Octacosanol composition used for improving microcirculation and lowering blood fat, and application
A technology of octacosanol and composition, which is applied in the direction of application, drug combination, active ingredient of hydroxyl compound, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0025] Example 1 Preparation of Octacosanol Composition for Improving Microcirculation and Lowering Blood Lipid
[0026] 5 g of octacosanol, 160 g of Panax notoginseng extract, 165 g of salvia miltiorrhiza extract, 4 g of silicon dioxide, and 26 g of lactose were evenly mixed to obtain a octacosanol composition for later use.
[0027] The obtained octacosanol composition powder was directly filled to prepare octacosanol composition capsules, each capsule containing 360 mg of the octacosanol composition.
Embodiment 2
[0028] Embodiment 2 Rat comparative test
[0029] Take 40 Wistar rats, half male and half male, first adapted to feeding for 3 days, weighed and divided into groups. Randomly divided into 4 groups (group 1-4), 10 in each group. Rats were gavaged once a day for 30 days. On the 31st day of the experiment, anesthetized by intraperitoneal injection of 10% chloral hydrate, blood was collected from the abdominal aorta, centrifuged (3000r / min) for 15min, and serum was separated and prepared.
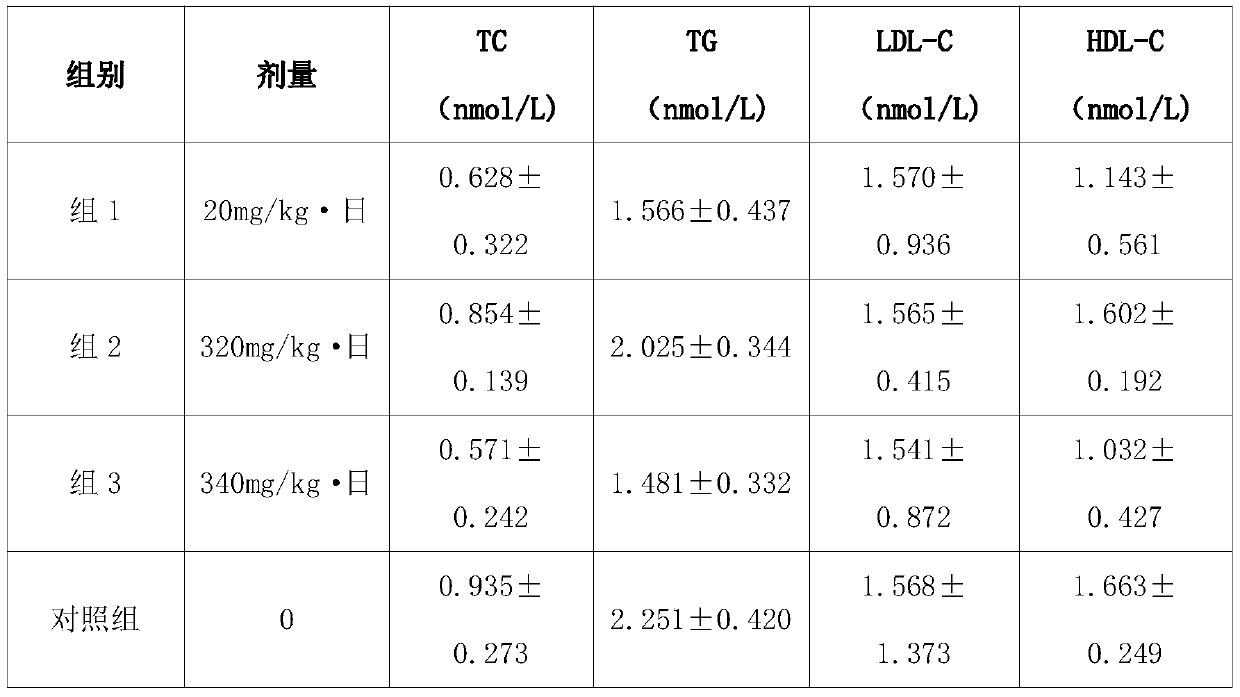
[0030] In this embodiment, the experimental grouping is as follows:
[0031] Group 1: octacosanol;
[0032] Group 2: Danshen extract + Astragalus extract (mass ratio 1:1);
[0033] Group 3: Octacosanol + Danshen extract + Astragalus extract (the mass ratio of the three is 1:32:33);
[0034] Control group: Feed only.
[0035] The contents of serum triglyceride (TG), low-density lipoprotein (LDL-C), high-density lipoprotein (HDL-C) and total cholesterol (TC) were measured by automatic bioch...
Embodiment 3 28
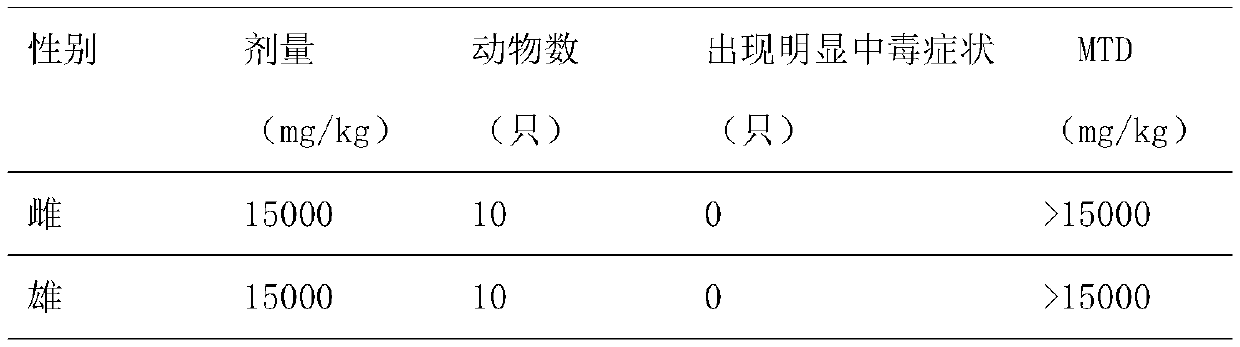
[0039] Example 3 Toxicity detection test of octacosanol composition
[0040] 1. Mouse detection:
[0041] 1.) Materials and methods:
[0042] 1.1) Sample:
[0043] Packed in plastic bottles, stored at room temperature, cool and dark, with a shelf life of 24 months, the test sample is the octacosanol composition capsule obtained in Example 1, and the capsule content is light brown powder.
[0044] 1.2) Test animals:
[0045] 40 healthy ICR mice, half male and half female, weighing 18.3-22.0 g; license number for barrier environment experimental animal facilities: SYXK (Su) 2012-0037; sterilized mouse feed and sterile water free to eat.
[0046] 1.3) Test method:
[0047] Maximum tolerated dose test method. Dose selection and test substance administration: the designed dose is 15000mg / kg b.wt., weigh 15000mg of sample and add purified water to 40ml to prepare the test substance. After fasting overnight, the mice were given two oral gavages, with an interval of 4 hours in b...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Maximum tolerated dose | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap