Aromatic amine compound and organic electroluminescent devices thereof
A compound and aromatic amine technology, applied in the field of organic electroluminescence, can solve the problems of inability to effectively block exciton luminescence efficiency, low light extraction efficiency of the cover layer, low glass transition temperature, etc., achieve good hole transport ability, and improve recombination rate. and the effect of high utilization rate and high refractive index
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0117] The synthesis of embodiment 1 compound 2
[0118]
[0119]Toluene solvent was added into the reaction flask, followed by 3-bromo-N-phenylcarbazole (33.85g, 200mmol), 4-aminobiphenyl (70.89g, 220mmol), sodium tert-butoxide (57.66g, 600mmol), After vacuuming and replacing with nitrogen for three times, add Pd(OAc) 2 (0.9g, 4.0mmol), after vacuumizing and nitrogen replacement three times, add P(t-Bu) 3 (6.4mL of 1.0M toluene solution, 6.4mmol), and nitrogen replacement was performed three times, and the mixture was refluxed for 2h under nitrogen atmosphere. After the reaction was stopped, the mixture was cooled to room temperature, filtered through diatomaceous earth to obtain the filtrate, and the filtrate was concentrated , adding 20 mL of methanol, standing for recrystallization, and filtering to obtain compound 2-1 (68.97 g, 84%), and the purity of the solid was detected by HPLC ≧99.9%.
[0120] Add 500mL of THF and raw material a-1 (35.01g, 200mmol) into the reac...
Embodiment 2
[0124] The synthesis of embodiment 2 compound 26
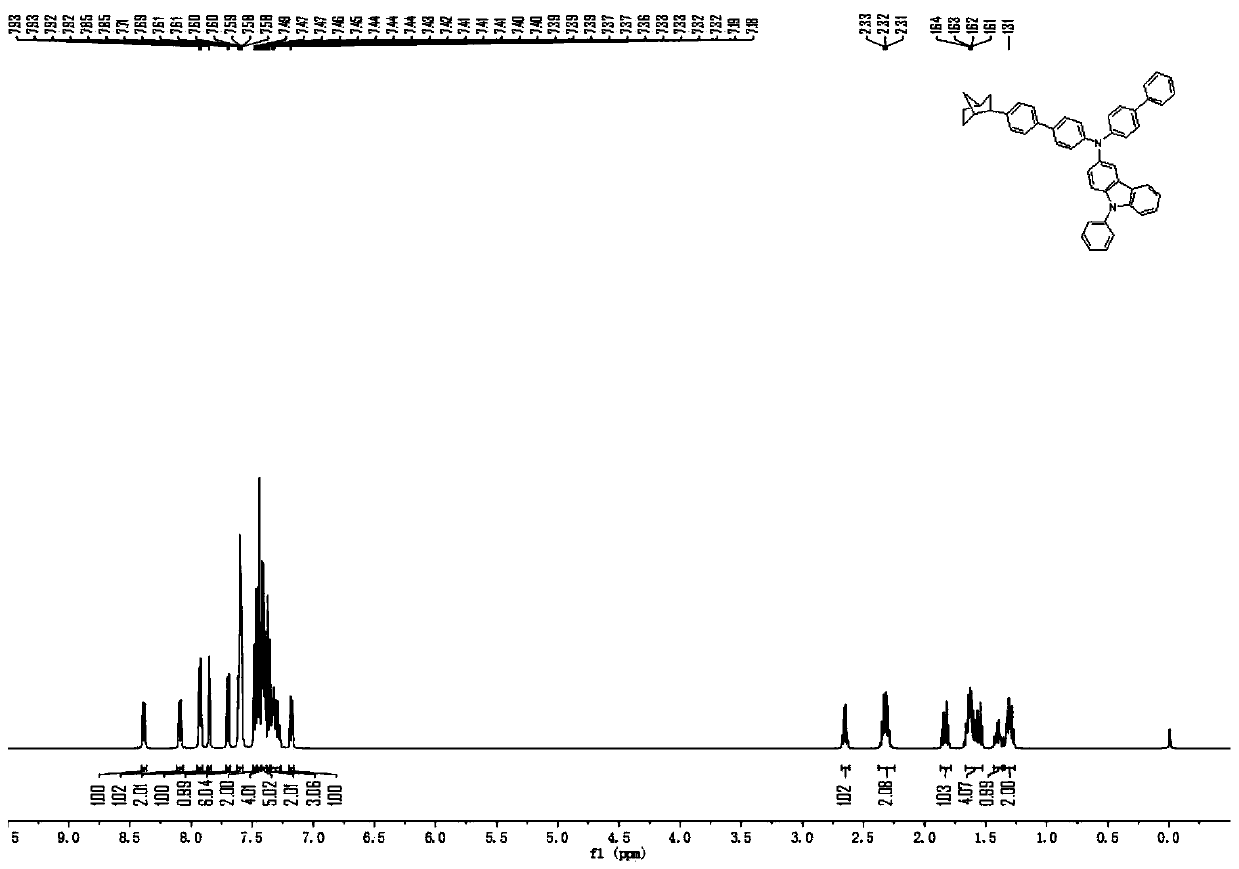
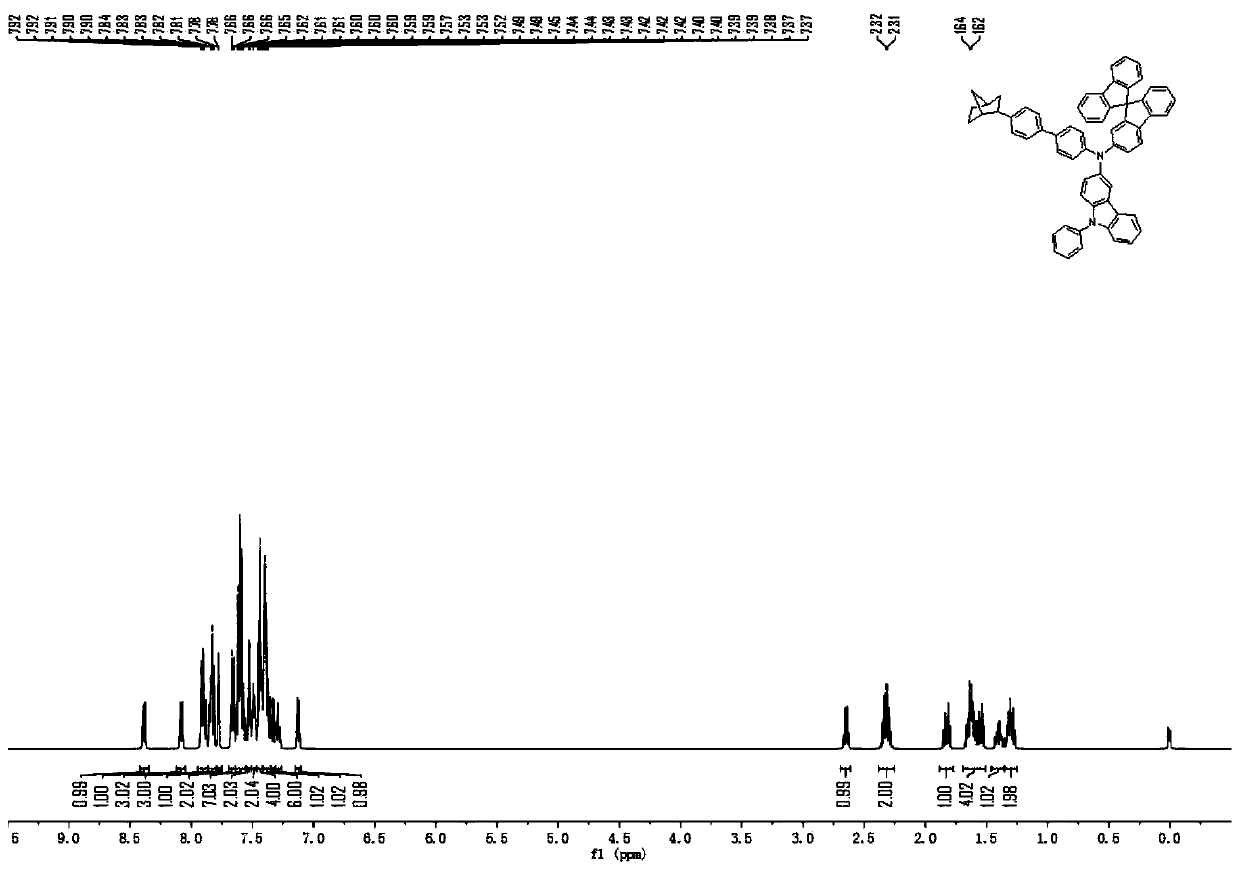
[0125] The raw material 4-aminobiphenyl in Example 1 was replaced with an equimolar amount of 2-amino-9,9-spirobifluorene, and compound 26 (51.76 g, 79%) was obtained according to the synthesis method of compound 2, and the purity of the solid was detected by HPLC ≧99.9%.
[0126] Mass Spectrum m / z: 818.43 (calculated: 818.37). Theoretical element content (%)C 62 h 46 N 2 : C, 90.92; H, 5.66; N, 3.42. Measured element content (%): C, 90.95; H, 5.65; N, 3.40. 1 H-NMR (500MHz, CDCl 3 )(δ,ppm): 8.39(dd,J=7.4,1.5Hz,1H),8.08(dd,J=7.4,1.5Hz,1H),7.95–7.86(m,3H),7.82(dd,J= 6.9,5.1Hz,3H),7.78(d,J=1.5Hz,1H),7.69–7.64(m,2H),7.64–7.55(m,J=7.3,5.7Hz,7H),7.55–7.51(m ,2H),7.49(d,J=1.6Hz,2H),7.46–7.41(m,4H),7.41–7.35(m,6H),7.35–7.31(m,J=7.5,1.5Hz,1H), 7.31–7.26(m, J=7.5, 2.1Hz, 1H), 7.12(dd, J=7.5, 1.4Hz, 1H), 2.65(q, J=7.1Hz, 1H), 2.32(d, J=4.8Hz , 2H), 1.86–1.79 (m, J=13.6, 7.0Hz, 1H), 1.63 (d, J=7.3Hz, 4H), 1.46–1.36 (m, 1H), 1.3...
Embodiment 3
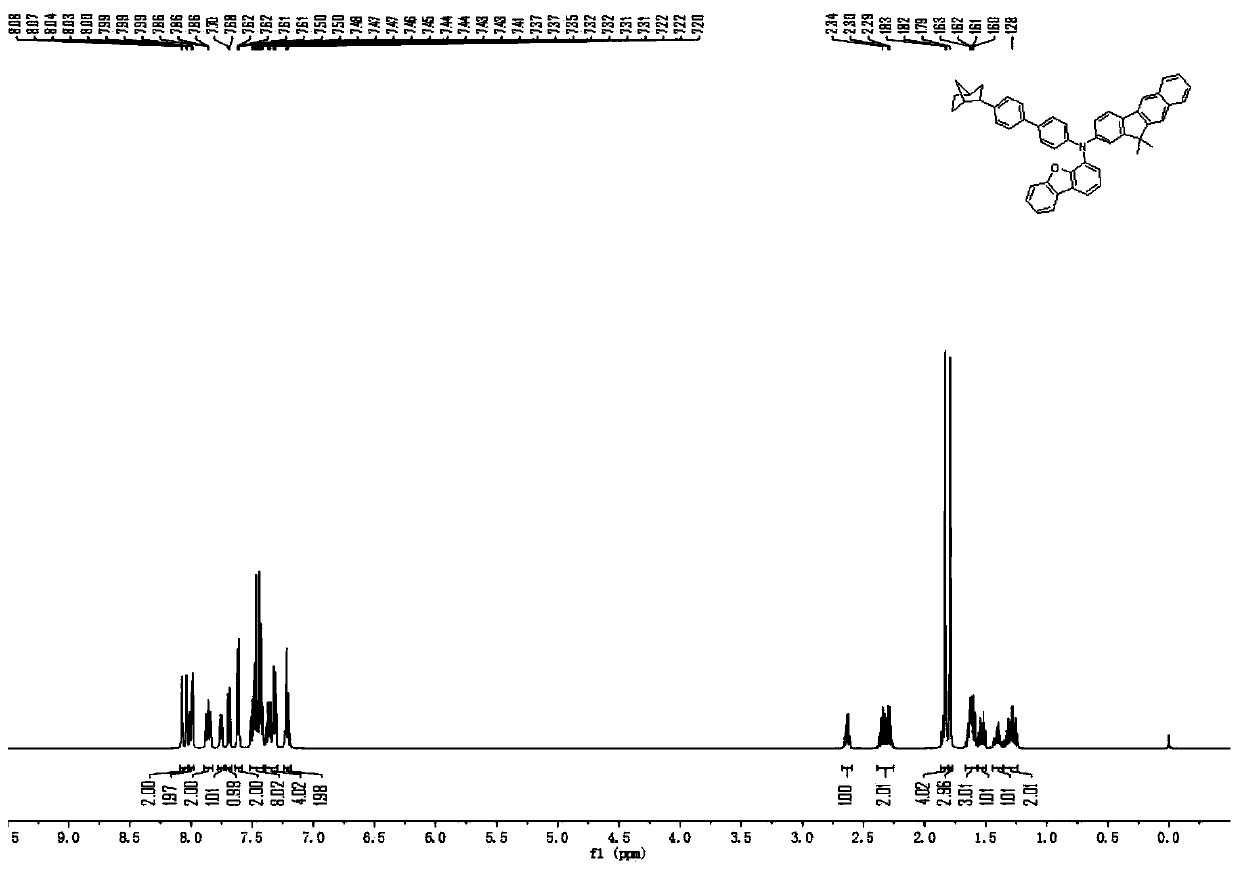
[0127] The synthesis of embodiment 3 compound 67
[0128] The raw material 4-aminobiphenyl in Example 1 was replaced with an equimolar amount of 3-aminodibenzofuran, and compound 67 (42.94 g, 80%) was obtained according to the synthesis method of compound 2, and the solid purity was detected by HPLC ≧99.9%.
[0129] Mass Spectrum m / z: 670.38 (calculated: 670.30). Theoretical element content (%)C 49 h 38 N 2 O: C, 87.73; H, 5.71; N, 4.18; O, 2.38. Measured element content (%): C, 87.75; H, 5.70; N, 4.18; O, 2.37. The above results confirmed that the obtained product was the target product.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Thickness | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More