100mL of organic phase.
[0032] This embodiment obtains 266.3g of dibutyltin dilaurate, the theoretical output value of dibutyltin dilaurate calculated according to the reaction equation is 276.0g, and the yield of dibutyltin dilaurate is 97.2...
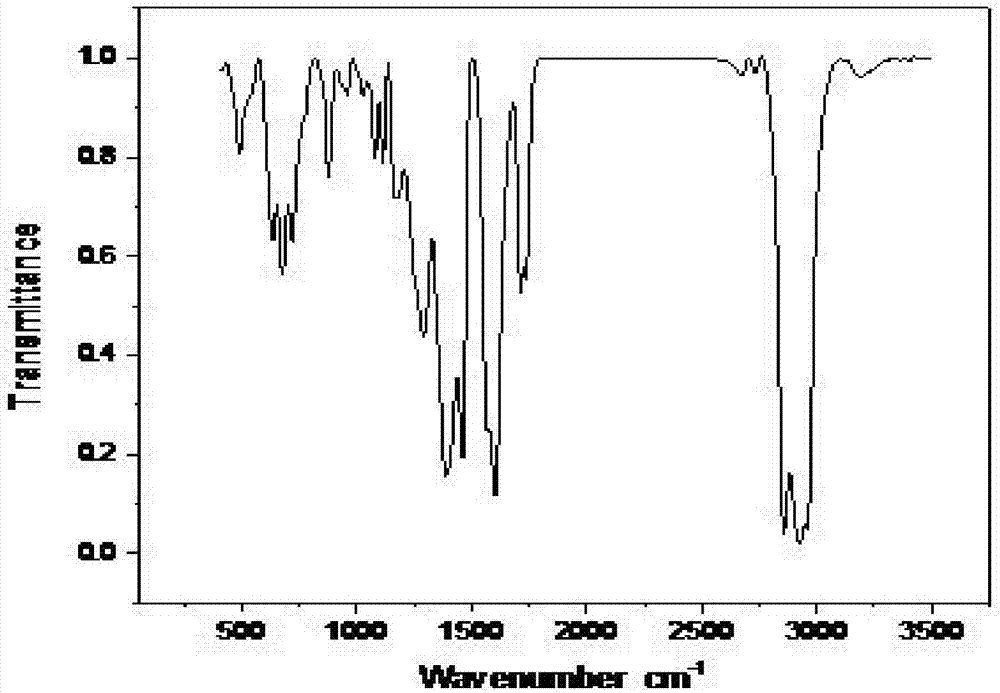
Embodiment 3
[0035] The synthetic method of the present embodiment dibutyltin dilaurate comprises the following steps:
[0036] Step 1, stirring the solid mixture of 264g lauric acid and 164g dibutyltin oxide at 30°C to convert the solid mixture into a liquid mixture;
[0037] Step 2, heating the liquid mixture obtained in step 1 to 70°C, and then reflux for 3h;
[0038] Step 3. Cool the liquid mixture after the reflux reaction in step 2 to 30° C., collect the organic phase after standing for stratification, add anhydrous magnesium sulfate to the organic phase to remove water for 10 hours, and then filter to obtain the filtrate It is dibutyltin dilaurate; the amount of anhydrous magnesium sulfate added is 10g of anhydrous magnesium sulfate per 100mL of organic phase.
[0039] This embodiment obtains 395.4g of dibutyltin dilaurate, the theoretical output value of dibutyltin dilaurate calculated according to the reaction equation is 416.2g, and the yield of dibutyltin dilaurate is 95.0%.
...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More