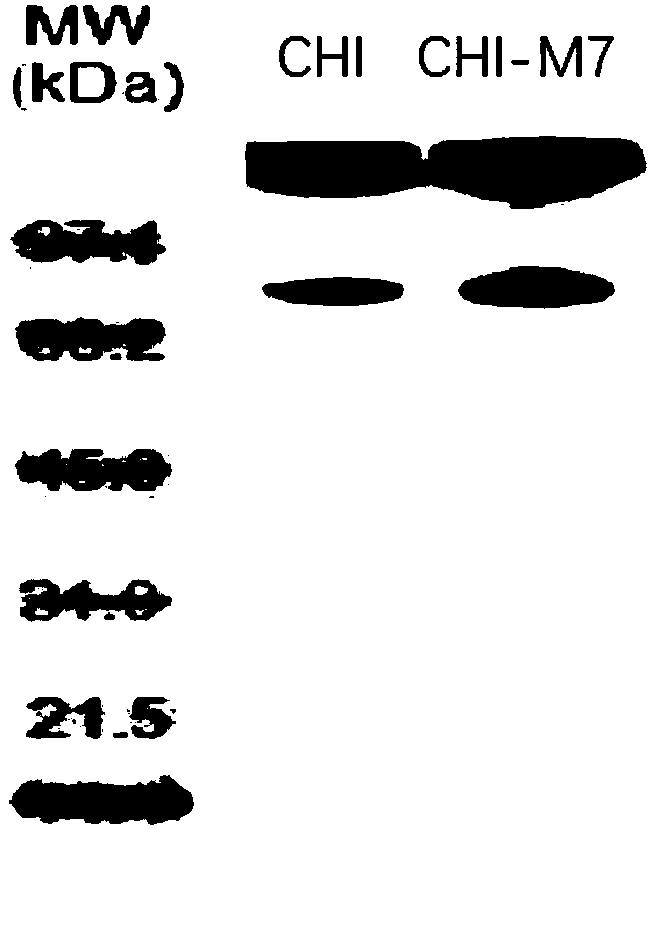Acid-resisting mutant of low-temperature resistant chitinase and application thereof
A chitinase, chitin technology, applied in application, enzyme, hydrolase and other directions, can solve problems such as acid sensitivity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0024] The gene cloning of embodiment 1 wild-type chitinase
[0025] The inventors have discovered the acid-resistant chitinase disclosed in the prior art, which is screened from Pseudoalteromonas sp., has an optimum temperature of 35°C, and can maintain 25% activity at 10°C. Belonging to acid-resistant enzymes, the chitinase whose amino acid sequence is shown in SEQ ID NO: 1 and whose coding nucleotide is shown in SEQ ID NO: 2 is expressed and prepared in Escherichia coli. In short, Shanghai Jierui Biotechnology Co., Ltd. was entrusted to artificially synthesize the gene, and then ligated into pET-28(+) vector (New England Biolabs) between the restriction sites of EcoR I and Hind III, and transformed into Escherichia coli DH5α was screened on an LB plate containing 50 mg / L ampicillin. The expression vector contained in the screened positive bacteria was named pET-chi, and the plasmid was extracted and identified correctly by sequencing.
Embodiment 2
[0026] Embodiment 2 mutation screening
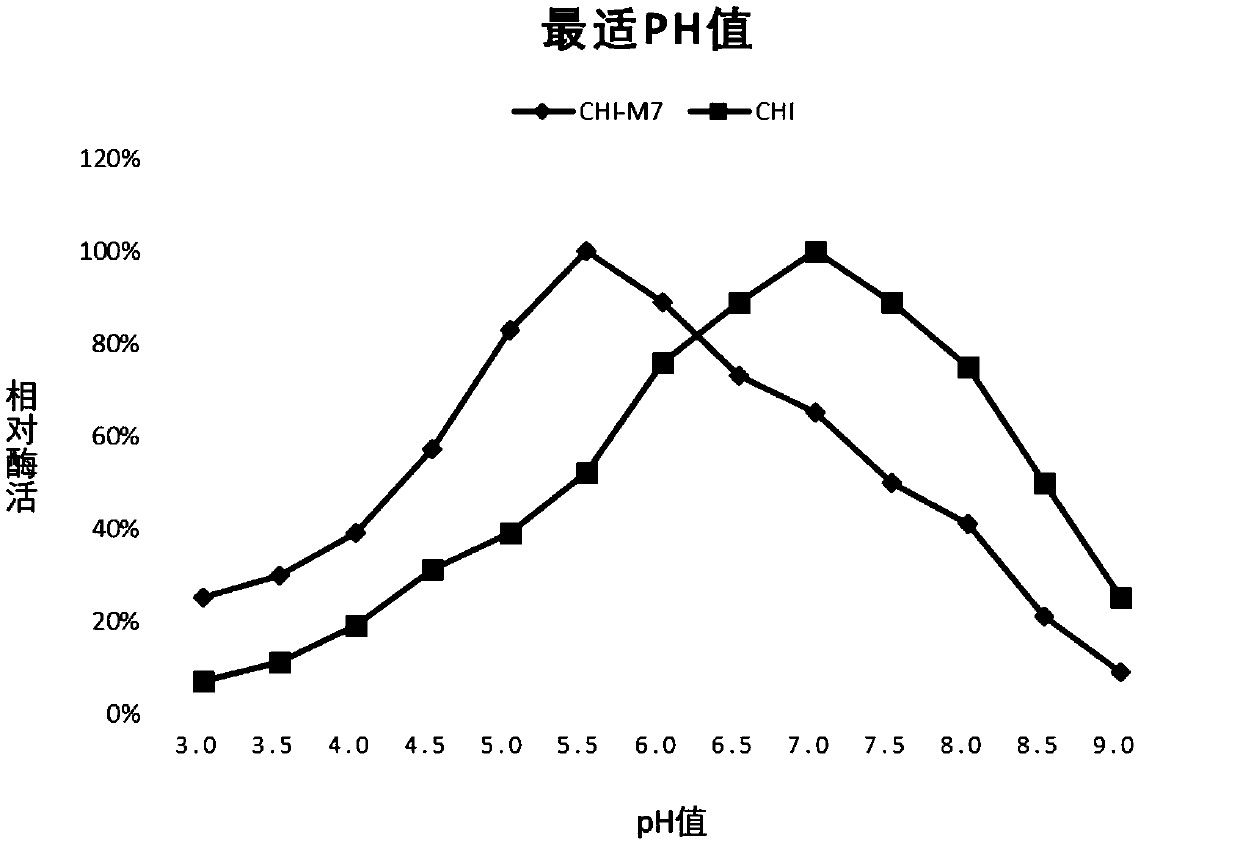
[0027] In order to further improve the acid resistance of the above-mentioned chitinase (the amino acid sequence is SEQ ID NO: 1), the applicant used the recombinant plasmid pET-chi constructed in Example 1 as a template to design random mutation primers for error-prone PCR. , the target gene was randomly mutated at multiple points, and it was found that some mutations had no effect on its acid resistance, some mutations even made the acid stability worse, and some mutations, although they could improve acid resistance, had low temperature resistance after mutation. The performance has been reduced and none of them meet the requirements. In the end, the applicant obtained an enzyme that can significantly improve acid resistance and maintain an acid resistance activity equivalent to that of the wild type. The specific mutation combinations are: I861S, A875C, V894P, G903R, H914K, A927N, M942W; specifically the 861st Ile is mutated to Ser...
Embodiment 3
[0030] Example 3 Expression Purification
[0031] Competent cells of Escherichia coli BL21 (DE3) were prepared by methods known in the art, and the wild-type expression vectors constructed in Example 1 and Example 2 and the expression vectors of mutants were respectively transformed into competent cells by the heat shock method, and positive screening results were obtained. The clones were verified by PCR and sequencing for later use.
[0032] Pick the positive bacteria from the inoculation needle and inoculate them in 5mL LB medium, culture at 30°C, 200r / min for 12h, then inoculate 2% (V / V) inoculum in 400mL LB medium, at 30°C, 200r / min / min for 8 hours. When the bacterial density OD600 reached 0.6, 0.5mM IPTG was added to induce expression. In order to prevent the formation of inclusion bodies, the expression condition was low temperature induction at 16°C overnight.
[0033] The bacteria were collected by centrifugation at 4°C, resuspended in PBS, sonicated on ice, and th...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com