Co-crystal of pyrazinamide and quercetin and preparation method of co-crystal
A technology of pyrazinamide and quercetin, which is applied in the field of pharmaceutical co-crystals of pyrazinamide and quercetin and its preparation, can solve the problems of limited clinical application and low bioavailability, and achieve high yield and purity, The effect of low cost and simple process
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
specific Embodiment approach 1
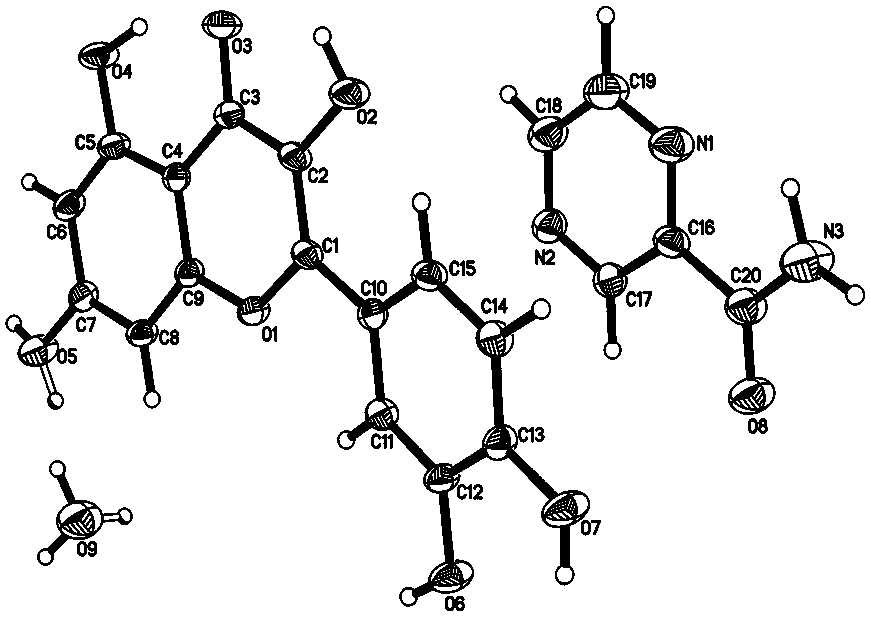
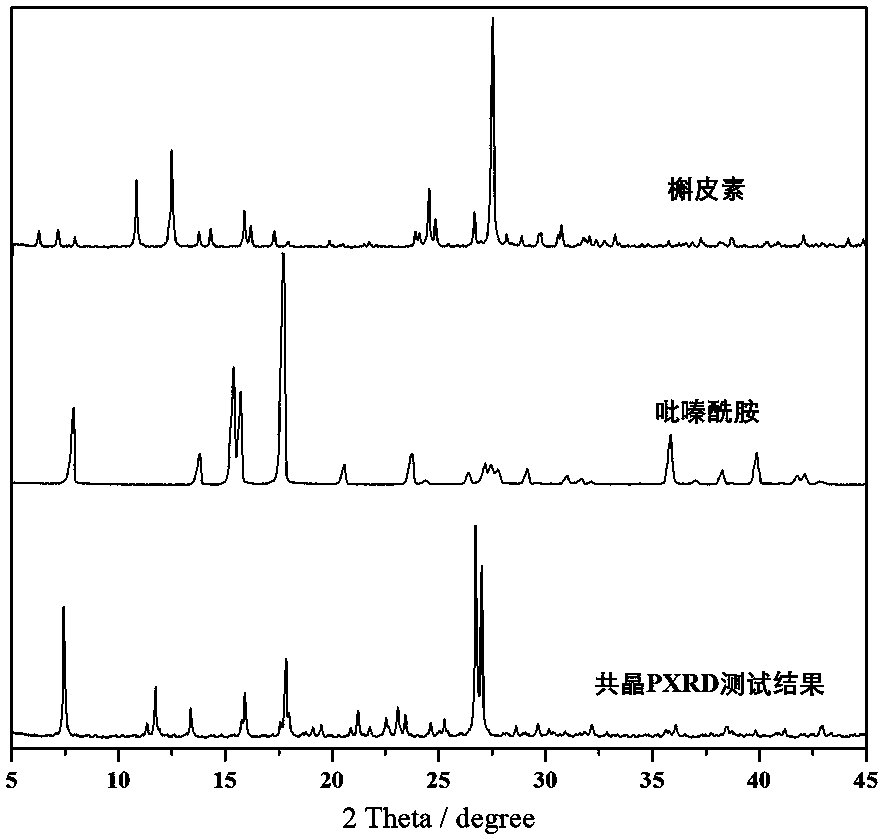
[0021] Specific embodiment one: the structural formula of pyrazinamide-quercetin co-crystal of this embodiment is [C 5 h 5 N 3 O·C 15 h 10 o 7 ·H 2 O], the basic structural unit is formed by a pyrazinamide molecule, a quercetin molecule, and a water molecule. The pyrazinamide-quercetin cocrystal is a monoclinic system with a space group of P 2 1 / n, the unit cell parameters are: a = 8.759 Å, b = 23.793 Å, c = 10.012 Å, α = 90°, β = 114.56°, γ = 90°. Its PXRD characteristic diffraction peaks appear at 7.422°, 11.702°, 13.362°, 15.901°, 17.799°, 21.183°, 23.042°, 23.402°, 26.703°, 26.978°.
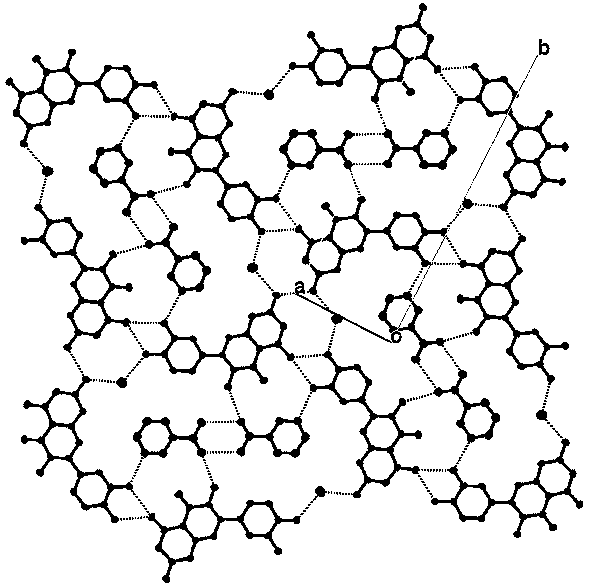
[0022] The pyrazinamide-quercetin co-crystal described in this embodiment is composed of a pyrazinamide molecule, a quercetin molecule and a water molecule combined by supramolecular interactions such as hydrogen bonds. Such as figure 2 As shown, the pyrazinamide molecules in the co-crystal form a hydrogen-bonded dimer through the amide-amide synthon; four quercetin molecules and ...
specific Embodiment approach 2
[0023] Specific embodiment two: the preparation method of pyrazinamide-quercetin co-crystal of this embodiment is implemented according to the following steps:
[0024] Mix pyrazinamide raw material drug and quercetin at a molar ratio of 1:1, add to ethanol solution at 60°C, stir for 1-2 hours and then filter while it is hot. The pyrazinamide-quercetin co-crystal was obtained.
specific Embodiment approach 3
[0025] Embodiment 3: This embodiment differs from Embodiment 2 in that the solid-to-liquid ratio of the mixture powder to ethanol in the system is 100 mg: (10-15) mL. Other steps and parameters are the same as in the second embodiment.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More